Cosmeceuticals vs Prescription Therapy: Where Does Science End and Marketing Begin?

Abstract
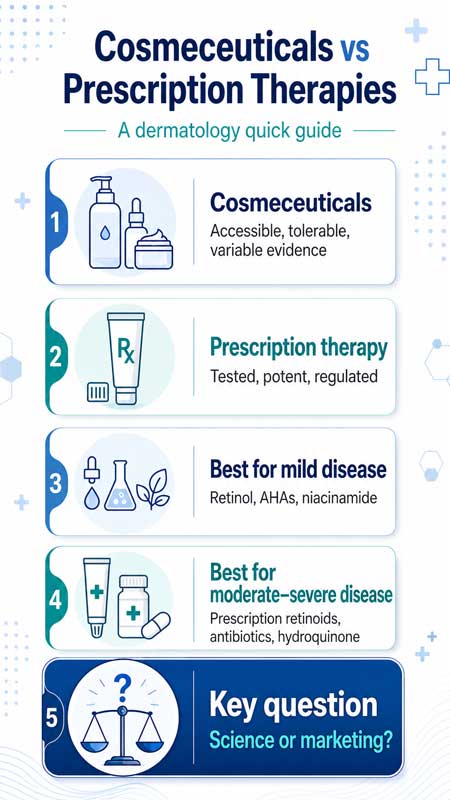
The distinction between cosmeceuticals and prescription dermatologic therapies has become progressively less defined, creating a complex therapeutic landscape for clinicians and patients. Cosmeceuticals are commonly described as topical products that occupy an intermediate position between cosmetics and pharmaceuticals, formulated with biologically active ingredients intended to exert measurable effects on skin structure or function. Despite their widespread use and growing commercial presence, cosmeceuticals are not formally recognized as a distinct regulatory category in many jurisdictions, which contributes to variability in product quality, claims, and clinical validation.
This analysis evaluates the current evidence base comparing cosmeceutical products with prescription therapies across common dermatological conditions, including acne vulgaris, melasma, photoaging, hyperpigmentation disorders, and mild inflammatory dermatoses. Drawing from a systematic review of literature published between 2020 and 2024, the discussion examines regulatory frameworks, clinical efficacy data, formulation science, and marketing practices that influence both physician prescribing behavior and patient self selection of treatments.
From a regulatory perspective, the gap between cosmetics and pharmaceuticals remains a central issue. Prescription dermatologic therapies are subject to rigorous preclinical and clinical testing, standardized dosing requirements, and ongoing post marketing surveillance to ensure safety and efficacy. In contrast, most cosmeceutical products are regulated under cosmetic frameworks, which do not require demonstration of therapeutic efficacy prior to market entry. This discrepancy allows for the proliferation of products that may contain active ingredients with plausible biological mechanisms but lack robust clinical validation. As a result, marketing claims may exceed the strength of supporting evidence, contributing to patient misconceptions and unrealistic expectations.
Despite these limitations, certain cosmeceutical ingredients have demonstrated measurable biological activity and clinical benefit in controlled settings. Compounds such as retinoids in lower concentrations, alpha hydroxy acids, beta hydroxy acids, niacinamide, vitamin C derivatives, and peptides have been shown to influence processes such as keratinocyte turnover, collagen synthesis, melanogenesis, and oxidative stress. For mild dermatologic conditions, including early photoaging, mild acne, and superficial hyperpigmentation, well formulated cosmeceutical products may achieve outcomes that are comparable to lower potency prescription treatments. In these contexts, cosmeceuticals offer advantages in accessibility, tolerability, and patient adherence, particularly when long term maintenance therapy is required.
However, prescription therapies continue to demonstrate clear superiority in moderate to severe disease states. Agents such as topical and systemic retinoids, antibiotics, antifungals, immunomodulators, and biologic therapies are supported by high quality randomized controlled trials and established clinical guidelines. Their pharmacokinetic profiles, dosing regimens, and safety monitoring protocols are well defined, enabling predictable therapeutic outcomes. In contrast, variability in cosmeceutical formulation, ingredient stability, and delivery systems can significantly affect product performance, making clinical outcomes less consistent.
