Biologics in Pregnancy and Lactation: How Aggressive Can We Safely Be?

Abstract
Biologic therapies have transformed the management of chronic inflammatory and autoimmune diseases by targeting specific components of the immune system. Their use has led to significant improvements in disease control, quality of life, and long term outcomes for conditions such as rheumatoid arthritis, inflammatory bowel disease, psoriasis, and other immune mediated disorders. However, the increasing use of biologic agents among women of reproductive age has raised important questions regarding their safety during pregnancy and lactation. Clinicians are often faced with complex decisions that require balancing the risks of maternal disease activity against potential fetal and neonatal exposure to these therapies.
This review evaluates current evidence on the use of biologic therapies during pregnancy and breastfeeding, drawing from recent observational studies, pregnancy registries, pharmacovigilance data, and established clinical guidelines. Particular attention is given to commonly used biologic classes, including tumor necrosis factor inhibitors, interleukin inhibitors, and other targeted immunomodulators. The analysis aims to clarify safety profiles, patterns of placental transfer, and implications for fetal development and neonatal outcomes.
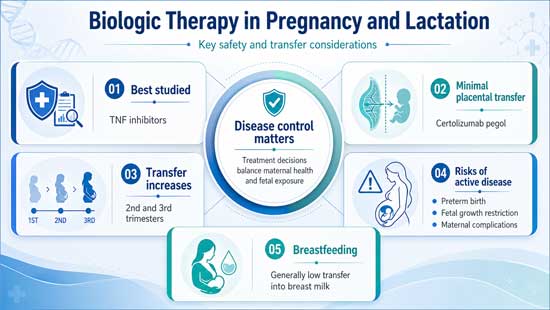
Among the biologic agents, tumor necrosis factor inhibitors remain the most extensively studied in pregnancy. Available evidence suggests that most anti tumor necrosis factor agents demonstrate acceptable safety profiles when used during pregnancy, particularly in the first and second trimesters. Placental transfer of these agents is limited in early pregnancy due to the immature expression of Fc receptors, although transfer increases in the third trimester. Agents such as certolizumab pegol, which lacks an Fc fragment, show minimal placental passage even in later stages of pregnancy, making them an attractive option for continued therapy when clinically indicated. Large registry studies and cohort analyses have not demonstrated a marked increase in major congenital malformations, although some variability exists in reported outcomes related to preterm birth and low birth weight, often confounded by underlying disease severity.
In contrast, newer biologic therapies targeting interleukin pathways, including interleukin 17 and interleukin 23 inhibitors, have more limited safety data in pregnancy. While preclinical studies and early clinical reports have not identified clear safety signals, the absence of robust longitudinal data necessitates cautious use. In most cases, these agents are either discontinued prior to conception or avoided during pregnancy unless disease severity warrants continued therapy and alternative options are inadequate.
The management of inflammatory disease during pregnancy must consider the well established risks associated with uncontrolled maternal disease. Active disease has been linked to adverse pregnancy outcomes, including miscarriage, preterm delivery, fetal growth restriction, and increased maternal morbidity. Consequently, maintaining disease remission or low disease activity is a central objective in the care of pregnant patients with autoimmune conditions. In many cases, the benefits of continuing biologic therapy outweigh the potential risks associated with fetal exposure, particularly in individuals with severe or refractory disease.
Lactation presents additional considerations, although available data are generally reassuring for many biologic agents. Due to their large molecular size, most biologics are present in breast milk at very low concentrations, and systemic absorption by the infant is expected to be minimal due to degradation within the gastrointestinal tract. Studies evaluating infant outcomes during breastfeeding have not demonstrated major adverse effects, although long term data remain limited. Clinical guidelines increasingly support the continuation of certain biologic therapies during breastfeeding when clinically indicated.
Clinical decision making in this context requires an individualized, patient centered approach. Factors such as disease type and severity, prior treatment response, timing of pregnancy, and patient preferences must all be carefully considered. Shared decision making is essential, with clear communication regarding the known benefits, potential risks, and areas of uncertainty associated with biologic therapy during pregnancy and lactation.
In conclusion, current evidence supports the cautious but often necessary use of biologic therapies in pregnant and breastfeeding patients with inflammatory diseases. Tumor necrosis factor inhibitors have the most established safety profiles, while newer agents require further investigation. Effective disease control remains a critical determinant of both maternal and fetal outcomes, and therapeutic decisions should prioritize maintaining disease stability while minimizing unnecessary risk. This review provides a framework for clinicians to navigate these complex decisions, emphasizing evidence based practice, individualized care, and ongoing evaluation as new data emerge.
Introduction
Pregnancy introduces complex clinical considerations for women with chronic inflammatory diseases who require ongoing biologic therapy. Determining the appropriate level of treatment intensity during pregnancy and lactation remains a critical challenge in maternal fetal medicine, rheumatology, gastroenterology, and dermatology. Clinicians must carefully balance the need for sustained maternal disease control with the potential risks of fetal and neonatal exposure to immunomodulatory agents.
