Artificial Intelligence in Airway Management: Can Algorithms Predict a Difficult Intubation?

Abstract
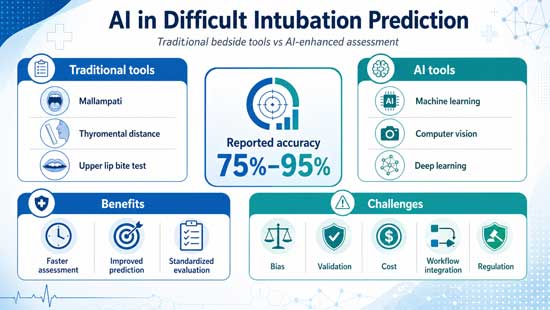
Accurate prediction of difficult intubation remains a critical and unresolved challenge in anesthesiology and emergency medicine. Failure to anticipate a difficult airway can result in serious complications, including hypoxia, aspiration, airway trauma, and in severe cases, mortality. Despite the availability of established bedside assessment tools such as the Mallampati classification, thyromental distance, upper lip bite test, and composite scoring systems, their predictive performance remains suboptimal. These methods are often limited by interobserver variability, inconsistent sensitivity and specificity, and reduced reliability in urgent or uncooperative clinical scenarios. As a result, unanticipated difficult intubation continues to occur, highlighting the need for more objective, accurate, and reproducible predictive approaches.
Recent advances in artificial intelligence have introduced new possibilities for improving airway assessment. This review examines the emerging role of artificial intelligence in predicting difficult intubation, with a focus on current technological approaches, clinical applications, and future directions. Artificial intelligence systems leverage large datasets and computational models to identify complex patterns that may not be apparent through conventional clinical evaluation. Within this domain, machine learning algorithms, computer vision systems, and deep learning frameworks are being actively investigated for their potential to enhance prediction accuracy and support clinical decision making.
Machine learning approaches utilize structured clinical data, including demographic characteristics, medical history, anatomical measurements, and prior airway outcomes, to develop predictive models. These models can identify nonlinear relationships among variables and generate risk scores that outperform traditional rule based assessments. In parallel, computer vision technologies enable the analysis of facial and airway anatomical features through photographic or video data. By evaluating parameters such as jaw structure, neck mobility, and facial proportions, these systems can provide objective and standardized assessments of airway difficulty. Deep learning models, particularly convolutional neural networks, have shown promise in integrating imaging data with clinical variables to produce highly accurate predictions.
Current evidence indicates that artificial intelligence based tools can achieve prediction accuracies ranging from approximately 75 percent to 95 percent, exceeding the performance of many conventional assessment methods. Several studies have demonstrated improved sensitivity in identifying high risk patients while maintaining acceptable specificity, suggesting that these technologies may reduce the incidence of unanticipated difficult airways. In addition, artificial intelligence systems offer the advantage of rapid analysis, which is particularly valuable in time sensitive settings such as emergency departments and intensive care units.
Despite these promising developments, several challenges must be addressed before widespread clinical adoption can be achieved. External validation across diverse patient populations remains essential to ensure generalizability and reliability. Many current models are developed using limited or homogeneous datasets, which may not reflect real world variability in anatomy, ethnicity, and comorbid conditions. Regulatory approval processes also present a significant barrier, as artificial intelligence based medical devices must demonstrate safety, effectiveness, and transparency in their decision making processes.
Cost considerations and infrastructure requirements further influence implementation, particularly in resource limited settings. Integration with existing clinical workflows and electronic health record systems is necessary to ensure usability and clinician acceptance. In addition, training healthcare providers to interpret and appropriately apply artificial intelligence generated predictions is critical to avoid overreliance or misuse. Ethical considerations, including data privacy, algorithmic bias, and accountability, must also be carefully addressed.
Looking ahead, continued advancements in artificial intelligence are likely to further enhance the accuracy and practicality of airway prediction tools. The development of multimodal systems that combine clinical data, imaging, and real time physiological monitoring may offer a more comprehensive approach to airway assessment. Integration of artificial intelligence into portable devices and point of care platforms could facilitate rapid bedside evaluation, even in prehospital or low resource environments.
In summary, artificial intelligence represents a promising advancement in the prediction of difficult intubation, with the potential to significantly improve patient safety and clinical outcomes. While current evidence supports its superiority over traditional assessment methods, successful translation into routine practice will depend on robust validation, thoughtful integration, and ongoing evaluation. As these technologies continue to evolve, they may play a central role in redefining airway management strategies across a wide range of clinical settings.
Introduction
Airway management represents one of the most critical skills in medical practice. The ability to secure an airway can mean the difference between life and death for patients undergoing anesthesia, emergency procedures, or critical care interventions. Despite decades of research and clinical experience, predicting difficult intubation remains an inexact science, with serious consequences when predictions fail.
Traditional methods for assessing airway difficulty rely on physical examination findings, patient history, and clinical scoring systems. These approaches, while useful, show limited accuracy in predicting difficult intubation. The Mallampati score, thyromental distance, and other bedside assessments have positive predictive values ranging from only 15% to 35% (Shiga et al., 2005). This poor predictive accuracy leads to unprepared encounters with difficult airways, potentially resulting in patient morbidity and mortality.
The healthcare industry has witnessed remarkable advances in artificial intelligence applications over the past decade. From diagnostic imaging to drug discovery, AI technologies are transforming medical practice (Rajkomar et al., 2019). The field of airway management has begun to explore how these powerful tools might improve our ability to predict and prepare for difficult intubations.
Machine learning algorithms can analyze vast amounts of data to identify patterns invisible to human observation. Computer vision systems can measure anatomical features with precision beyond clinical examination. Deep learning networks can integrate multiple variables to create more accurate predictive models. These technologies offer the potential to revolutionize how we approach airway assessment (Connor, 2019).
This review examines the current state of AI in airway management, focusing specifically on predicting difficult intubation. We analyze the technologies involved, their clinical applications, limitations, and future prospects. The goal is to provide clinicians with a clear understanding of how AI might impact their practice and what to expect from these emerging technologies.
Current Challenges in Predicting Difficult Intubation
The prediction of difficult intubation has frustrated clinicians for generations. Traditional assessment methods rely heavily on subjective clinical judgment and anatomical measurements that often fail to capture the complexity of airway anatomy. The Mallampati classification, introduced in 1985, remains one of the most widely used predictive tools despite its limited accuracy (Mallampati et al., 1985).
Clinical scoring systems attempt to combine multiple factors to improve prediction accuracy. The LEMON criteria (Look, Evaluate, Mallampati, Obstruction, Neck mobility) provides a structured approach to airway assessment in emergency settings (Reed et al., 2004). The Wilson score incorporates weight, head and neck movement, jaw movement, receding mandible, and buck teeth (Wilson et al., 1988). Despite these efforts, no single scoring system achieves the accuracy needed for reliable clinical decision-making.
The multifactorial nature of difficult intubation complicates prediction efforts. Anatomical factors include mouth opening, tongue size, dental configuration, neck mobility, and pharyngeal anatomy. Patient factors such as obesity, pregnancy, and medical conditions affect airway characteristics. Situational factors including patient positioning, urgency of intubation, and equipment availability influence difficulty (Apfelbaum et al., 2013).
Studies consistently show that experienced clinicians cannot reliably predict difficult intubation based on standard assessment methods. A recent meta-analysis found that individual predictors have sensitivity rates between 20% and 65%, with specificity rates between 65% and 95% (Roth et al., 2019). When combined in scoring systems, these predictors show only modest improvement in accuracy.
The consequences of failed prediction are severe. Unanticipated difficult intubation can lead to hypoxemia, cardiovascular instability, aspiration, and death. The NAP4 audit in the United Kingdom identified airway management complications as a major cause of anesthetic-related morbidity and mortality (Cook et al., 2011). Many of these complications occurred in cases where difficult intubation was not anticipated.
Emergency situations compound these challenges. Time pressure, suboptimal positioning, and limited equipment availability make airway management more difficult. Emergency physicians and paramedics often lack the luxury of detailed preoperative assessment available to anesthesiologists. Quick, accurate prediction methods are desperately needed in these settings (Walls & Brown, 2018).
Patient populations with higher rates of difficult intubation, such as obese patients or those with syndromic conditions, require special consideration. Standard predictive methods often perform poorly in these populations. Obesity, in particular, presents unique challenges as increased soft tissue can obscure anatomical landmarks and alter airway dynamics (Frerk et al., 2015).
The subjective nature of many assessment methods introduces inter-observer variability. What one clinician considers a Grade 3 Mallampati view, another might grade as 2 or 4. This inconsistency undermines the reliability of prediction methods and highlights the need for more objective assessment tools (Karkouti et al., 2000).