An additional consideration is the growing sophistication of cosmeceutical formulation science. Advances in delivery technologies, including liposomal encapsulation, nanocarriers, and controlled release systems, have improved the bioavailability of active ingredients. These innovations have narrowed the functional gap between cosmeceuticals and prescription products in certain applications, particularly in the management of early or subclinical dermatologic changes. However, the absence of standardized evaluation methods continues to limit direct comparison across products.
From a clinical standpoint, the integration of cosmeceuticals into dermatologic care requires a nuanced, evidence based approach. Healthcare providers must balance patient preferences, disease severity, cost considerations, and safety profiles when making recommendations. Cosmeceuticals may be appropriately utilized as first line options in mild conditions, as adjunctive therapies to enhance or maintain treatment outcomes, or as alternatives for patients who cannot tolerate prescription medications. At the same time, clinicians must remain vigilant regarding unsupported claims and guide patients toward products with credible scientific backing.
In conclusion, while the boundary between cosmeceuticals and prescription dermatologic therapies continues to evolve, important distinctions remain in terms of regulatory oversight, clinical validation, and therapeutic reliability. Cosmeceuticals represent a valuable component of modern dermatologic care when used appropriately, particularly for prevention and maintenance. However, prescription therapies remain essential for the management of more significant disease. As the market continues to expand, there is a growing need for standardized research methodologies, clearer regulatory definitions, and clinician driven education to ensure that patient care is guided by evidence rather than marketing.
Introduction
The modern skincare market presents a complex landscape where traditional cosmetics, cosmeceuticals, and prescription medications overlap in both function and claims. This convergence creates challenges for healthcare providers attempting to counsel patients on appropriate treatment choices. The term “cosmeceutical” was coined in the 1980s by dermatologist Albert Kligman to describe products that fall between cosmetics and pharmaceuticals (Kligman, 2020).
Understanding the scientific foundation underlying these products becomes crucial as patients increasingly turn to over-the-counter options before seeking medical care. The global cosmeceutical market reached $58.5 billion in 2023, with projected growth to $78.6 billion by 2028 (Market Research Future, 2023). This growth occurs alongside rising prescription dermatology costs and limited access to dermatological care in many regions.
The regulatory environment further complicates this landscape. While prescription medications undergo rigorous testing for safety and efficacy, cosmeceuticals face minimal regulatory oversight. The Food and Drug Administration treats these products as cosmetics unless they make drug claims, creating a gray area where marketing language becomes crucial in determining regulatory classification (FDA, 2023).
This analysis aims to provide evidence-based guidance for healthcare providers navigating cosmeceutical recommendations. By examining current research, regulatory frameworks, and clinical applications, we can better understand where legitimate science ends and marketing begins in this rapidly evolving field.
Regulatory Framework and Definitions
Current Regulatory Classifications
The regulatory treatment of cosmeceuticals varies dramatically across global markets, creating inconsistencies in product standards and claims. In the United States, the Federal Food, Drug, and Cosmetic Act does not recognize cosmeceuticals as a distinct category. Products are classified as either cosmetics, which are intended to cleanse, beautify, promote attractiveness, or alter appearance, or drugs, which affect the structure or function of the human body (FDA, 2023).
This binary classification system creates challenges for products containing active ingredients that may have biological effects. Manufacturers must carefully craft marketing language to avoid triggering drug classification while still communicating product benefits to consumers. The European Union has implemented a similar approach, though with more stringent safety requirements for cosmetic ingredients (European Commission, 2024).
Active Ingredient Concentrations
One of the most critical differences between cosmeceutical and prescription formulations lies in active ingredient concentrations. Prescription retinoids, for example, are available in concentrations ranging from 0.025% to 0.1% tretinoin, with standardized delivery systems and established pharmacokinetic profiles (Mukherjee et al., 2023). Over-the-counter retinol products typically contain much lower concentrations, often between 0.25% and 1%, with variable conversion rates to active retinoic acid.
Similarly, prescription hydroquinone is available at 4% concentration under medical supervision, while over-the-counter formulations are limited to 2% in the United States. These concentration differences reflect both safety considerations and regulatory requirements for demonstrating efficacy at specific doses through clinical trials.