Biologic therapies have fundamentally transformed the management of immune mediated inflammatory conditions such as rheumatoid arthritis, inflammatory bowel disease, psoriasis, and ankylosing spondylitis. These agents are designed to selectively target key components of the immune response, including tumor necrosis factor, interleukins, integrins, and other signaling pathways involved in chronic inflammation. Their ability to achieve sustained remission and prevent disease progression has remarkably improved quality of life and long term outcomes for many patients. As a result, an increasing number of women of reproductive age are conceiving while receiving these therapies or require their continuation during pregnancy to maintain disease stability.
The clinical decision making process surrounding biologic use in pregnancy has become more nuanced as the evidence base has expanded. Pregnancy registries, observational cohort studies, and post marketing surveillance data have provided important insights into the safety profiles of various biologic agents. While early concerns centered on potential teratogenicity and immunosuppression in the developing fetus, current data suggest that many biologics, particularly tumor necrosis factor inhibitors, have relatively favorable safety profiles when used appropriately. However, important differences exist between agents in terms of placental transfer, timing of exposure, and pharmacokinetics.
Placental transport of biologic agents is largely mediated by the neonatal Fc receptor, with minimal transfer occurring during the first trimester and increasing significantly in the second and third trimesters. This has important clinical implications, as fetal exposure is generally limited during the period of organogenesis but may be substantial later in pregnancy, particularly with monoclonal antibodies that have high affinity for Fc receptors. Agents with modified structures or shorter half lives may exhibit reduced placental passage, which can influence therapeutic selection and timing of discontinuation when appropriate.
Maintaining maternal disease control is a central priority in pregnancy management. Active inflammatory disease is consistently associated with adverse obstetric outcomes, including preterm birth, fetal growth restriction, low birth weight, and increased rates of operative delivery. In conditions such as inflammatory bowel disease and rheumatoid arthritis, disease flares during pregnancy can also lead to increased hospitalization and maternal morbidity. Consequently, discontinuation of effective therapy solely due to pregnancy concerns may result in greater overall risk than continued treatment.
At the same time, potential risks related to fetal and neonatal exposure to biologic agents must be carefully considered. Although most available data do not demonstrate a significant increase in major congenital anomalies with commonly used biologics, questions remain regarding subtle effects on immune system development, infection risk in early life, and long term health outcomes. Infants exposed to biologic agents in utero, particularly during late pregnancy, may have detectable drug levels at birth, which has implications for vaccination strategies, especially with live attenuated vaccines.
Lactation introduces an additional layer of complexity. Many biologic agents are large protein molecules with limited transfer into breast milk and poor oral bioavailability in the infant, suggesting a low likelihood of systemic absorption. Available evidence generally supports the compatibility of several biologics with breastfeeding, although data remain limited for newer agents. Shared decision making is therefore essential, taking into account maternal preferences, disease severity, and the specific pharmacologic profile of the therapy.
This review synthesizes current evidence on the safety and clinical use of biologic therapies during pregnancy and lactation. It aims to provide practical, evidence based guidance for clinicians navigating treatment decisions in this population. Key considerations include preconception counseling, risk stratification, selection of appropriate agents, timing of therapy continuation or discontinuation, and coordination of care across specialties.
In summary, the management of biologic therapy in pregnancy requires a careful and individualized approach that prioritizes both maternal and fetal well being. Advances in clinical research and real world data have improved confidence in the use of several biologic agents, yet important knowledge gaps remain. Ongoing studies and long term follow up data will be essential to refine clinical guidelines and optimize outcomes for both mother and child in this increasingly common clinical scenario.
Overview of Biologic Medications
Biologic medications represent a class of drugs derived from living organisms that target specific components of the immune system. Unlike traditional disease-modifying antirheumatic drugs (DMARDs) that broadly suppress immune function, biologics work by blocking specific inflammatory pathways.
The most commonly used biologics during pregnancy belong to several major categories. TNF inhibitors include adalimumab, etanercept, infliximab, certolizumab pegol, and golimumab. These medications block TNF-alpha, a key inflammatory protein involved in many autoimmune conditions. Interleukin inhibitors target various interleukin pathways, including IL-1, IL-6, IL-12/23, IL-17, and IL-23. B-cell depleting agents like rituximab target CD20 on B lymphocytes.
Understanding the pharmacokinetic properties of different biologics helps predict their potential effects during pregnancy. Most biologics are large proteins that do not cross the placenta easily during early pregnancy. However, active transport of IgG antibodies across the placenta increases dramatically after 20 weeks of gestation. This timing difference has important implications for treatment planning.