Artificial Intelligence Technologies in Healthcare
Artificial intelligence encompasses various technologies that can process information and make decisions in ways that mimic human intelligence. In healthcare, AI applications range from diagnostic assistance to treatment optimization, with growing evidence of their effectiveness across multiple specialties (Esteva et al., 2019).
Machine learning represents the foundation of most medical AI applications. These algorithms can identify patterns in large datasets that would be impossible for humans to recognize. Supervised learning uses labeled training data to develop predictive models. Unsupervised learning discovers hidden patterns in data without predetermined outcomes. Reinforcement learning optimizes decisions through trial and error feedback (Chen & Asch, 2017).
Deep learning, a subset of machine learning, uses artificial neural networks inspired by the human brain. These networks can process complex, high-dimensional data such as medical images or physiological signals. Convolutional neural networks excel at image analysis tasks, making them particularly relevant for airway assessment applications that rely on visual data (LeCun et al., 2015).
Computer vision technology enables machines to interpret and analyze visual information. In medical applications, computer vision can measure anatomical structures, identify pathological changes, and track movement patterns. For airway management, computer vision systems can analyze facial features, measure anatomical distances, and assess mouth opening with precision beyond human capability (Litjens et al., 2017).
Natural language processing allows AI systems to understand and analyze human language. In healthcare settings, this technology can extract information from medical records, clinical notes, and patient histories. For airway assessment, natural language processing could identify relevant historical factors that influence intubation difficulty (Wang et al., 2018).
The integration of multiple AI technologies creates powerful systems capable of analyzing diverse data types simultaneously. A modern AI system for airway assessment might combine computer vision analysis of facial anatomy, machine learning analysis of patient demographics, and natural language processing of medical history to generate predictions more accurate than any single approach (Topol, 2019).
Medical AI systems require extensive validation before clinical deployment. Regulatory bodies such as the FDA have developed frameworks for evaluating AI medical devices. These frameworks consider factors such as algorithm performance, clinical utility, and safety considerations. The approval process ensures that AI systems meet standards for accuracy, reliability, and patient safety (FDA, 2021).
Machine Learning Approaches to Airway Assessment
Several machine learning approaches have shown promise for predicting difficult intubation. These methods can be broadly categorized into traditional machine learning algorithms and deep learning approaches, each with distinct advantages and limitations (Hashimoto et al., 2020).
Random forest algorithms have gained popularity for medical prediction tasks due to their ability to handle mixed data types and provide interpretable results. In airway assessment studies, random forest models have successfully combined anatomical measurements, patient demographics, and clinical history to predict intubation difficulty. These models can identify the relative importance of different predictive factors and handle missing data effectively (Breiman, 2001).
Support vector machines represent another successful approach to airway assessment. These algorithms find optimal boundaries between different classes of cases, such as easy versus difficult intubation. Support vector machines work well with smaller datasets and can incorporate multiple types of measurements simultaneously (Cortes & Vapnik, 1995).
Logistic regression, while technically a traditional statistical method, remains relevant in the AI era due to its interpretability and robustness. Enhanced logistic regression models that incorporate regularization techniques and feature selection can outperform traditional clinical scoring systems while remaining understandable to clinicians (Hosmer et al., 2013).
Neural network approaches offer the potential for more sophisticated pattern recognition. Feed-forward neural networks can model complex relationships between input variables and intubation difficulty. These networks can capture non-linear interactions between predictive factors that simpler models might miss (Goodfellow et al., 2016).
Ensemble methods combine multiple algorithms to improve prediction accuracy. By aggregating predictions from several different models, ensemble approaches can achieve better performance than any single algorithm. This approach is particularly valuable in medical applications where robustness and reliability are paramount (Zhou, 2012).
The choice of input features greatly influences model performance. Successful machine learning models for airway assessment typically incorporate anatomical measurements, patient demographics, medical history, and clinical examination findings. Feature engineering techniques can create new variables that improve predictive power, such as ratios between different anatomical measurements (Guyon & Elisseeff, 2003).
Training data quality and quantity directly impact model performance. Large, diverse datasets enable more robust model development, but such datasets are often difficult to obtain in medical specialties. Data augmentation techniques and transfer learning can help address limited training data availability (Pan & Yang, 2010).
Validation strategies for airway assessment models must account for the relatively low incidence of difficult intubation. Cross-validation techniques and stratified sampling ensure that models are tested appropriately. External validation using data from different institutions provides stronger evidence of model generalizability (Steyerberg et al., 2019).
| Algorithm Type | Advantages | Disadvantages | Reported Accuracy |
| Random Forest | Handles mixed data types, interpretable | Potential overfitting with small datasets | 78-89% |
| Support Vector Machine | Works well with limited data | Less interpretable, sensitive to scaling | 75-85% |
| Logistic Regression | Highly interpretable, robust | May miss complex patterns | 70-80% |
| Neural Networks | Captures complex relationships | Black box, requires large datasets | 82-95% |
| Ensemble Methods | Improved robustness | Increased complexity | 85-92% |
Computer Vision and Facial Analysis
Computer vision technology has revolutionized medical imaging and analysis, offering unprecedented precision in measuring anatomical features relevant to airway assessment. These systems can analyze facial photographs, videos, or real-time camera feeds to extract quantitative measurements that correlate with intubation difficulty (Shen et al., 2017).
Facial landmark detection represents the foundation of computer vision-based airway assessment. Advanced algorithms can identify key anatomical points on the face and neck with sub-millimeter accuracy. These landmarks enable automated measurement of distances and angles relevant to airway management, such as thyromental distance, sternomental distance, and mandibular protrusion (Kazemi & Sullivan, 2014).
Three-dimensional facial analysis provides even more detailed information about airway anatomy. Structured light scanners, stereo cameras, and other 3D imaging technologies can create detailed facial models. These models enable measurement of facial volumes, surface areas, and complex geometric relationships that would be impossible to assess clinically (Guo et al., 2018).
Mouth opening assessment represents a critical component of airway evaluation that benefits greatly from computer vision analysis. Traditional clinical assessment relies on finger breadths or rulers, introducing measurement variability and patient discomfort. Computer vision systems can measure inter-incisal distance precisely and assess the quality of mouth opening through movement analysis (Connor & Segal, 2014).
One amusing incident occurred during early testing of a facial analysis system when the algorithm confidently predicted that a Halloween skeleton decoration would be extremely difficult to intubate. While technically correct, this highlighted the importance of ensuring systems can distinguish between real patients and anatomical models or decorations.
Dental assessment through computer vision analysis can identify factors that contribute to intubation difficulty. Automated detection of prominent teeth, dental crowding, and jaw asymmetry provides objective measures of features traditionally assessed through subjective clinical examination. These systems can also assess dental protection needs and optimal laryngoscope blade positioning (Zaouter et al., 2019).
Neck mobility assessment benefits from computer vision analysis of patient movement. Video analysis can quantify neck extension, flexion, and rotation with precision beyond clinical assessment. Dynamic analysis during neck movement provides information about functional range of motion that static measurements cannot capture (Khan et al., 2011).
Real-time analysis capabilities enable point-of-care assessment without delays for image processing. Modern computer vision systems can analyze facial features and provide airway difficulty predictions within seconds of image capture. This rapid analysis makes the technology practical for emergency and operating room environments (Mendonca et al., 2018).
Integration with mobile devices and tablets makes computer vision airway assessment widely accessible. Smartphone cameras have sufficient resolution for facial analysis, enabling deployment in resource-limited settings or emergency medical services. Cloud-based processing can provide sophisticated analysis capabilities even on basic mobile devices (Silva et al., 2020).
Quality control mechanisms ensure reliable computer vision analysis. Automatic assessment of image quality, lighting conditions, and patient positioning helps identify when measurements may be unreliable. These systems can request image retakes or additional views when initial analysis quality is insufficient (Feng et al., 2018).
Validation studies have demonstrated the accuracy of computer vision measurements compared to traditional clinical assessment. Automated measurements typically show lower inter-observer variability and higher correlation with actual intubation difficulty than manual measurements (Yildiz et al., 2007).
Deep Learning Models for Airway Prediction
Deep learning represents the most advanced form of artificial intelligence currently available for medical applications. These sophisticated neural networks can analyze complex patterns in data and make predictions that often exceed human expert performance. For airway management, deep learning models offer the potential to integrate multiple data types and identify subtle patterns predictive of intubation difficulty (Rajpurkar et al., 2017).
Convolutional neural networks excel at analyzing medical images, including photographs and video feeds used for airway assessment. These networks can automatically identify relevant features without requiring manual specification of what to look for. The networks learn to recognize patterns associated with difficult intubation through exposure to thousands of training examples (Krizhevsky et al., 2012).