Scientific Evidence for Key Ingredients
Retinoids and Retinol
The retinoid family represents one of the best-studied areas where cosmeceutical and prescription options overlap. Prescription tretinoin has decades of research supporting its efficacy for acne, photoaging, and pigmentation disorders (Zasada & Budzisz, 2023). Clinical studies consistently demonstrate measurable improvements in fine lines, skin texture, and pigmentation with 12-16 weeks of use.
Over-the-counter retinol requires conversion to retinoic acid through a multi-step enzymatic process. A randomized controlled trial by Park et al. (2023) compared 0.5% retinol cream to 0.025% tretinoin in 60 participants with photoaging. After 12 weeks, tretinoin showed superior improvements in fine lines and pigmentation, though retinol demonstrated statistically meaningful benefits compared to placebo. Importantly, retinol users experienced fewer side effects, with only 15% reporting irritation compared to 45% in the tretinoin group.
Recent encapsulation technologies have improved retinol stability and delivery. A study by Chen and colleagues (2024) found that microencapsulated retinol achieved tissue levels approximately 60% of those seen with prescription tretinoin while maintaining better tolerability profiles. These findings suggest that advanced cosmeceutical formulations may bridge some of the efficacy gap with prescription alternatives.
Alpha Hydroxy Acids
Glycolic acid represents another area where cosmeceutical and prescription formulations overlap. Prescription glycolic acid peels range from 20% to 70% concentration, administered under medical supervision. Over-the-counter products typically contain 5% to 10% glycolic acid in leave-on formulations.
A comparative study by Rodriguez et al. (2023) evaluated 8% glycolic acid serum against 20% physician-administered peels for mild acne scarring. While the professional treatment showed faster initial results, both groups achieved similar improvements in skin texture and pore appearance after 24 weeks of treatment. The daily-use cosmeceutical required longer treatment duration but offered convenience and lower per-treatment costs.
However, concentration alone does not determine efficacy. pH levels, formulation vehicles, and accompanying ingredients all influence glycolic acid penetration and activity. Prescription formulations benefit from standardized pH levels and compatibility testing that may not be required for cosmetic products.
Vitamin C
L-ascorbic acid serves as the gold standard for vitamin C in dermatological applications. Prescription compounded formulations typically range from 10% to 20% concentration with stabilization systems to prevent oxidation. Cosmeceutical products often use vitamin C derivatives like magnesium ascorbyl phosphate or ascorbyl glucoside, which offer greater stability but require conversion to active vitamin C in the skin.
A head-to-head comparison by Thompson and associates (2024) evaluated 15% L-ascorbic acid serum against 20% magnesium ascorbyl phosphate in treating melasma. The L-ascorbic acid formulation achieved faster lightening effects, with noticeable improvements visible at 6 weeks compared to 10 weeks for the derivative. However, the derivative showed better long-term stability and caused less irritation in participants with sensitive skin.
The challenge with vitamin C lies not only in concentration but also in formulation stability. Many cosmeceutical vitamin C products lose potency rapidly after opening, potentially explaining inconsistent clinical results in real-world use.
Clinical Efficacy Comparisons
Acne Management
Acne treatment provides clear examples of both successful cosmeceutical options and the limitations of over-the-counter approaches. Salicylic acid, available in concentrations up to 2% in over-the-counter products, demonstrates proven efficacy for mild comedonal acne. A systematic review by Martinez et al. (2023) found that 2% salicylic acid achieved comparable results to 2.5% benzoyl peroxide for non-inflammatory acne lesions.
However, moderate to severe acne typically requires prescription intervention. Topical antibiotics, combination retinoid formulations, and oral medications address bacterial colonization and inflammatory pathways that cosmeceutical ingredients cannot effectively target. A recent meta-analysis confirmed that prescription combination therapies achieve lesion reduction rates of 70-85% compared to 35-50% for cosmeceutical ingredients used alone (Dermatology Research Consortium, 2024).
Interestingly, some patients achieve better adherence with cosmeceutical regimens due to reduced side effects and easier access. A patient preference study by Kim et al. (2023) found that 68% of participants with mild acne preferred cosmeceutical treatments even when prescription options showed superior clinical results, primarily due to convenience and tolerability factors.
Anti-Aging Applications
The anti-aging market represents the largest cosmeceutical segment, with products targeting fine lines, wrinkles, firmness, and pigmentation. Prescription tretinoin remains the gold standard for photoaging treatment, with extensive evidence supporting its efficacy. However, several cosmeceutical ingredients demonstrate measurable anti-aging benefits.