The half-life of different biologics varies considerably, affecting how long they remain in maternal and fetal circulation. Certolizumab pegol has unique properties among TNF inhibitors because it lacks the Fc portion of the antibody, resulting in minimal placental transfer even in later pregnancy.
Safety Data for Specific Biologic Classes
TNF Inhibitors
TNF inhibitors have the most extensive pregnancy safety data among biologic medications. Multiple pregnancy registries and observational studies have tracked thousands of pregnancies exposed to these medications.
Adalimumab, one of the most commonly prescribed biologics, has been studied in over 2000 pregnancies. Registry data shows no increased risk of major birth defects compared to disease-matched controls or the general population. The rate of major malformations ranges from 3-5%, which falls within the background risk for the general population of 2-4%.
Etanercept has similarly reassuring data from pregnancy registries. Studies of over 1000 exposed pregnancies show no increased risk of birth defects or adverse pregnancy outcomes. The fusion protein structure of etanercept may result in less placental transfer compared to monoclonal antibodies, though this theoretical advantage has not translated into clinically meaningful differences in safety outcomes.
Infliximab has been associated with slightly higher rates of placental transfer due to its IgG1 structure, but extensive registry data shows no increased risk of birth defects. Some studies suggest a possible increased risk of infections in infants exposed to infliximab in utero, but this finding has not been consistent across all studies.
Certolizumab pegol offers unique advantages during pregnancy due to its pegylated Fab fragment structure. Multiple studies confirm minimal placental transfer, with undetectable or very low levels in cord blood even with continued use throughout pregnancy. This pharmacologic profile makes certolizumab particularly attractive for pregnant patients requiring TNF inhibition.
Golimumab has more limited pregnancy data due to its more recent approval, but available studies suggest a safety profile similar to other TNF inhibitors. Registry data includes several hundred pregnancies with no obvious safety signals.
Interleukin Inhibitors
Interleukin inhibitors represent a diverse group of biologics with varying amounts of pregnancy safety data. The IL-1 inhibitor anakinra has limited pregnancy data but shows no obvious safety signals in small case series. Its short half-life and different mechanism of action may offer theoretical advantages, but insufficient data exists for firm conclusions.
IL-6 inhibitors like tocilizumab have more substantial pregnancy data, particularly in patients with rheumatoid arthritis. Registry studies of several hundred pregnancies show no increased risk of birth defects. However, some reports suggest possible effects on fetal growth, though these findings require confirmation in larger studies.
Newer IL-17 inhibitors including secukinumab, ixekizumab, and brodalumab have very limited pregnancy data. Case reports and small case series suggest no obvious safety signals, but the numbers are too small for definitive conclusions. These medications are increasingly used for psoriasis and psoriatic arthritis in women of childbearing age, making pregnancy data collection a priority.
IL-23 inhibitors like ustekinumab, guselkumab, and risankizumab also have limited pregnancy data. Ustekinumab has the most information with registry data on over 100 pregnancies showing no increased risk of major malformations. However, the sample sizes remain small for rare outcomes.
Other Biologic Classes
B-cell depleting agents like rituximab present unique considerations during pregnancy. The medication can cause prolonged B-cell depletion in both mother and infant, potentially affecting immune system development. Registry data suggests no increased risk of birth defects, but concerns exist about infection risk and vaccine responses in exposed infants.
Abatacept, a T-cell costimulation modulator, has limited pregnancy data but no obvious safety signals in available studies. The medication’s mechanism of action raises theoretical concerns about immune system development, but clinical data is insufficient for firm conclusions.
Pregnancy Outcomes and Registry Data
Pregnancy registries provide the most robust safety data for biologic medications during pregnancy. These voluntary reporting systems collect data on pregnancy outcomes in women exposed to specific medications, comparing results to background rates in the general population and disease-matched controls.
The OTIS (Organization of Teratology Information Specialists) autoimmune diseases in pregnancy study represents one of the largest prospective cohorts. This ongoing study has followed thousands of pregnancies in women with autoimmune diseases, including those exposed to various biologic therapies. Results consistently show no increased risk of major birth defects with TNF inhibitor exposure.
Manufacturer-sponsored registries for individual medications provide additional safety data. These registries often include larger numbers of exposed pregnancies but may have different ascertainment methods and comparison groups. Despite these limitations, the consistency of findings across multiple registries strengthens confidence in the overall safety profile.
Meta-analyses of registry data and published studies provide the highest level of evidence available for pregnancy safety. A recent meta-analysis of TNF inhibitor use in pregnancy included over 3000 exposed pregnancies and found no increased risk of major malformations, spontaneous abortion, or preterm birth compared to controls.
Registry data does have important limitations that affect interpretation. Underreporting of adverse outcomes, particularly for newer medications, may bias results. Confounding by indication remains a concern, as women with more severe disease may be more likely to continue biologic therapy during pregnancy. Small sample sizes for newer medications limit the ability to detect rare adverse outcomes.