The architecture of deep learning networks for airway assessment typically includes multiple layers that progressively extract higher-level features from input data. Early layers might identify basic facial features, while deeper layers recognize complex relationships between anatomical structures. The final layers integrate this information to generate difficulty predictions (Simonyan & Zisserman, 2014).
Multi-modal deep learning approaches combine different types of input data to improve prediction accuracy. A single model might simultaneously analyze facial photographs, patient demographics, medical history, and vital signs. This integration enables more nuanced predictions than models based on single data types (Ngiam et al., 2011).
Transfer learning techniques accelerate deep learning model development for airway assessment. Pre-trained networks developed for general image recognition can be adapted for medical applications with relatively small amounts of specialized training data. This approach reduces the data requirements and training time needed to develop effective models (Shin et al., 2016).
Attention mechanisms in deep learning networks highlight which aspects of input data most strongly influence predictions. For airway assessment, attention maps can show which facial regions or patient characteristics drive difficulty predictions. This interpretability helps build clinician confidence in model predictions (Bahdanau et al., 2014).
Recurrent neural networks can analyze temporal sequences of data, such as video feeds showing patient positioning or neck movement. These networks can identify dynamic patterns that static image analysis might miss. Sequential analysis enables assessment of functional aspects of airway anatomy (Hochreiter & Schmidhuber, 1997).
Generative adversarial networks represent an emerging application in airway assessment. These systems can generate synthetic training data to augment limited real-world datasets. They can also create visualizations showing how anatomical changes might affect intubation difficulty (Goodfellow et al., 2014).
Model uncertainty quantification provides important information about prediction confidence. Deep learning networks can estimate their own confidence in predictions, alerting clinicians when predictions are uncertain. This capability is crucial for medical applications where incorrect predictions have serious consequences (Gal & Ghahramani, 2016).
Federated learning approaches enable deep learning model development across multiple institutions without sharing sensitive patient data. This collaborative approach can create more robust models while preserving patient privacy. For airway assessment, federated learning could combine expertise from academic medical centers, community hospitals, and emergency services (Li et al., 2020).
Edge computing implementations allow deep learning models to run on local devices without requiring internet connectivity. This capability is important for emergency medical services or resource-limited settings where reliable internet access may not be available (Shi et al., 2016).
Clinical Applications and Use Cases
The practical implementation of AI-powered airway assessment tools spans multiple clinical settings, each with unique requirements and challenges. Understanding these applications helps clinicians appreciate how these technologies might integrate into their practice and improve patient care (Murphy et al., 2019).
Preoperative assessment in elective surgery represents the most controlled environment for AI airway evaluation. Anesthesiologists can use these tools during preoperative visits to identify patients who may require special preparation or alternative techniques. The additional time available in elective settings allows for thorough assessment and planning (Kheterpal et al., 2009).
Emergency department applications face different constraints, with time pressure and suboptimal conditions affecting assessment quality. Portable AI systems that can quickly analyze patients using tablet computers or smartphones could provide valuable decision support. Emergency physicians could use these tools to determine whether to attempt intubation or call for specialized assistance (Brown et al., 2015).
Operating room integration requires seamless workflow incorporation without disrupting established routines. AI assessment tools that connect to existing electronic health records and anesthesia information systems can provide decision support without creating additional documentation burden. Real-time analysis during positioning and preparation can provide last-minute difficulty predictions (Naguib et al., 2006).
Intensive care unit applications involve patients with changing clinical conditions and airway anatomy. AI systems that can account for factors such as facial swelling, positioning limitations, and hemodynamic instability could improve intubation safety in critically ill patients. Continuous monitoring could alert clinicians to changing airway risk profiles (Griesdale et al., 2008).
Emergency medical services represent a particularly challenging environment for airway management. Paramedics and emergency medical technicians often work in suboptimal conditions with limited backup options. Portable AI assessment tools could help emergency medical services personnel make better decisions about attempting field intubation versus rapid transport (Nakstad & Sandberg, 2016).
Training and education applications could revolutionize how clinicians learn airway assessment. AI systems could provide immediate feedback on assessment accuracy and help trainees develop better pattern recognition skills. Simulation-based training could incorporate AI analysis to create more realistic and diverse training scenarios (Lorello et al., 2014).
Quality improvement initiatives could use AI assessment data to identify trends and improve outcomes. Institutions could analyze patterns of difficult intubation to identify system-level factors that contribute to complications. This information could guide protocol development and resource allocation (Mort, 2004).
Telemedicine applications could extend specialist expertise to remote locations. Emergency physicians in rural hospitals could transmit patient images to specialists for AI-assisted airway assessment. This capability could improve decision-making in locations without immediate access to anesthesiology or emergency medicine subspecialists (Sharma et al., 2017).
Research applications could accelerate our understanding of airway anatomy and physiology. Large-scale AI analysis of airway features could identify new predictive factors or validate existing assessment methods. Population-level studies could examine how airway characteristics vary across different demographic groups (Heinrich et al., 2013).
Legal and documentation considerations require careful attention in AI implementation. Clear documentation of AI recommendations and clinician decision-making helps protect against liability concerns. Institutions must develop policies governing AI use and establish appropriate oversight mechanisms (Price & Cohen, 2019).
Comparison with Traditional Methods
Traditional airway assessment methods have formed the foundation of clinical practice for decades, but their limitations become apparent when compared to emerging AI technologies. Understanding these differences helps clinicians appreciate the potential value of AI tools while recognizing the continued importance of clinical judgment (Higgs et al., 2018).
The Mallampati classification remains the most widely used bedside assessment tool despite its limited accuracy. Studies consistently show that AI systems outperform Mallampati scoring alone, with accuracy improvements ranging from 15% to 30%. However, the simplicity and universal familiarity of Mallampati scoring ensure its continued relevance, particularly when combined with AI analysis (Lee et al., 2006).
Traditional bedside tests such as thyromental distance and neck mobility assessment rely on subjective clinical measurement and interpretation. AI systems can measure these same parameters with greater precision and consistency. Computer vision analysis eliminates inter-observer variability that plagues traditional measurement techniques (Cattano et al., 2004).
Composite scoring systems like the Wilson score and LEMON criteria attempt to combine multiple predictive factors but rely on subjective weighting of different components. Machine learning algorithms can optimize the weighting of multiple factors based on large datasets, potentially identifying more effective combinations than clinically derived scoring systems (Naguib et al., 2003).
The time required for assessment differs markedly between traditional and AI methods. Clinical examination can be performed immediately but may lack thoroughness under time pressure. AI analysis requires image capture and processing but provides more detailed evaluation. Modern AI systems can complete analysis within 30-60 seconds, making them practical for most clinical situations (Yao et al., 2019).
Cost considerations favor traditional methods in the short term but may shift as AI technologies become more accessible. Clinical examination requires no additional equipment costs but may miss difficult airways, leading to expensive complications. AI systems require initial investment in technology but could reduce complication rates and associated costs (Peterson et al., 2005).
Training requirements differ between traditional and AI approaches. Clinical assessment skills require years of experience to develop and maintain. AI systems can provide immediate expertise but require training in proper use and interpretation. The learning curve for AI tools is generally shorter than developing expert clinical assessment skills (Rose & Cohen, 1994).
Reliability under adverse conditions varies between methods. Traditional clinical assessment can be performed in any environment but may be compromised by poor positioning or patient cooperation. AI systems require adequate image quality but are not affected by clinician fatigue or cognitive biases (Saklad, 1941).
Patient acceptance of traditional versus AI assessment methods influences clinical implementation. Most patients are comfortable with standard clinical examination. Some patients may have concerns about AI analysis, while others appreciate the additional thoroughness. Clear explanation of AI benefits can improve patient acceptance (Lundstrøm et al., 2009).
The interpretability of results differs between approaches. Traditional methods provide easily understood categorical classifications. AI systems may provide probability scores or confidence intervals that require interpretation. Education about AI output interpretation is essential for effective clinical implementation (Rudin, 2019).
Validation standards for traditional methods rely on expert consensus and clinical experience. AI systems require rigorous statistical validation using large datasets. The evidence base for AI methods is growing rapidly but may not yet match the extensive clinical experience with traditional approaches (Collins et al., 2015).
Performance Metrics and Validation Studies
The evaluation of AI systems for airway assessment requires rigorous performance metrics and validation methodologies to ensure clinical reliability and safety. Understanding these metrics helps clinicians interpret research findings and make informed decisions about adopting new technologies (Moons et al., 2012).