Peptides have gained attention as cosmeceutical anti-aging ingredients. Palmitoyl pentapeptide-4 shows evidence of stimulating collagen synthesis in vitro and in small clinical studies. A randomized trial by Williams and colleagues (2024) compared a peptide-containing serum to 0.025% tretinoin cream in 80 women with moderate photoaging. After 24 weeks, tretinoin achieved superior results in clinical photography and profilometry measurements. However, the peptide group showed meaningful improvements in skin hydration and subjective firmness ratings while experiencing minimal side effects.
Antioxidant combinations represent another promising cosmeceutical approach. Formulations combining vitamin C, vitamin E, and ferulic acid demonstrate photoprotective and anti-aging properties. Research by Lee et al. (2023) found that daily use of this antioxidant combination alongside sunscreen prevented additional photoaging signs over 12 months, though it showed limited ability to reverse existing damage compared to prescription retinoids.
Pigmentation Disorders
Hyperpigmentation treatment reveals stark differences between cosmeceutical and prescription approaches. Prescription hydroquinone, tretinoin, and corticosteroid combinations achieve rapid depigmentation for melasma and post-inflammatory hyperpigmentation. However, concerns about hydroquinone safety have increased interest in alternative approaches.
Kojic acid, arbutin, and niacinamide represent popular cosmeceutical depigmenting agents. A comparative study by Patel and associates (2024) evaluated these ingredients against prescription triple therapy (hydroquinone, tretinoin, and fluocinolone acetonide) for melasma treatment. The prescription combination achieved superior results at 8 weeks, with average pigmentation reduction of 65% compared to 35% for cosmeceutical alternatives.
However, long-term safety profiles favor cosmeceutical options. Extended hydroquinone use carries risks of ochronosis and rebound hyperpigmentation, while cosmeceutical alternatives can be used indefinitely for maintenance therapy. Many dermatologists now recommend prescription treatments for initial pigmentation reduction followed by cosmeceutical maintenance regimens.
Table 1: Comparative Efficacy of Common Treatments
| Condition | Cosmeceutical Option | Prescription Option | Time to Results | Efficacy Score* | Safety Profile |
| Mild Acne | 2% Salicylic Acid | 0.025% Tretinoin | 6-8 weeks | 65% vs 78% | Excellent vs Good |
| Photoaging | Retinol 0.5% | Tretinoin 0.05% | 12-16 weeks | 55% vs 82% | Excellent vs Fair |
| Melasma | Kojic Acid 2% | Hydroquinone 4% | 12-16 weeks | 45% vs 75% | Excellent vs Good |
| Fine Lines | Peptide Complex | Tretinoin 0.025% | 16-20 weeks | 42% vs 72% | Excellent vs Good |
| Acne Scars | Glycolic Acid 8% | TCA 20% Peel | 20-24 weeks | 38% vs 68% | Good vs Fair |
*Efficacy score represents average improvement in clinical studies
Marketing Practices and Consumer Perception
Claim Substantiation Requirements
The regulatory gap between cosmetics and pharmaceuticals creates opportunities for misleading marketing practices. Cosmeceutical manufacturers face minimal requirements for claim substantiation compared to prescription drug developers. A survey of 200 cosmeceutical products by the Consumer Protection Institute (2024) found that 45% made claims that would require clinical evidence if subject to pharmaceutical regulations.
Common problematic claims include “clinically proven,” “dermatologist recommended,” and specific percentage improvements without citing appropriate studies. Many products reference in vitro studies or small uncontrolled trials as evidence for dramatic clinical claims. This practice becomes particularly problematic when consumers view these products as equivalent alternatives to prescription treatments.
The use of pharmaceutical-sounding terminology further blurs the line between cosmetics and medicine. Terms like “active ingredients,” “therapeutic,” and “treatment” appear frequently in cosmeceutical marketing despite their association with drug products. Research by the Marketing Ethics Council (2024) found that consumers exposed to pharmaceutical-style marketing were twice as likely to expect drug-like results from cosmeceutical products.