Disease-Specific Considerations
Rheumatoid Arthritis
Rheumatoid arthritis affects approximately 1% of women of childbearing age, making pregnancy considerations crucial for treatment planning. The disease often improves during pregnancy due to hormonal changes, but approximately 25% of women experience persistent or worsening symptoms requiring continued treatment.
Uncontrolled rheumatoid arthritis during pregnancy is associated with increased risks of preterm birth, low birth weight, and cesarean delivery. These risks must be balanced against potential medication effects when making treatment decisions. Studies consistently show that women with well-controlled disease have pregnancy outcomes similar to healthy controls.
TNF inhibitors represent first-line biologic therapy for pregnant women with rheumatoid arthritis requiring biologic treatment. Certolizumab pegol is often preferred due to minimal placental transfer, but other TNF inhibitors are reasonable alternatives based on individual patient factors and previous response.
Timing of medication discontinuation varies based on disease activity and patient preferences. Some women can safely discontinue biologics in early pregnancy if disease activity is low. Others require continued treatment throughout pregnancy to maintain disease control.
Inflammatory Bowel Disease
Inflammatory bowel disease, including Crohn’s disease and ulcerative colitis, commonly affects women during their reproductive years. Active disease during pregnancy poses substantial risks including increased rates of preterm birth, low birth weight, and pregnancy complications.
The principle of treating through pregnancy has gained acceptance in inflammatory bowel disease management. Maintaining clinical remission often requires continued biologic therapy throughout pregnancy. Registry data supports the safety of this approach, with pregnancy outcomes in women on anti-TNF therapy similar to those in remission without medication.
Adalimumab and infliximab have the most extensive safety data in inflammatory bowel disease pregnancy registries. Both medications effectively maintain remission during pregnancy with acceptable safety profiles. Vedolizumab, an integrin inhibitor, has more limited pregnancy data but shows no obvious safety signals in available studies.
The timing of last biologic dose before delivery requires individualized decision-making. Some practitioners discontinue medication 6-10 weeks before delivery to minimize infant exposure, while others continue treatment throughout pregnancy if disease activity requires it.
Psoriasis and Psoriatic Arthritis
Psoriasis affects 2-3% of the population, with psoriatic arthritis developing in approximately 30% of patients. Unlike rheumatoid arthritis, psoriasis typically worsens during pregnancy, making treatment decisions more challenging.
Topical therapies and phototherapy represent first-line treatments during pregnancy, but severe disease may require systemic therapy. Traditional systemic medications like methotrexate are contraindicated during pregnancy, making biologics important treatment options.
TNF inhibitors are the best-studied biologics for psoriasis during pregnancy. Registry data supports their safety profile, though studies specifically in pregnant women with psoriasis are more limited than in rheumatoid arthritis populations.
Newer biologics like IL-17 and IL-23 inhibitors are increasingly used for psoriasis but have very limited pregnancy data. Case reports suggest no obvious safety signals, but larger studies are needed for definitive conclusions.
The impact of untreated severe psoriasis on pregnancy outcomes is less well studied than other inflammatory conditions. However, the systemic inflammation associated with severe disease may contribute to pregnancy complications, supporting treatment of severe cases.
Ankylosing Spondylitis
Ankylosing spondylitis affects men more commonly than women, but approximately 30% of cases occur in women. The disease typically begins in young adulthood, making pregnancy considerations relevant for many patients.
Disease activity often remains stable or improves modestly during pregnancy, but some women require continued biologic therapy for symptom control. TNF inhibitors represent the primary biologic treatment option, with similar safety data to other inflammatory conditions.
The physical changes of pregnancy can complicate disease management, as growing abdominal size and changes in posture may worsen symptoms. Maintaining mobility and function during pregnancy requires balancing disease control with medication safety considerations.
Lactation and Biologic Therapy
Breastfeeding safety represents another important consideration for women using biologic therapy. Most biologics are large proteins that are poorly absorbed from the infant’s gastrointestinal tract, suggesting minimal systemic exposure even if present in breast milk.
Transfer of biologics into breast milk varies by medication but is generally low. TNF inhibitors are present in breast milk in small amounts, but infant serum levels are typically undetectable or very low. The theoretical risk of immunosuppression in breastfed infants must be balanced against the well-established benefits of breastfeeding.
Professional society guidelines generally support breastfeeding in women using compatible biologic medications. The American College of Rheumatology and European League Against Rheumatism both state that most biologics are compatible with breastfeeding based on available evidence.
Individual patient counseling should address the limited long-term data on breastfeeding safety while emphasizing the low levels of drug transfer and potential benefits of continued disease control. Some women may choose to pump and discard breast milk around the time of biologic administration to further minimize infant exposure.