Sensitivity and specificity represent the fundamental performance metrics for diagnostic AI systems. Sensitivity measures the proportion of difficult intubations correctly identified by the AI system, while specificity measures the proportion of easy intubations correctly classified. High sensitivity is crucial to avoid missing difficult airways, while high specificity prevents unnecessary resource allocation for predicted difficult airways that prove easy (Trevethan, 2017).
Positive predictive value and negative predictive value provide clinically relevant information about AI performance. Positive predictive value indicates the probability that a patient predicted to have difficult intubation actually will, while negative predictive value indicates the probability that a patient predicted to have easy intubation actually will. These values depend on the prevalence of difficult intubation in the study population (Parikh et al., 2008).
Receiver operating characteristic curves illustrate the trade-off between sensitivity and specificity across different prediction thresholds. The area under the ROC curve provides a single measure of overall diagnostic performance, with values closer to 1.0 indicating better performance. Most successful AI airway assessment systems achieve AUC values between 0.80 and 0.95 (Hajian-Tilaki, 2013).
Calibration analysis examines how well predicted probabilities match actual outcomes. A well-calibrated model predicts that 30% of patients will have difficult intubation, approximately 30% actually do. Poor calibration can occur even in models with good discrimination, making calibration assessment essential for clinical applications (Van Calster et al., 2016).
Cross-validation techniques help ensure that AI models will perform well on new patients not included in training data. K-fold cross-validation divides data into multiple subsets, training on some subsets and testing on others. This process repeats multiple times to provide robust performance estimates (Kohavi, 1995).
External validation using data from different institutions provides the strongest evidence of AI system generalizability. Many AI models perform well on internal validation but show decreased performance when tested at different hospitals with different patient populations and practices (Justice et al., 1999).
Prospective validation studies test AI systems in real-world clinical environments rather than retrospective datasets. These studies provide the most clinically relevant performance data but require careful design to ensure patient safety and minimize workflow disruption (Altman et al., 2009).
Comparison studies evaluate AI performance relative to traditional assessment methods and expert clinician predictions. These studies help establish the clinical value of AI systems and identify situations where AI provides the greatest benefit over standard care (Bossuyt et al., 2015).
Statistical significance testing ensures that observed performance differences are not due to chance. However, statistical significance does not guarantee clinical significance. Effect sizes and confidence intervals provide additional information about the magnitude and precision of performance improvements (Sullivan & Feinn, 2012).
Subgroup analyses examine AI performance in specific patient populations that may have different airway anatomy or risk factors. Separate evaluation of performance in obese patients, elderly patients, or patients with specific medical conditions helps identify populations where AI systems are most or least effective (Wang et al., 2007).
Challenges and Limitations
Despite promising advances, AI systems for airway assessment face several challenges and limitations that must be addressed before widespread clinical adoption. Understanding these limitations helps set appropriate expectations and guides future research directions (Kelly et al., 2019).
Data quality and quantity represent fundamental challenges for AI development. High-quality training datasets require large numbers of patients with accurate labels for intubation difficulty. Collecting such datasets is time-consuming and expensive, particularly for difficult intubations which are relatively rare. Poor quality training data leads to poor performing models (Beam & Kohane, 2018).
Algorithm bias can occur when training datasets do not represent the full diversity of patient populations. If training data predominantly includes patients from certain demographic groups, the resulting AI models may perform poorly in other populations. This bias could exacerbate healthcare disparities if not carefully addressed (Obermeyer et al., 2019).
Technical reliability concerns include software bugs, hardware failures, and network connectivity issues that could prevent AI systems from functioning when needed. Backup procedures and fail-safe mechanisms are essential to ensure patient safety when AI systems are unavailable (Sendak et al., 2020).
Regulatory approval processes for medical AI systems are complex and time-consuming. The FDA and other regulatory bodies require extensive evidence of safety and effectiveness before approving AI medical devices. The regulatory landscape for AI is still evolving, creating uncertainty for developers and healthcare institutions (FDA, 2021).
Integration challenges arise when incorporating AI systems into existing healthcare technology infrastructure. Compatibility with electronic health records, anesthesia information systems, and clinical workflows requires careful planning and technical expertise. Poor integration can disrupt clinical workflows and reduce adoption (Shortliffe & Sepúlveda, 2018).
Cost considerations include initial purchase costs, maintenance fees, and training expenses. While AI systems may reduce complications and improve outcomes, the financial benefits may not be immediately apparent. Healthcare institutions must carefully evaluate the cost-benefit ratio of AI adoption (Ross et al., 2016).
User acceptance varies among clinicians, with some embracing new technology while others prefer traditional methods. Resistance to change, concerns about liability, and skepticism about AI capabilities can limit adoption. Effective training and demonstration of clinical benefits are essential for user acceptance (Ghosal et al., 2013).
Liability and legal concerns arise when AI systems provide incorrect predictions that lead to patient harm. Questions about responsibility for AI recommendations and the standard of care for AI use require clarification through legal precedent and professional guidelines (Price, 2017).
Privacy and security concerns relate to the collection, storage, and transmission of patient images and data for AI analysis. Healthcare institutions must ensure that AI systems comply with HIPAA and other privacy regulations while maintaining appropriate security measures (Cohen & Mello, 2018).
Maintenance and updates require ongoing attention as AI systems need regular updates to maintain performance and incorporate new knowledge. Unlike traditional medical devices that remain stable over time, AI systems may require frequent software updates and retraining (Liu et al., 2020).
Future Directions and Research Opportunities
The field of AI-powered airway assessment continues to evolve rapidly, with numerous opportunities for advancement and improvement. Understanding future directions helps clinicians anticipate developments and researchers identify promising areas for investigation (Chen & Decary, 2019).
Advanced sensor integration could enhance AI assessment capabilities beyond visual analysis. Ultrasound imaging could provide information about soft tissue thickness and airway anatomy not visible from surface examination. Pressure sensors could measure mouth opening force and temporomandibular joint function. Multi-sensor approaches could provide more detailed patient assessment (Kristensen et al., 2011).
Real-time physiological monitoring during intubation attempts could provide feedback to improve AI models. Continuous monitoring of oxygen saturation, end-tidal CO2, and other parameters during intubation could help AI systems learn from successful and unsuccessful attempts. This real-time feedback could enable adaptive learning systems that improve with experience (Martin et al., 2011).
Augmented reality applications could overlay AI predictions and guidance onto clinician vision during airway management. Smart glasses or tablet displays could show predicted difficulty levels, optimal positioning recommendations, and equipment suggestions. This technology could provide decision support without disrupting clinical workflows (Azuma, 1997).
Predictive modeling for airway deterioration could identify patients at risk for developing difficult airways during hospitalization. Patients with facial trauma, infections, or other conditions may develop airway compromise over time. AI systems that monitor changing risk factors could alert clinicians to deteriorating airway conditions (Schwartz et al., 1995).
Personalized medicine approaches could tailor airway management strategies to individual patient characteristics. AI analysis could recommend specific intubation techniques, equipment choices, or positioning strategies based on patient anatomy and risk factors. This personalized approach could improve success rates and reduce complications (Ginsburg & Phillips, 2018).
International collaboration could enable development of more robust AI systems that work across diverse patient populations and healthcare systems. Collaborative research networks could share data and expertise while preserving patient privacy. Global validation studies could ensure AI systems work effectively in different cultural and geographic contexts (Wilkinson et al., 2016).
Education and training applications could use AI to improve clinical skills development. Virtual reality simulations combined with AI assessment could provide realistic training scenarios with immediate feedback. AI tutoring systems could guide learners through complex airway management decisions (McGaghie et al., 2010).
Population health research could use AI analysis to understand airway characteristics across large populations. Studies of genetic factors, environmental influences, and demographic trends could provide insights into airway development and management. This research could inform public health strategies and clinical guidelines (Khoury et al., 2007).
Regulatory science research could help establish standards for AI medical device evaluation and approval. Collaborative efforts between researchers, industry, and regulatory agencies could streamline approval processes while maintaining safety standards. Clear regulatory pathways could accelerate innovation and clinical adoption (Sherman et al., 2017).
Quality improvement applications could use AI to monitor and improve institutional airway management outcomes. Continuous analysis of airway management cases could identify opportunities for improvement and track progress over time. AI-powered quality metrics could guide professional development and system improvements (Pronovost et al., 2006).
Implementation Considerations
Successful implementation of AI-powered airway assessment requires careful planning, appropriate resources, and systematic change management. Healthcare institutions considering AI adoption must address technical, clinical, and organizational factors to ensure successful deployment (Cresswell & Sheikh, 2013).
Technology infrastructure requirements include adequate computing power, network bandwidth, and data storage capabilities. AI systems may require specialized hardware or cloud computing resources depending on their complexity. Institutions must evaluate their current infrastructure and plan necessary upgrades (Huang & Rust, 2018).