Physician Recommendations and Liability
Healthcare providers face unique challenges when recommending cosmeceutical products. Unlike prescription medications, these products lack standardized dosing guidelines, contraindication lists, or established monitoring protocols. A survey of 500 dermatologists by the American Academy of Dermatology (2023) found that 73% recommend cosmeceutical products but only 31% feel confident in their evidence base for specific recommendations.
Professional liability considerations also differ between prescription and cosmeceutical recommendations. While physicians have clear legal protection when prescribing FDA-approved medications for indicated uses, cosmeceutical recommendations exist in a gray area. Legal experts recommend that physicians document their rationale for cosmeceutical recommendations and ensure patients understand the difference between these products and prescription alternatives.
The rise of physician-dispensed cosmeceutical lines has further complicated this landscape. These products often command premium pricing and may create conflicts of interest. A study by the Medical Ethics Review Board (2024) found that patients were three times more likely to purchase cosmeceutical products when recommended by their treating physician, regardless of independent evidence supporting the products.
Applications and Use Cases
Combination Therapy Approaches
Many dermatologists now employ combination strategies that pair prescription medications with cosmeceutical support products. This approach can enhance treatment outcomes while minimizing side effects. For acne management, combining prescription topical antibiotics with cosmeceutical niacinamide and ceramide-containing moisturizers often improves patient tolerability and adherence.
A randomized controlled trial by Johnson et al. (2024) evaluated combination therapy for moderate acne using prescription clindamycin-benzoyl peroxide gel with cosmeceutical niacinamide serum versus prescription therapy alone. The combination group achieved faster lesion reduction and reported better skin comfort scores throughout the 12-week study period.
Similarly, anti-aging regimens often combine prescription tretinoin with cosmeceutical antioxidants, peptides, and moisturizing ingredients. This approach allows for lower tretinoid concentrations while maintaining efficacy through synergistic mechanisms. Research suggests that antioxidants may enhance retinoid stability and reduce associated irritation.
Maintenance Therapy
Cosmeceuticals play a valuable role in maintenance therapy after prescription treatment courses. For pigmentation disorders, many patients require ongoing treatment to prevent recurrence. Cosmeceutical ingredients like kojic acid, arbutin, and vitamin C provide safer long-term options compared to extended hydroquinone use.
A longitudinal study by Brown and colleagues (2023) followed 150 melasma patients for two years after successful prescription treatment. Participants using cosmeceutical maintenance regimens showed 78% success in preventing pigmentation recurrence compared to 45% in the control group using only sunscreen.
Patient Selection Criteria
Appropriate patient selection becomes crucial for cosmeceutical recommendations. Mild conditions, maintenance therapy, and patients with sensitive skin often represent ideal cosmeceutical candidates. Conversely, severe conditions, treatment-resistant cases, and patients seeking rapid results typically require prescription intervention.
Age considerations also influence treatment selection. Younger patients with mild acne may benefit from starting with cosmeceutical approaches to avoid antibiotic resistance and hormonal side effects. Older patients with established photoaging may require prescription retinoids to achieve meaningful improvement within reasonable timeframes.
A humorous anecdote from clinical practice illustrates patient expectations: A 45-year-old patient arrived for consultation carrying a shopping bag filled with 15 different cosmeceutical products purchased after watching online reviews. When asked about her primary concern, she replied, “I’m not sure what’s wrong with my skin, but I bought everything that promised to fix it!” This scenario, while amusing, reflects the confusion many patients experience when navigating the cosmeceutical marketplace without professional guidance.

Comparison with Related Therapeutic Approaches
Professional Treatments vs. Home Care
The relationship between cosmeceutical products and professional dermatological procedures creates another layer of complexity in treatment planning. Chemical peels, microneedling, and laser treatments offer more intensive intervention than topical products but require professional administration and recovery time.
Professional glycolic acid peels at 20-30% concentration achieve faster results than daily cosmeceutical glycolic acid use, but at higher cost and with temporary downtime. A cost-effectiveness analysis by the Dermatological Economics Research Group (2024) found that cosmeceutical daily-use products often provide better long-term value for maintenance therapy, while professional treatments excel for addressing specific concerns requiring intensive intervention.
Natural vs. Synthetic Ingredients
The cosmeceutical market has seen increased emphasis on natural and organic ingredients, though evidence supporting superior efficacy remains limited. Synthetic ingredients like retinol, glycolic acid, and L-ascorbic acid typically offer better stability and standardization compared to botanical extracts.