Risk-Benefit Analysis Framework
Clinical decision-making regarding biologic use during pregnancy requires a systematic approach to risk-benefit analysis. This framework should consider maternal disease activity, pregnancy complications from uncontrolled disease, medication safety data, and individual patient factors.
Maternal disease activity assessment includes current symptoms, objective measures of inflammation, and risk of disease flare if treatment is discontinued. High disease activity during pregnancy carries substantial risks for both mother and fetus, often justifying continued biologic therapy.
The timing of pregnancy planning affects treatment decisions. Preconception counseling allows for medication optimization and disease stabilization before pregnancy. Unplanned pregnancies may require different approaches depending on current disease status and medication timing.
Individual patient factors include previous pregnancy outcomes, response to alternative medications, and personal preferences regarding medication use during pregnancy. Some women may prefer to minimize medication exposure even at the cost of increased disease activity, while others prioritize disease control.
| Medication | Pregnancy Risk | Placental Transfer | Registry Data | Lactation Compatibility |
| Adalimumab | Low | Moderate (after 20 weeks) | >2000 pregnancies | Compatible |
| Etanercept | Low | Low-Moderate | >1000 pregnancies | Compatible |
| Infliximab | Low | High (after 20 weeks) | >1500 pregnancies | Compatible |
| Certolizumab | Low | Minimal | >1000 pregnancies | Compatible |
| Golimumab | Low | Moderate | <500 pregnancies | Probably Compatible |
| Tocilizumab | Low-Moderate | Moderate | <300 pregnancies | Probably Compatible |
| Ustekinumab | Unknown | Unknown | <200 pregnancies | Unknown |
| Secukinumab | Unknown | Unknown | <100 pregnancies | Unknown |
Timing Considerations and Treatment Planning
The timing of biologic administration during pregnancy requires careful consideration of pharmacokinetic properties and placental transfer patterns. Most IgG antibodies show minimal placental transfer during the first 20 weeks of pregnancy, followed by rapidly increasing transfer in the third trimester.
This transfer pattern suggests that early pregnancy exposure carries minimal risk of fetal immunosuppression, while later exposure may result in detectable drug levels in the newborn. However, registry data does not show increased adverse outcomes with continued use throughout pregnancy for most TNF inhibitors.
Some practitioners recommend discontinuing biologics around 20 weeks of gestation to minimize fetal exposure while maintaining disease control during the critical early pregnancy period. Others continue treatment throughout pregnancy if required for disease control, particularly given the reassuring safety data from registries.
Postpartum planning should address the timing of treatment resumption and breastfeeding considerations. Many women can safely resume biologic therapy immediately after delivery if required for disease control. Breastfeeding compatibility allows for continued treatment in most cases.
One particularly memorable case involved a pregnant rheumatologist who continued her own adalimumab throughout pregnancy after extensively reviewing the literature. She joked that she had become her own best patient, applying evidence-based medicine to her personal situation while experiencing the anxiety that comes with any treatment decision during pregnancy. Her healthy delivery of twins reinforced her confidence in the safety data, though she admitted the decision felt different when applied to her own pregnancy rather than counseling patients.
Monitoring and Safety Protocols
Patients using biologic therapy during pregnancy require enhanced monitoring to ensure both maternal and fetal wellbeing. This monitoring should include regular assessment of disease activity, screening for infections, and fetal growth surveillance.
Disease activity monitoring helps ensure that biologic therapy is effectively controlling inflammation. Validated disease activity measures should be used when available, supplemented by clinical assessment and laboratory markers of inflammation.
Infection screening becomes particularly important during pregnancy due to the immunosuppressive effects of biologic therapy. Patients should receive appropriate vaccinations before pregnancy when possible and avoid live vaccines during pregnancy.
Fetal monitoring should follow standard obstetric guidelines with additional attention to growth and development. While registry data is reassuring, individual monitoring helps detect potential complications early.
Coordination between specialists is essential for optimal outcomes. Rheumatologists, gastroenterologists, or dermatologists should work closely with obstetricians to ensure comprehensive care throughout pregnancy.
Challenges and Limitations in Current Evidence
Despite the growing body of evidence supporting biologic safety during pregnancy, several important limitations affect clinical decision-making. Registry data, while valuable, has inherent limitations including voluntary reporting, potential selection bias, and limited long-term follow-up.
The rarity of major birth defects means that even large registries may lack sufficient power to detect small increases in risk. Background rates of major malformations range from 2-4% in the general population, requiring very large sample sizes to detect clinically meaningful increases.
Confounding by indication represents a persistent challenge in observational studies. Women with more severe disease may be more likely to continue biologic therapy during pregnancy, making it difficult to separate medication effects from disease effects on pregnancy outcomes.
Long-term developmental outcomes in children exposed to biologics in utero remain largely unknown. Most registry studies focus on birth outcomes with limited follow-up beyond infancy. Potential effects on immune system development, growth, or cognitive development require longer follow-up studies.