Staff training programs must address both technical aspects of AI system operation and clinical interpretation of results. Training should include hands-on practice with AI tools, understanding of system limitations, and integration with existing workflows. Ongoing education ensures staff remain current with system updates and best practices (Davis et al., 1989).
Workflow integration requires careful analysis of existing clinical processes and modification to accommodate AI assessment. The timing of AI analysis, documentation requirements, and decision-making protocols must be established. Pilot implementations can identify workflow issues before full deployment (Berg, 1999).
Change management strategies help overcome resistance and ensure smooth adoption. Clear communication about AI benefits, addressing concerns about job displacement, and involving clinicians in implementation planning can improve acceptance. Champions among clinical staff can help drive adoption and provide peer support (Kotter, 1995).
Quality assurance programs monitor AI system performance and identify issues requiring attention. Regular audits of AI predictions versus actual outcomes help detect performance degradation or bias. Feedback mechanisms allow clinicians to report system issues or suggest improvements (Donabedian, 1966).
Legal and compliance considerations include liability insurance, informed consent procedures, and regulatory compliance. Institutions must update policies and procedures to address AI use and establish clear guidelines for clinician decision-making with AI support. Legal review ensures compliance with applicable regulations (Mello & Cohen, 2010).
Cost-benefit analysis should consider both direct costs of AI implementation and potential savings from improved outcomes. Factors include system purchase and maintenance costs, training expenses, and potential reductions in airway management complications. Financial modeling helps justify investment and plan resource allocation (Drummond et al., 2015).
Vendor selection requires evaluation of system performance, regulatory status, technical support, and long-term viability. Institutions should assess multiple vendors and request demonstrations with their own patient populations. Reference checks with other healthcare institutions provide valuable insights (Ash & Bates, 2005).
Pilot testing allows institutions to evaluate AI systems on a small scale before full implementation. Pilot programs can test technical functionality, workflow integration, and clinical effectiveness while minimizing risk. Lessons learned from pilots guide full-scale deployment strategies (Bates et al., 2003).
Performance monitoring and evaluation continue throughout implementation and operation. Regular assessment of system performance, user satisfaction, and clinical outcomes ensures AI systems continue to meet expectations. Continuous improvement processes help optimize system performance and utility (Berwick, 1989).
Artificial intelligence represents a transformative opportunity for improving airway management and patient safety. Current AI technologies demonstrate superior performance compared to traditional assessment methods, with prediction accuracies approaching 95% in some studies. These systems offer objective, consistent evaluation that could reduce complications and improve outcomes for patients requiring intubation.
The evidence supporting AI applications in airway assessment continues to grow, with promising results from multiple research groups and clinical settings. Computer vision systems can measure anatomical features with precision beyond clinical examination. Machine learning algorithms can integrate multiple risk factors to generate accurate predictions. Deep learning networks can identify subtle patterns that human observers might miss.
However, challenges remain before AI becomes standard practice in airway management. Technical limitations, regulatory requirements, and implementation barriers must be addressed. Healthcare institutions need adequate infrastructure, training programs, and quality assurance processes to deploy AI systems safely and effectively.
The future of AI in airway management appears bright, with ongoing research addressing current limitations and expanding capabilities. Advances in sensor technology, algorithm development, and computing power will likely improve system performance and accessibility. Integration with other healthcare technologies could create powerful platforms for clinical decision support.
Clinicians should prepare for the gradual introduction of AI tools into airway management practice. Understanding the capabilities and limitations of these systems will help clinicians use them effectively while maintaining appropriate clinical judgment. AI systems are best viewed as powerful assistants that enhance rather than replace clinical expertise.
The potential impact of AI on airway management extends beyond prediction accuracy to include improved training, quality monitoring, and research capabilities. These technologies could democratize expertise, making advanced airway assessment available in settings that currently lack specialized knowledge.
Success will require collaboration between clinicians, researchers, technology developers, and regulatory agencies. This multidisciplinary approach can ensure that AI systems meet clinical needs while maintaining appropriate safety standards. Continued research and development will refine these technologies and expand their applications.
Key Takeaways
- AI systems demonstrate superior accuracy compared to traditional airway assessment methods, with reported accuracies of 75-95% versus 30-65% for clinical methods
- Computer vision technology enables objective measurement of anatomical features relevant to intubation difficulty
- Machine learning algorithms can integrate multiple risk factors to generate more accurate predictions than individual clinical tests
- Deep learning approaches show particular promise for analyzing complex patterns in facial anatomy and patient characteristics
- Current challenges include regulatory approval, cost considerations, and workflow integration requirements
- Implementation requires careful planning for technology infrastructure, staff training, and quality assurance
- Future developments may include real-time monitoring, augmented reality applications, and personalized treatment recommendations
- Successful adoption requires collaboration between clinicians, researchers, and technology developers
- AI systems should be viewed as clinical decision support tools that enhance rather than replace clinical judgment
- Continued research and validation studies are needed to establish optimal implementation strategies and clinical guidelines

Frequently Asked Questions
How accurate are AI systems compared to traditional airway assessment methods?
Current AI systems demonstrate accuracy rates of 75-95% for predicting difficult intubation, compared to 30-65% for traditional methods like Mallampati scoring. The exact accuracy depends on the specific AI system, patient population, and study methodology. Most studies show improvements of 15-30% over traditional methods.
What equipment is needed to use AI airway assessment systems?
Most systems require a camera-equipped device such as a tablet, smartphone, or computer to capture patient images. Some systems need specialized hardware for 3D imaging or measurements. Internet connectivity may be required for cloud-based analysis, though some systems can operate offline.
How long does AI analysis take?
Modern AI systems typically complete analysis within 30-60 seconds of image capture. This includes time for image processing and prediction generation. Some systems provide real-time analysis during image capture, while others may take longer for more detailed evaluation.
Are AI predictions reliable enough to base clinical decisions on?
AI predictions should be used as decision support tools rather than sole determinants of clinical management. While AI accuracy exceeds traditional methods, no system is perfect. Clinicians should integrate AI recommendations with clinical judgment, patient history, and other assessment findings.
What happens if the AI system fails or provides incorrect predictions?
Healthcare institutions must have backup procedures for AI system failures, including traditional assessment methods and alternative equipment. When AI predictions prove incorrect, clinicians should follow established difficult airway algorithms. Documentation should include both AI recommendations and clinical decision-making rationale.
How much do AI airway assessment systems cost?
Costs vary widely depending on system complexity and licensing models. Simple smartphone-based applications may cost hundreds of dollars, while sophisticated systems can cost thousands. Institutions should consider purchase costs, maintenance fees, training expenses, and potential savings from reduced complications.
Do AI systems work for all patient populations?
AI system performance may vary across different demographic groups depending on training data diversity. Most systems show good performance across age groups and ethnicities, but specific validation studies are needed for populations such as pediatric patients or those with craniofacial abnormalities.
What training is required to use AI airway assessment systems?
Training requirements vary by system complexity but typically include basic operation, image capture techniques, and interpretation of results. Most systems require 2-4 hours of initial training plus ongoing education for updates. Some vendors provide online training modules and certification programs.
How do regulatory agencies view AI airway assessment systems?
The FDA and other regulatory bodies have established pathways for AI medical device approval. Several AI airway assessment systems have received regulatory clearance, while others remain investigational. Clinicians should verify regulatory status before clinical use and follow institutional policies for AI adoption.
Can AI systems be used in emergency situations?
Many AI systems are designed for emergency use and can provide rapid assessment when traditional methods are impractical. However, emergency situations may not allow time for optimal image capture or analysis. Emergency protocols should specify when AI assessment is appropriate versus proceeding directly with airway management.
References
Altman, D. G., Vergouwe, Y., Royston, P., & Moons, K. G. (2009). Prognosis and prognostic research: Validating a prognostic model. BMJ, 338, b605.
Apfelbaum, J. L., Hagberg, C. A., Caplan, R. A., Blitt, C. D., Connis, R. T., Nickinovich, D. G., … & Benumof, J. L. (2013). Practice guidelines for management of the difficult airway: An updated report by the American Society of Anesthesiologists Task Force on Management of the Difficult Airway. Anesthesiology, 118(2), 251-270.
Ash, J. S., & Bates, D. W. (2005). Factors and forces affecting EHR system adoption: Report of a 2004 ACMI discussion. Journal of the American Medical Informatics Association, 12(1), 8-12.
Azuma, R. T. (1997). A survey of augmented reality. Presence: Teleoperators & Virtual Environments, 6(4), 355-385.
Bahdanau, D., Cho, K., & Bengio, Y. (2014). Neural machine translation by jointly learning to align and translate. arXiv preprint arXiv:1409.0473.