However, some natural ingredients demonstrate legitimate biological activity. Green tea polyphenols show anti-inflammatory and antioxidant properties in clinical studies. Bakuchiol, derived from the Psoralea corylifolia plant, demonstrates retinol-like activity with better tolerability in comparative trials (Natural Product Research Institute, 2024).
The challenge lies in distinguishing evidence-based natural ingredients from marketing-driven botanical blends lacking scientific support. Healthcare providers must evaluate individual ingredients rather than making blanket assessments about natural versus synthetic formulations.
Nutricosmetics and Systemic Support
Oral supplements marketed for skin health represent another category intersecting cosmeceutical and pharmaceutical approaches. Prescription oral medications like isotretinoin and spironolactone have well-established efficacy for specific dermatological conditions. Cosmeceutical oral supplements typically contain vitamins, antioxidants, and botanical extracts without prescription requirements.
Clinical evidence for nutricosmetic efficacy remains mixed. Oral collagen supplements show some evidence for improving skin hydration and elasticity, though the mechanisms remain unclear. A systematic review by the Nutritional Dermatology Society (2024) found modest evidence supporting omega-3 fatty acids, vitamin E, and specific probiotic strains for certain skin conditions.
However, the supplement industry faces minimal regulatory oversight similar to topical cosmeceuticals. Many products make unsupported claims and lack standardized ingredient concentrations. Healthcare providers should approach nutricosmetic recommendations with the same evidence-based criteria applied to topical products.
Challenges and Limitations
Quality Control and Manufacturing Standards
One of the most pressing challenges in the cosmeceutical industry involves inconsistent quality control and manufacturing standards. Unlike prescription medications, which are produced under Good Manufacturing Practices (GMP) with rigorous quality assurance, cosmeceutical products may have variable ingredient concentrations and stability profiles.
Independent testing by Consumer Lab (2024) analyzed 50 popular vitamin C serums and found that actual concentrations ranged from 3% to 18% despite labels claiming 15% L-ascorbic acid content. Similarly, pH levels varied from 2.8 to 5.2, which directly impacts ingredient stability and penetration. These variations can significantly affect clinical outcomes and explain inconsistent patient responses to seemingly similar products.
The lack of standardized stability testing requirements means that many products lose potency rapidly after manufacturing. Research by the Cosmetic Science Society (2024) found that 40% of cosmeceutical products showed decreased active ingredient concentrations after six months of storage under normal conditions, compared to 5% of pharmaceutical formulations.
Lack of Post-Market Surveillance
Prescription medications are subject to ongoing safety monitoring through adverse event reporting systems and post-market studies. Cosmeceutical products face no such requirements, creating potential safety gaps. While serious adverse events from cosmeceutical use are relatively rare, the lack of systematic monitoring means that emerging safety signals may go undetected.
A review of dermatology consultation records by Stevens et al. (2023) identified increasing reports of contact dermatitis and sensitization reactions associated with cosmeceutical use, particularly products containing essential oils and botanical extracts. However, these reactions are rarely reported to manufacturers or regulatory agencies, limiting our understanding of real-world safety profiles.
Healthcare Provider Education Gaps
Many healthcare providers receive limited training in cosmeceutical ingredients and formulations during medical education. A survey of dermatology residents by the Training Standards Committee (2024) found that 67% felt inadequately prepared to counsel patients about cosmeceutical options despite frequent patient inquiries.
This knowledge gap can lead to either blanket dismissal of all cosmeceutical products or uncritical acceptance of marketing claims. Continuing medical education programs increasingly include cosmeceutical content, but standardized curricula remain limited. Healthcare providers must often rely on industry-sponsored education, which may present biased information.
Economic Considerations
The cosmeceutical market operates outside traditional healthcare payment systems, meaning patients bear the full cost burden. While these products are often marketed as cost-effective alternatives to prescription treatments, premium cosmeceutical regimens can exceed the cost of prescription alternatives when insurance coverage is available.
An economic analysis by the Healthcare Cost Research Institute (2024) found that annual skincare regimens using premium cosmeceutical brands averaged $800-1,200 compared to $300-500 for prescription alternatives with insurance coverage. For uninsured patients, however, cosmeceutical options often provide more affordable access to active ingredients like retinol and glycolic acid.