Publication bias may affect the literature, as case reports of adverse outcomes may be more likely to be published than routine outcomes. This bias could potentially overestimate risks associated with biologic use during pregnancy.
Future Directions and Research Needs
Several important areas require additional research to improve evidence-based decision-making for biologic use during pregnancy. Long-term follow-up studies of children exposed to biologics in utero are needed to assess developmental outcomes and immune system function.
Newer biologic medications require larger pregnancy registries to establish safety profiles comparable to TNF inhibitors. IL-17 inhibitors, IL-23 inhibitors, and other novel biologics need systematic data collection to guide clinical decision-making.
Pharmacokinetic studies during pregnancy could help optimize dosing and timing of biologic therapy. Changes in drug clearance and distribution during pregnancy may affect optimal dosing regimens.
Biomarker research might identify predictors of disease flare during pregnancy, helping to guide decisions about continuing or discontinuing biologic therapy. Genetic markers or inflammatory biomarkers could potentially stratify risk and personalize treatment decisions.
Comparative effectiveness research could help identify optimal biologic choices during pregnancy. While current evidence suggests similar safety profiles across TNF inhibitors, subtle differences might emerge with larger datasets or longer follow-up.
Clinical Practice Guidelines and Recommendations
Major professional societies have developed guidelines for biologic use during pregnancy based on available evidence. These guidelines generally support the continued use of compatible biologics when required for disease control.
The American College of Rheumatology 2020 guidelines recommend that TNF inhibitors can be continued during pregnancy for women with rheumatoid arthritis, psoriatic arthritis, or ankylosing spondylitis when benefits outweigh risks. Certolizumab is preferred when possible due to minimal placental transfer.
European League Against Rheumatism guidelines similarly support TNF inhibitor use during pregnancy when required for disease control. The guidelines emphasize the importance of preconception counseling and shared decision-making between patients and providers.
Gastroenterology societies generally recommend continuing anti-TNF therapy throughout pregnancy for women with inflammatory bowel disease to maintain remission. The risks of active disease are considered to outweigh potential medication risks based on available evidence.
These guidelines emphasize that treatment decisions should be individualized based on disease activity, patient preferences, and available alternatives. Regular updates to guidelines reflect the evolving evidence base and changing clinical practice.
Economic and Healthcare System Considerations
The cost-effectiveness of biologic therapy during pregnancy involves complex calculations of medication costs, monitoring expenses, and potential savings from prevented complications. Uncontrolled inflammatory disease during pregnancy can result in substantial healthcare costs from pregnancy complications and poor outcomes.
Access to biologic therapy during pregnancy varies significantly across healthcare systems and insurance plans. Prior authorization requirements and formulary restrictions may limit access to preferred medications, potentially affecting clinical decision-making.
The need for enhanced monitoring during pregnancy adds to healthcare costs but may be offset by improved outcomes. Coordination between specialists requires additional resources but likely improves care quality and patient satisfaction.
Healthcare provider education regarding biologic safety during pregnancy remains variable. Many providers lack familiarity with the growing evidence base, potentially leading to unnecessary medication discontinuation and disease flares.
Global Perspectives and Regulatory Considerations
Regulatory approval and labeling of biologics for pregnancy use varies across countries and may not reflect current evidence. Many medications continue to carry pregnancy category warnings that do not align with registry data and clinical experience.
International pregnancy registries provide opportunities for larger datasets and diverse populations but face challenges with standardization and data quality. Harmonization of data collection methods could improve the evidence base for regulatory decision-making.
Cultural attitudes toward medication use during pregnancy vary significantly and may influence treatment decisions independent of medical evidence. Provider education and patient counseling must account for these cultural factors to optimize shared decision-making.
Resource limitations in some healthcare systems may limit access to biologic therapy or adequate monitoring during pregnancy. These disparities highlight the need for cost-effective approaches and healthcare system strengthening.
Patient Education and Counseling Strategies
Effective patient education regarding biologic use during pregnancy requires clear communication about risks, benefits, and uncertainties. Patients need accurate information to participate in shared decision-making while avoiding unnecessary anxiety.
Risk communication should use natural frequencies rather than relative risks to help patients understand absolute risk levels. For example, stating that major birth defects occur in 3-4 out of 100 pregnancies both with and without medication exposure may be more meaningful than relative risk statistics.
Decision aids and educational materials can help patients process complex information and clarify their values regarding treatment choices. These tools should present balanced information about risks and benefits while acknowledging areas of uncertainty.
Support groups and patient organizations provide valuable resources for women facing these treatment decisions. Connecting patients with others who have faced similar decisions can provide emotional support and practical insights.
The question of how aggressive biologic therapy can safely be during pregnancy and lactation has evolved considerably as evidence has accumulated from pregnancy registries and clinical experience. Current data supports the safety of TNF inhibitors during pregnancy when required for maternal disease control, with no increased risk of major birth defects demonstrated across thousands of pregnancies.