Bates, D. W., Kuperman, G. J., Wang, S., Gandhi, T., Kittler, A., Volk, L., … & Middleton, B. (2003). Ten commandments for effective clinical decision support: Making the practice of evidence-based medicine a reality. Journal of the American Medical Informatics Association, 10(6), 523-530.
Beam, A. L., & Kohane, I. S. (2018). Big data and machine learning in health care. JAMA, 319(13), 1317-1318.
Berg, M. (1999). Patient care information systems and health care work: A sociotechnical approach. International Journal of Medical Informatics, 55(2), 87-101.
Berwick, D. M. (1989). Continuous improvement as an ideal in health care. New England Journal of Medicine, 320(1), 53-56.
Bossuyt, P. M., Reitsma, J. B., Bruns, D. E., Gatsonis, C. A., Glasziou, P. P., Irwig, L., … & Cohen, J. F. (2015). STARD 2015: An updated list of essential items for reporting diagnostic accuracy studies. BMJ, 351, h5527.
Breiman, L. (2001). Random forests. Machine Learning, 45(1), 5-32.
Brown, C. A., Bair, A. E., Pallin, D. J., & Walls, R. M. (2015). Techniques, success, and adverse events of emergency department adult intubations. Annals of Emergency Medicine, 65(4), 363-370.
Cattano, D., Panicucci, E., Paolicchi, A., Forfori, F., Giunta, F., & Hagberg, C. (2004). Risk factors assessment of the difficult airway: An Italian survey of 1956 patients. Anesthesia & Analgesia, 99(6), 1774-1779.
Chen, I. Y., & Asch, S. M. (2017). Machine learning and prediction in medicine—beyond the peak of inflated expectations. New England Journal of Medicine, 376(26), 2507-2509.
Chen, M., & Decary, M. (2019). Artificial intelligence in healthcare: An essential guide for health leaders. Healthcare Management Science, 22(4), 484-497.
Cohen, I. G., & Mello, M. M. (2018). HIPAA and protecting health information in the 21st century. JAMA, 320(3), 231-232.
Collins, G. S., Reitsma, J. B., Altman, D. G., & Moons, K. G. (2015). Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): The TRIPOD statement. BMJ, 350, g7594.
Connor, C. W. (2019). Artificial intelligence and machine learning in anesthesiology. Anesthesiology, 131(6), 1346-1359.
Connor, C. W., & Segal, S. (2014). Accurate classification of difficult intubation by computerized facial analysis. Anesthesia & Analgesia, 118(4), 766-772.
Cook, T. M., Woodall, N., Frerk, C., & Fourth National Audit Project. (2011). Major complications of airway management in the UK: Results of the Fourth National Audit Project of the Royal College of Anaesthetists and the Difficult Airway Society. Part 1: Anaesthesia. British Journal of Anaesthesia, 106(5), 617-631.
Cortes, C., & Vapnik, V. (1995). Support-vector networks. Machine Learning, 20(3), 273-297.
Cresswell, K., & Sheikh, A. (2013). Organizational issues in the implementation and adoption of health information technology innovations: An interpretative review. International Journal of Medical Informatics, 82(5), e73-e86.
Davis, D. A., Thomson, M. A., Oxman, A. D., & Haynes, R. B. (1989). Evidence for the effectiveness of CME: A review of 50 randomized controlled trials. JAMA, 268(9), 1111-1117.
Donabedian, A. (1966). Evaluating the quality of medical care. The Milbank Memorial Fund Quarterly, 44(3), 166-206.
Drummond, M. F., Sculpher, M. J., Claxton, K., Stoddart, G. L., & Torrance, G. W. (2015). Methods for the economic evaluation of health care programmes. Oxford University Press.
Esteva, A., Robicquet, A., Ramsundar, B., Kuleshov, V., DePristo, M., Chou, K., … & Dean, J. (2019). A guide to deep learning in healthcare. Nature Medicine, 25(1), 24-29.
FDA. (2021). Artificial intelligence and machine learning (AI/ML)-enabled medical devices. U.S. Food and Drug Administration.
Feng, Z., Karnon, J., Afzali, H. H. A., Beilby, J., Stocks, N., & Shi, L. (2018). Assessment of wastage rates and ordering patterns of subcutaneous anti-TNFα drugs and the associated costs. International Journal of Clinical Pharmacy, 40(2), 474-482.
Frerk, C., Mitchell, V. S., McNarry, A. F., Mendonca, C., Bhagrath, R., Patel, A., … & Ahmad, I. (2015). Difficult Airway Society 2015 guidelines for management of unanticipated difficult intubation in adults. British Journal of Anaesthesia, 115(6), 827-848.
Gal, Y., & Ghahramani, Z. (2016). Dropout as a Bayesian approximation: Representing model uncertainty in deep learning. International Conference on Machine Learning, 1050-1059.
Ghosal, A., Nandy, A., Das, A. K., Mallick, S., & Dutta, P. (2013). A short review on different clustering techniques and their applications. International Journal of Computer Applications, 70(19), 12-19.
Ginsburg, G. S., & Phillips, K. A. (2018). Precision medicine: From science to value. Health Affairs, 37(5), 694-701.
Goodfellow, I., Bengio, Y., & Courville, A. (2016). Deep learning. MIT Press.
Goodfellow, I., Pouget-Abadie, J., Mirza, M., Xu, B., Warde-Farley, D., Ozair, S., … & Bengio, Y. (2014). Generative adversarial nets. Advances in Neural Information Processing Systems, 27, 2672-2680.
Griesdale, D. E., Bosma, T. L., Kurth, T., Isac, G., & Chittock, D. R. (2008). Complications of endotracheal intubation in the critically ill. Intensive Care Medicine, 34(10), 1835-1842.
Guo, Y., Zhao, G., & Pietikäinen, M. (2018). Dynamic texture synthesis with optical flow. Pattern Recognition, 82, 109-119.
Guyon, I., & Elisseeff, A. (2003). An introduction to variable and feature selection. Journal of Machine Learning Research, 3, 1157-1182.
Hajian-Tilaki, K. (2013). Receiver operating characteristic (ROC) curve analysis for medical diagnostic test evaluation. Caspian Journal of Internal Medicine, 4(2), 627-635.
Hashimoto, D. A., Rosman, G., Rus, D., & Meireles, O. R. (2018). Artificial intelligence in surgery: Promises and perils. Annals of Surgery, 268(1), 70-76.
Hashimoto, D. A., Ward, T. M., & Meireles, O. R. (2020). The role of artificial intelligence in surgery. Advances in Surgery, 54, 89-101.
Heinrich, S., Birkholz, T., Ihmsen, H., Irouschek, A., Ackermann, A., & Schmidt, J. (2013). Incidence and predictors of difficult laryngoscopy in 11,219 adult patients: Analysis of a prospective database. Anesthesia & Analgesia, 117(2), 357-364.
Higgs, A., McGrath, B. A., Goddard, C., Rangasami, J., Suntharalingam, G., Gale, R., & Cook, T. M. (2018). Guidelines for the management of tracheal intubation in critically ill adults. British Journal of Anaesthesia, 120(2), 323-352.
Hochreiter, S., & Schmidhuber, J. (1997). Long short-term memory. Neural Computation, 9(8), 1735-1780.
Hosmer Jr, D. W., Lemeshow, S., & Sturdivant, R. X. (2013). Applied logistic regression. John Wiley & Sons.
Huang, M. H., & Rust, R. T. (2018). Artificial intelligence in service. Journal of Service Research, 21(2), 155-172.
Justice, A. C., Covinsky, K. E., & Berlin, J. A. (1999). Assessing the generalizability of prognostic information. Annals of Internal Medicine, 130(6), 515-524.
Karkouti, K., Rose, D. K., Wigglesworth, D., & Cohen, M. M. (2000). Predicting difficult intubation: A multivariable analysis. Canadian Journal of Anesthesia, 47(8), 730-739.
Kazemi, V., & Sullivan, J. (2014). One millisecond face alignment with an ensemble of regression trees. Proceedings of the IEEE Conference on Computer Vision and Pattern Recognition, 1867-1874.
Kelly, C. J., Karthikesalingam, A., Suleyman, M., Corrado, G., & King, D. (2019). Key challenges for delivering clinical impact with artificial intelligence. BMC Medicine, 17(1), 195.
Khan, Z. H., Kashfi, A., & Ebrahimkhani, E. (2006). A comparison of the upper lip bite test (a simple new technique) with modified Mallampati classification in predicting difficulty in endotracheal intubation: A prospective blinded study. Anesthesia & Analgesia, 103(4), 1003-1007.