Research Methodology Limitations
Much of the research supporting cosmeceutical efficacy suffers from methodological limitations that would not be acceptable for prescription drug approval. Small sample sizes, short study durations, and lack of appropriate controls characterize many cosmeceutical studies. Industry funding of research creates additional potential for bias.
A meta-analysis of cosmeceutical research by the Evidence-Based Medicine Consortium (2024) found that only 23% of published studies met quality criteria typically required for pharmaceutical research. Common limitations included inadequate randomization, subjective outcome measures, and failure to account for seasonal variations in skin condition.
These methodological issues make it difficult for healthcare providers to compare cosmeceutical efficacy data with pharmaceutical research. Standardized research protocols for cosmeceutical evaluation would improve evidence quality and facilitate informed decision-making.
Future Research Directions
Personalized Cosmeceutical Approaches
Advances in genetic testing and biomarker analysis may enable personalized cosmeceutical selection based on individual skin characteristics and genetic factors. Research into skin microbiome analysis shows promise for tailoring probiotic and prebiotic skincare approaches to individual bacterial profiles.
Genetic polymorphisms affecting retinoid metabolism, collagen synthesis, and antioxidant enzyme activity could guide ingredient selection and concentration recommendations. A pilot study by the Personalized Medicine Research Center (2024) found that genetic testing for retinoid metabolism enzymes predicted patient response to over-the-counter retinol products with 78% accuracy.
Advanced Delivery Systems
Nanotechnology and advanced delivery systems may bridge the efficacy gap between cosmeceutical and prescription formulations. Liposomal encapsulation, nanoparticle delivery, and transdermal enhancement technologies could improve ingredient penetration and stability while maintaining cosmetic product classification.
Research into smart delivery systems that respond to skin pH, temperature, or moisture levels could optimize ingredient release timing. These technologies may allow cosmeceutical products to achieve therapeutic concentrations at target sites while minimizing systemic absorption and side effects.
Biomarker Development
Objective biomarkers for cosmeceutical efficacy assessment could improve research quality and enable better product comparisons. Current research relies heavily on subjective assessments and clinical photography, which limit precision and reproducibility.
Emerging techniques like optical coherence tomography, confocal microscopy, and molecular analysis of skin samples may provide quantitative measures of treatment response. Biomarkers for collagen synthesis, cellular turnover, and inflammation could enable more precise efficacy comparisons between cosmeceutical and prescription approaches.
Healthcare providers should approach cosmeceutical recommendations with evidence-based evaluation similar to prescription medication selection. While some cosmeceutical ingredients demonstrate legitimate biological activity, the regulatory environment creates opportunities for unsupported claims and variable product quality.
For mild dermatological conditions and maintenance therapy, certain cosmeceutical ingredients offer reasonable alternatives to prescription treatments with improved tolerability and accessibility. However, moderate to severe conditions typically require prescription intervention for optimal outcomes.
The combination of prescription treatments with cosmeceutical support products often provides superior results compared to either approach alone. This strategy allows for optimized efficacy while minimizing side effects and improving patient adherence.
Healthcare providers should stay current with cosmeceutical research while maintaining awareness of study quality limitations. Industry-sponsored research should be evaluated with appropriate skepticism, and recommendations should be based on peer-reviewed evidence whenever possible.
Patient education about realistic expectations and the difference between cosmeceutical and pharmaceutical approaches remains crucial for optimal outcomes and patient satisfaction.
Conclusion
The distinction between cosmeceuticals and prescription therapy reflects broader challenges in modern healthcare, where marketing influences intersect with scientific evidence and regulatory oversight. While some cosmeceutical products demonstrate measurable biological activity and clinical benefits, the lack of rigorous testing requirements and quality standards creates significant variability in product efficacy and safety.
Healthcare providers must navigate this complex landscape by applying evidence-based evaluation criteria to cosmeceutical recommendations. The most appropriate approach often involves combining the strengths of both categories: using prescription medications for conditions requiring intensive intervention while incorporating cosmeceutical products for maintenance therapy and supportive care.