The principle that has emerged from this evidence base emphasizes maintaining disease control during pregnancy while using medications with the best safety profiles available. Uncontrolled inflammatory disease poses substantial risks to both mother and fetus that often outweigh theoretical medication risks.
Certolizumab pegol offers unique advantages among TNF inhibitors due to minimal placental transfer, but other anti-TNF agents represent reasonable alternatives based on individual patient factors. Newer biologics require additional safety data before firm recommendations can be made, though early evidence suggests no obvious safety signals.
Clinical decision-making should involve careful risk-benefit analysis considering disease activity, patient preferences, and individual circumstances. Preconception counseling allows for optimal treatment planning, while unplanned pregnancies require individualized approaches based on current disease status.
The growing body of evidence supports a more aggressive approach to maintaining biologic therapy during pregnancy when clinically indicated. This represents a substantial shift from earlier conservative approaches that often resulted in disease flares and poor pregnancy outcomes.
Key Takeaways
Healthcare providers can safely maintain biologic therapy during pregnancy for women with inflammatory diseases when benefits outweigh risks. TNF inhibitors have the most extensive safety data and represent first-line biologic options during pregnancy. Certolizumab pegol offers theoretical advantages due to minimal placental transfer but other TNF inhibitors are reasonable alternatives.
Disease control during pregnancy should be prioritized, as uncontrolled inflammation poses substantial risks to maternal and fetal health. Preconception counseling allows for treatment optimization and should be standard care for women of childbearing age using biologic therapy.
Newer biologics including IL-17 and IL-23 inhibitors require additional safety data before routine use during pregnancy can be recommended. Most biologics appear compatible with breastfeeding based on limited transfer into breast milk and poor infant absorption.
Patient education and shared decision-making are essential components of care, requiring clear communication about risks, benefits, and areas of uncertainty. Regular monitoring during pregnancy helps ensure both disease control and early detection of any complications.

Frequently Asked Questions
Q: Are TNF inhibitors safe during pregnancy?
A: Current evidence from pregnancy registries including thousands of pregnancies shows no increased risk of major birth defects with TNF inhibitor use during pregnancy. The medications appear safe when required for disease control.
Q: Which biologic is safest during pregnancy?
A: Certolizumab pegol has unique pharmacologic properties resulting in minimal placental transfer, making it theoretically safer than other biologics. However, safety data for adalimumab, etanercept, and infliximab is also reassuring.
Q: Should I stop my biologic if I become pregnant?
A: This decision should be made in consultation with your healthcare provider based on your disease activity, medication type, and individual circumstances. Many women can safely continue biologic therapy throughout pregnancy.
Q: Can I breastfeed while taking a biologic?
A: Most biologics appear compatible with breastfeeding based on limited transfer into breast milk and poor absorption by infants. TNF inhibitors are generally considered safe during breastfeeding.
Q: What about newer biologics like IL-17 inhibitors?
A: Newer biologics have limited pregnancy safety data compared to TNF inhibitors. While early reports are reassuring, larger studies are needed before routine use during pregnancy can be recommended.
Q: Will my baby be immunocompromised if I take biologics during pregnancy?
A: Registry data does not show increased infection rates in infants exposed to TNF inhibitors during pregnancy. Some medications may cross the placenta but do not appear to cause clinically meaningful immunosuppression.
Q: When should I restart my biologic after delivery?
A: Most women can safely resume biologic therapy immediately after delivery if required for disease control. Breastfeeding compatibility allows for continued treatment in most cases.
Q: Do I need extra monitoring during pregnancy if I take biologics?
A: Enhanced monitoring is recommended including regular disease activity assessment, infection screening, and standard fetal monitoring. Coordination between specialists helps ensure optimal care.
References
Andreoli, L., Bertsias, GK., Agmon-Levin, N., Brown, S., Cervera, R., Costedoat-Chalumeau, N., … & Tincani, A. (2017). EULAR recommendations for women’s health and the management of family planning, assisted reproduction, pregnancy and menopause in patients with systemic lupus erythematosus and/or antiphospholipid syndrome. Annals of the Rheumatic Diseases, 76(3), 476-485.
Clowse, MEB., Förger, F., Hwang, C., Thorp, J., Dolhain, RJEM., van Tubergen, A., … & Viktil, KK. (2018). Minimal to no transfer of certolizumab pegol into breast milk: results from CRADLE, a prospective, postmarketing, multicentre, pharmacokinetic study. Annals of the Rheumatic Diseases, 77(6), 835-840.
Götestam Skorpen, C., Hoeltzenbein, M., Tincani, A., Fischer-Betz, R., Elefant, E., Chambers, C., … & Østensen, M. (2016). The EULAR points to consider for use of antirheumatic drugs before pregnancy, and during pregnancy and lactation. Annals of the Rheumatic Diseases, 75(5), 795-810.