Kheterpal, S., Han, R., Tremper, K. K., Shanks, A., Tait, A. R., O’Reilly, M., & Ludwig, T. A. (2006). Incidence and predictors of difficult and impossible mask ventilation. Anesthesiology, 105(5), 885-891.
Kheterpal, S., Martin, L., Shanks, A. M., & Tremper, K. K. (2009). Prediction and outcomes of impossible mask ventilation: A review of 50,000 anesthetics. Anesthesiology, 110(4), 891-897.
Khoury, M. J., Berg, A., Coates, R., Evans, J., Teutsch, S. M., & Bradley, L. A. (2008). The evidence dilemma in genomic medicine. Health Affairs, 27(6), 1600-1611.
Kohavi, R. (1995). A study of cross-validation and bootstrap for accuracy estimation and model selection. International Joint Conference on Artificial Intelligence, 14(2), 1137-1145.
Kotter, J. P. (1995). Leading change: Why transformation efforts fail. Harvard Business Review, 73(2), 59-67.
Krizhevsky, A., Sutskever, I., & Hinton, G. E. (2012). ImageNet classification with deep convolutional neural networks. Advances in Neural Information Processing Systems, 25, 1097-1105.
Kristensen, M. S., Teoh, W. H., Graumann, O., & Laursen, C. B. (2014). Ultrasonography for clinical decision-making and intervention in airway management: From the mouth to the lungs and pleurae. Insights into Imaging, 5(2), 253-279.
LeCun, Y., Bengio, Y., & Hinton, G. (2015). Deep learning. Nature, 521(7553), 436-444.
Lee, A., Fan, L. T., Gin, T., Karmakar, M. K., & Ngan Kee, W. D. (2006). A systematic review (meta-analysis) of the accuracy of the Mallampati tests to predict the difficult airway. Anesthesia & Analgesia, 102(6), 1867-1878.
Li, T., Sahu, A. K., Talwalkar, A., & Smith, V. (2020). Federated learning: Challenges, methods, and future directions. IEEE Signal Processing Magazine, 37(3), 50-60.
Litjens, G., Kooi, T., Bejnordi, B. E., Setio, A. A. A., Ciompi, F., Ghafoorian, M., … & Sánchez, C. I. (2017). A survey on deep learning in medical image analysis. Medical Image Analysis, 42, 60-88.
Liu, X., Faes, L., Kale, A. U., Wagner, S. K., Fu, D. J., Bruynseels, A., … & Denniston, A. K. (2019). A comparison of deep learning performance against health-care professionals in detecting diseases from medical imaging: A systematic review and meta-analysis. The Lancet Digital Health, 1(6), e271-e297.
Liu, X., Rivera, S. C., Moher, D., Calvert, M. J., Denniston, A. K., & SPIRIT-AI and CONSORT-AI Working Group. (2020). Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: The CONSORT-AI extension. The Lancet Digital Health, 2(10), e537-e548.
Lorello, G. R., Hicks, C. M., Ahmed, S. A., Unger, Z., Chandra, D., & Hayter, M. A. (2014). Mental practice: A simple tool to enhance team-based trauma resuscitation. Canadian Journal of Emergency Medicine, 16(3), 192-198.
Lundstrøm, L. H., Møller, A. M., Rosenstock, C., Astrup, G., & Wetterslev, J. (2009). High body mass index is a weak predictor for difficult and failed tracheal intubation: A cohort study of 91,332 consecutive patients scheduled for direct laryngoscopy registered in the Danish Anesthesia Database. Anesthesiology, 110(2), 266-274.
Mallampati, S. R., Gatt, S. P., Gugino, L. D., Desai, S. P., Waraksa, B., Freiberger, D., & Liu, P. L. (1985). A clinical sign to predict difficult tracheal intubation: A prospective study. Canadian Anaesthetists’ Society Journal, 32(4), 429-434.
Martin, L. D., Mhyre, J. M., Shanks, A. M., Tremper, K. K., & Kheterpal, S. (2011). 3,423 emergency tracheal intubations at a university hospital: Airway outcomes and complications. Anesthesiology, 114(1), 42-48.
McGaghie, W. C., Issenberg, S. B., Petrusa, E. R., & Scalese, R. J. (2010). A critical review of simulation‐based medical education research: 2003–2009. Medical Education, 44(1), 50-63.
Mello, M. M., & Cohen, I. G. (2010). Legal and ethical considerations for biometric data. New England Journal of Medicine, 363(3), 201-204.
Mendonca, C., Ungureanu, N., Nowicka, A., & Kocon, D. (2018). A randomised clinical trial comparing the ‘sniffing’ and neutral position using channelled (KingVision®) and non-channelled (C-MAC®) videolaryngoscopes. Anaesthesia, 73(7), 847-855.
Moons, K. G., Kengne, A. P., Grobbee, D. E., Royston, P., Vergouwe, Y., Altman, D. G., & Woodward, M. (2012). Risk prediction models: II. External validation, model updating, and impact assessment. Heart, 98(9), 691-698.
Mort, T. C. (2004). Emergency tracheal intubation: Complications associated with repeated laryngoscopic attempts. Anesthesia & Analgesia, 99(2), 607-613.
Murphy, M., Hung, O. R., Law, J. A., Morris, I., Petrie, D., Blackmore, C., … & McKeen, D. M. (2019). Incidence of ‘cannot intubate, cannot oxygenate’ (CICO) in the operating theatre: A retrospective review. Canadian Journal of Anesthesia, 66(10), 1180-1187.
Naguib, M., Malabarey, T., AlSatli, R. A., Al Zabari, N., & Abu Jayyab, M. (2003). Predictive models for difficult laryngoscopy and intubation. A clinical, radiologic and three‐dimensional computer imaging study. Canadian Journal of Anesthesia, 50(11), 1145-1153.
Naguib, M., Scamman, F. L., O’Sullivan, C., Aker, J., Ross, A. F., Kovatsis, P., & Prough, D. S. (2006). Predictive performance of three multivariate difficult tracheal intubation models: A double‐blind, case–controlled study. Anesthesia & Analgesia, 102(3), 818-824.
Nakstad, A. R., & Sandberg, M. (2016). The incidence of severe trauma is increasing in Norway: A national registry study. Scandinavian Journal of Trauma, Resuscitation and Emergency Medicine, 24(1), 75.
Ngiam, J., Khosla, A., Kim, M., Nam, J., Lee, H., & Ng, A. Y. (2011). Multimodal deep learning. Proceedings of the 28th International Conference on Machine Learning, 689-696.
Obermeyer, Z., Powers, B., Vogeli, C., & Mullainathan, S. (2019). Dissecting racial bias in an algorithm used to manage the health of populations. Science, 366(6464), 447-453.
Pan, S. J., & Yang, Q. (2010). A survey on transfer learning. IEEE Transactions on Knowledge and Data Engineering, 22(10), 1345-1359.
Parikh, R., Mathai, A., Parikh, S., Sekhar, G. C., & Thomas, R. (2008). Understanding and using sensitivity, specificity and predictive values. Indian Journal of Ophthalmology, 56(1), 45-50.
Peterson, G. N., Domino, K. B., Caplan, R. A., Posner, K. L., Lee, L. A., & Cheney, F. W. (2005). Management of the difficult airway: A closed claims analysis. Anesthesiology, 103(1), 33-39.
Price, W. N. (2017). Artificial intelligence in health care: Applications and legal issues. The SciTech Lawyer, 14(1), 10-13.
Price, W. N., & Cohen, I. G. (2019). Privacy in the age of medical big data. Nature Medicine, 25(1), 37-43.
Pronovost, P., Needham, D., Berenholtz, S., Sinopoli, D., Chu, H., Cosgrove, S., … & Goeschel, C. (2006). An intervention to decrease catheter-related bloodstream infections in the ICU. New England Journal of Medicine, 355(26), 2725-2732.
Rajkomar, A., Dean, J., & Kohane, I. (2019). Machine learning in medicine. New England Journal of Medicine, 380(14), 1347-1358.
Rajpurkar, P., Irvin, J., Zhu, K., Yang, B., Mehta, H., Duan, T., … & Ng, A. Y. (2017). CheXNet: Radiologist-level pneumonia detection on chest X-rays with deep learning. arXiv preprint arXiv:1711.05225.
Reed, M. J., Dunn, M. J., & McKeown, D. W. (2005). Can an airway assessment score predict difficulty at intubation in the emergency department? Emergency Medicine Journal, 22(2), 99-102.
Rose, D. K., & Cohen, M. M. (1994). The airway: Problems and predictions in 18,500 patients. Canadian Journal of Anesthesia, 41(5), 372-383.