Mahadevan, U., Wolf, DC., Dubinsky, M., Cortot, A., Lee, SD., Siegel, CA., … & Sandborn, WJ. (2013). Placental transfer of anti-tumor necrosis factor agents in pregnant patients with inflammatory bowel disease. Clinical Gastroenterology and Hepatology, 11(3), 286-292.
Mariette, X., Förger, F., Abraham, B., Flynn, AD., Moltó, A., Flipo, RM., … & Chakravarty, E. (2018). Lack of placental transfer of certolizumab pegol during pregnancy: results from CRIB, a prospective, postmarketing, pharmacokinetic study. Annals of the Rheumatic Diseases, 77(2), 228-233.
Matro, R., Martin, CF., Wolf, D., Shah, SA., Mahadevan, U. (2013). Exposure concentrations of infants breastfed by women receiving biologic therapies for inflammatory bowel diseases and effects of breastfeeding on infections and development. Gastroenterology, 145(5), 1058-1067.
Nielsen, OH., Loftus, EV Jr., Jess, T. (2013). Safety of TNF-α inhibitors during IBD pregnancy: a systematic review. BMC Medicine, 11, 174.
Sammaritano, LR., Bermas, BL., Chakravarty, EE., Chambers, C., Clowse, MEB., Lockshin, MD., … & Ostensen, M. (2020). 2020 American College of Rheumatology guideline for the management of reproductive health in rheumatic and musculoskeletal diseases. Arthritis & Rheumatism, 72(4), 529-556.
Verstappen, SMM., King, Y., Watson, KD., Symmons, DPM., Hyrich, KL. (2011). Anti-TNF therapies and pregnancy: outcome of 130 pregnancies in the British Society for Rheumatology Biologics Register. Annals of the Rheumatic Diseases, 70(5), 823-826.
Weber-Schoendorfer, C., Chambers, C., Wacker, E., Beghin, D., Bernard, N., Shechtman, S., … & Schaefer, C. (2015). Pregnancy outcome after methotrexate treatment for rheumatic disease prior to or during early pregnancy: a prospective multicenter cohort study. Arthritis & Rheumatology, 67(7), 1846-1853.
Recent Articles


Integrative Perspectives on Cognition, Emotion, and Digital Behavior

Sleep-related:
Longevity/Nutrition & Diet:
Philosophical / Happiness / Social:
Other:
Modern Mind Unveiled
Developed under the direction of David McAuley, Pharm.D., this collection explores what it means to think, feel, and connect in the modern world. Drawing upon decades of clinical experience and digital innovation, Dr. McAuley and the GlobalRPh initiative translate complex scientific ideas into clear, usable insights for clinicians, educators, and students.
The series investigates essential themes—cognitive bias, emotional regulation, digital attention, and meaning-making—revealing how the modern mind adapts to information overload, uncertainty, and constant stimulation.
At its core, the project reflects GlobalRPh’s commitment to advancing evidence-based medical education and clinical decision support. Yet it also moves beyond pharmacotherapy, examining the psychological and behavioral dimensions that shape how healthcare professionals think, learn, and lead.
Through a synthesis of empirical research and philosophical reflection, Modern Mind Unveiled deepens our understanding of both the strengths and vulnerabilities of the human mind. It invites readers to see medicine not merely as a science of intervention, but as a discipline of perception, empathy, and awareness—an approach essential for thoughtful practice in the 21st century.
The Six Core Themes
I. Human Behavior and Cognitive Patterns
Examining the often-unconscious mechanisms that guide human choice—how we navigate uncertainty, balance logic with intuition, and adapt through seemingly irrational behavior.
II. Emotion, Relationships, and Social Dynamics
Investigating the structure of empathy, the psychology of belonging, and the influence of abundance and selectivity on modern social connection.
III. Technology, Media, and the Digital Mind
Analyzing how digital environments reshape cognition, attention, and identity—exploring ideas such as gamification, information overload, and cognitive “nutrition” in online spaces.
IV. Cognitive Bias, Memory, and Decision Architecture
Exploring how memory, prediction, and self-awareness interact in decision-making, and how external systems increasingly serve as extensions of thought.
V. Habits, Health, and Psychological Resilience
Understanding how habits sustain or erode well-being—considering anhedonia, creative rest, and the restoration of mental balance in demanding professional and personal contexts.
VI. Philosophy, Meaning, and the Self
Reflecting on continuity of identity, the pursuit of coherence, and the construction of meaning amid existential and informational noise.
Keywords
Cognitive Science • Behavioral Psychology • Digital Media • Emotional Regulation • Attention • Decision-Making • Empathy • Memory • Bias • Mental Health • Technology and Identity • Human Behavior • Meaning-Making • Social Connection • Modern Mind
Video Section 