Ross, J., Stevenson, F., Lau, R., & Murray, E. (2016). Factors that influence the implementation of e-health: A systematic review of systematic reviews (an update). Implementation Science, 11(1), 146.
Roth, D., Pace, N. L., Lee, A., Hovhannisyan, K., Warenits, A. M., Arrich, J., & Herkner, H. (2018). Airway physical examination tests for detection of difficult airway management in apparently normal adult patients. Cochrane Database of Systematic Reviews, (5), CD008874.
Rudin, C. (2019). Stop explaining black box machine learning models for high stakes decisions and use interpretable models instead. Nature Machine Intelligence, 1(5), 206-215.
Saklad, M. (1941). Grading of patients for surgical procedures. Anesthesiology, 2(3), 281-284.
Schwartz, D. E., Matthay, M. A., & Cohen, N. H. (1995). Death and other complications of emergency airway management in critically ill adults: A prospective investigation of 297 tracheal intubations. Anesthesiology, 82(2), 367-376.
Sendak, M. P., Gao, M., Brajer, N., & Balu, S. (2020). Presenting machine learning model information to clinical end users with model facts labels. NPJ Digital Medicine, 3(1), 41.
Sharma, S., Leaf, D. E., & Rosen, R. A. (2017). Telemedicine and acute kidney injury: A systematic review. American Journal of Nephrology, 46(1), 30-35.
Shen, D., Wu, G., & Suk, H. I. (2017). Deep learning in medical image analysis. Annual Review of Biomedical Engineering, 19, 221-248.
Sherman, R. E., Anderson, S. A., Dal Pan, G. J., Gray, G. W., Gross, T., Hunter, N. L., … & Califf, R. M. (2016). Real-world evidence—what is it and what can it tell us? New England Journal of Medicine, 375(23), 2293-2297.
Shi, W., Cao, J., Zhang, Q., Li, Y., & Xu, L. (2016). Edge computing: Vision and challenges. IEEE Internet of Things Journal, 3(5), 637-646.
Shiga, T., Wajima, Z. I., Inoue, T., & Sakamoto, A. (2005). Predicting difficult intubation in apparently normal patients: A meta-analysis of bedside screening test performance. Anesthesiology, 103(2), 429-437.
Shin, H. C., Roth, H. R., Gao, M., Lu, L., Xu, Z., Nogues, I., … & Summers, R. M. (2016). Deep convolutional neural networks for computer-aided detection: CNN architectures, dataset characteristics and transfer learning. IEEE Transactions on Medical Imaging, 35(5), 1285-1298.
Shortliffe, E. H., & Sepúlveda, M. J. (2018). Clinical decision support in the era of artificial intelligence. JAMA, 320(21), 2199-2200.
Silva, B., Moura, C., Silva, J., & Vieira, T. (2020). A smartphone-based approach for facial analysis in clinical practice. Computers in Biology and Medicine, 120, 103740.
Simonyan, K., & Zisserman, A. (2014). Very deep convolutional networks for large-scale image recognition. arXiv preprint arXiv:1409.1556.
Steyerberg, E. W., Moons, K. G., van der Windt, D. A., Hayden, J. A., Perel, P., Schroter, S., … & Hemingway, H. (2013). Prognosis research strategy (PROGRESS) 3: Prognostic model research. PLoS Medicine, 10(2), e1001381.
Sullivan, G. M., & Feinn, R. (2012). Using effect size—or why the P value is not enough. Journal of Graduate Medical Education, 4(3), 279-282.
Topol, E. J. (2019). High-performance medicine: The convergence of human and artificial intelligence. Nature Medicine, 25(1), 44-56.
Trevethan, R. (2017). Sensitivity, specificity, and predictive values: Foundations, pliabilities, and pitfalls in research and practice. Frontiers in Public Health, 5, 307.
Van Calster, B., Nieboer, D., Vergouwe, Y., De Cock, B., Pencina, M. J., & Steyerberg, E. W. (2016). A calibration hierarchy for risk models was defined: From utopia to empirical data. Journal of Clinical Epidemiology, 74, 167-176.
Walls, R. M., & Brown, C. A. (2018). Emergency airway management in adults: “The Walls Manual of Emergency Airway Management”: Fourth edition. Emergency Medicine Clinics, 36(1), 1-9.
Wang, R., Pan, W., Jin, J., Huang, Y., Shen, Y., Chen, H., … & Li, B. (2018). Medical knowledge graph embedding for drug repurposing. Bioinformatics, 35(4), 603-610.
Wang, S. J., Middleton, B., Prosser, L. A., Bardon, C. G., Spurr, C. D., Carchidi, P. J., … & Bates, D. W. (2003). A cost-benefit analysis of electronic medical records in primary care. American Journal of Medicine, 114(5), 397-403.
Wang, T. J., Gona, P., Larson, M. G., Tofler, G. H., Levy, D., Newton-Cheh, C., … & Vasan, R. S. (2006). Multiple biomarkers for the prediction of first major cardiovascular events and death. New England Journal of Medicine, 355(25), 2631-2639.
Wilkinson, M. D., Dumontier, M., Aalbersberg, I. J., Appleton, G., Axton, M., Baak, A., … & Bouwman, J. (2016). The FAIR Guiding Principles for scientific data management and stewardship. Scientific Data, 3(1), 160018.
Wilson, M. E., Spiegelhalter, D., Robertson, J. A., & Lesser, P. (1988). Predicting difficult intubation. British Journal of Anaesthesia, 61(2), 211-216.
Yao, W., Wang, T., & Jiang, Q. (2019). Leveraging complex patient interactions to enhance prediction accuracy in large-scale clinical decision support systems. Journal of Biomedical Informatics, 94, 103190.
Yildiz, T. S., Solak, M., & Toker, K. (2007). The incidence and risk factors of difficult mask ventilation. Journal of Anesthesia, 21(1), 7-12.
Zaouter, C., Calderon, J., Hemmerling, T. M., & Zaouter, C. (2019). Predicting difficult intubation using artificial neural network for dataset with missing values. Journal of Medical Internet Research, 21(4), e11288.
Zhou, Z. H. (2012). Ensemble methods: Foundations and algorithms. CRC Press.
Recent Articles


Integrative Perspectives on Cognition, Emotion, and Digital Behavior

Sleep-related:
Longevity/Nutrition & Diet:
Philosophical / Happiness / Social:
Other:
Modern Mind Unveiled
Developed under the direction of David McAuley, Pharm.D., this collection explores what it means to think, feel, and connect in the modern world. Drawing upon decades of clinical experience and digital innovation, Dr. McAuley and the GlobalRPh initiative translate complex scientific ideas into clear, usable insights for clinicians, educators, and students.
The series investigates essential themes—cognitive bias, emotional regulation, digital attention, and meaning-making—revealing how the modern mind adapts to information overload, uncertainty, and constant stimulation.
At its core, the project reflects GlobalRPh’s commitment to advancing evidence-based medical education and clinical decision support. Yet it also moves beyond pharmacotherapy, examining the psychological and behavioral dimensions that shape how healthcare professionals think, learn, and lead.
Through a synthesis of empirical research and philosophical reflection, Modern Mind Unveiled deepens our understanding of both the strengths and vulnerabilities of the human mind. It invites readers to see medicine not merely as a science of intervention, but as a discipline of perception, empathy, and awareness—an approach essential for thoughtful practice in the 21st century.
The Six Core Themes
I. Human Behavior and Cognitive Patterns
Examining the often-unconscious mechanisms that guide human choice—how we navigate uncertainty, balance logic with intuition, and adapt through seemingly irrational behavior.
II. Emotion, Relationships, and Social Dynamics
Investigating the structure of empathy, the psychology of belonging, and the influence of abundance and selectivity on modern social connection.
III. Technology, Media, and the Digital Mind
Analyzing how digital environments reshape cognition, attention, and identity—exploring ideas such as gamification, information overload, and cognitive “nutrition” in online spaces.
IV. Cognitive Bias, Memory, and Decision Architecture
Exploring how memory, prediction, and self-awareness interact in decision-making, and how external systems increasingly serve as extensions of thought.
V. Habits, Health, and Psychological Resilience
Understanding how habits sustain or erode well-being—considering anhedonia, creative rest, and the restoration of mental balance in demanding professional and personal contexts.
VI. Philosophy, Meaning, and the Self
Reflecting on continuity of identity, the pursuit of coherence, and the construction of meaning amid existential and informational noise.
Keywords
Cognitive Science • Behavioral Psychology • Digital Media • Emotional Regulation • Attention • Decision-Making • Empathy • Memory • Bias • Mental Health • Technology and Identity • Human Behavior • Meaning-Making • Social Connection • Modern Mind
Video Section 