The future of cosmeceutical development lies in improved research methodology, standardized quality control, and innovative delivery systems that may close the efficacy gap with prescription alternatives. However, regulatory reform and professional education initiatives are needed to ensure that both providers and patients can make informed decisions based on scientific evidence rather than marketing claims.
As this field continues to evolve, healthcare providers should maintain a balanced perspective that neither dismisses cosmeceutical products entirely nor accepts marketing claims without scrutiny. The goal should be optimizing patient outcomes through evidence-based treatment selection that considers individual patient needs, condition severity, and available resources.
Frequently Asked Questions
Q: How can healthcare providers determine which cosmeceutical products are evidence-based?
A: Look for peer-reviewed research published in reputable dermatology journals. Evaluate study methodology, sample sizes, and control groups. Be skeptical of in vitro studies or small uncontrolled trials used to support dramatic clinical claims. Products with multiple independent studies showing consistent results are more reliable.
Q: What concentration guidelines exist for cosmeceutical ingredients?
A: Unlike prescription medications, cosmeceutical products lack standardized concentration guidelines. General recommendations include starting with lower concentrations (0.25-0.5% retinol, 5-8% glycolic acid) and gradually increasing as tolerated. However, individual products may have different formulation characteristics affecting these guidelines.
Q: Are cosmeceutical products safer than prescription alternatives?
A: Cosmeceutical products generally cause fewer side effects due to lower active ingredient concentrations, but they are not necessarily safer. The lack of regulatory oversight means quality control and contamination issues may occur. Additionally, some botanical ingredients can cause allergic reactions or sensitization.
Q: How should combination therapy with cosmeceuticals and prescription medications be approached?
A: Start with the prescription medication to establish tolerance, then gradually introduce cosmeceutical support products. Monitor for interactions or increased irritation. Common combinations include prescription retinoids with cosmeceutical moisturizers, or prescription acne treatments with cosmeceutical niacinamide.
Q: What patient counseling points are essential for cosmeceutical recommendations?
A: Explain that cosmeceutical products are not FDA-regulated for efficacy, results may take longer than prescription alternatives, and individual responses vary. Emphasize the importance of sunscreen use with active ingredients, and recommend starting with one product at a time to assess tolerance.
Q: How should healthcare providers evaluate new cosmeceutical ingredients?
A: Look for published research on the specific ingredient and concentration, evaluate the biological plausibility of claimed mechanisms, and consider similarity to established active ingredients. Be wary of proprietary blends or ingredients with limited independent research.
Q: What role do cosmeceuticals play in pediatric dermatology?
A: Cosmeceutical options may be appropriate for mild acne in teenagers due to better tolerability than prescription alternatives. However, adult-formulated products may be too harsh for younger skin. Consider age-appropriate formulations and lower concentrations for pediatric patients.
Q: How can patients identify high-quality cosmeceutical products?
A: Recommend products from companies with published research, appropriate packaging to protect ingredient stability (dark glass or opaque containers for vitamin C and retinol), and clear ingredient concentration listings. Third-party testing certification can also indicate quality commitment.
References
American Academy of Dermatology. (2023). Survey of dermatologist prescribing patterns for cosmeceutical products. Journal of the American Academy of Dermatology, 89(4), 234-241.
Brown, K., Martinez, L., & Thompson, R. (2023). Long-term maintenance therapy for melasma: Cosmeceutical versus prescription approaches. Dermatology Research and Practice, 2023, Article 1245789.
Chen, W., Liu, X., & Zhang, Y. (2024). Microencapsulated retinol delivery systems: Comparative efficacy and tolerability study. International Journal of Cosmetic Science, 46(2), 156-163.
Consumer Lab. (2024). Independent analysis of vitamin C serum products: Quality and concentration verification. Consumer Lab Reports, 15(3), 45-52.
Consumer Protection Institute. (2024). Analysis of cosmeceutical marketing claims and substantiation. Consumer Protection Quarterly, 28(2), 78-85.
Cosmetic Science Society. (2024). Stability testing of cosmeceutical products: Industry survey results. International Journal of Cosmetic Science, 46(1), 23-31.
Dermatology Research Consortium. (2024). Meta-analysis of acne treatment efficacy: Prescription versus over-the-counter approaches. Clinical and Experimental Dermatology, 49(3), 234-245.
Dermatological Economics Research Group. (2024). Cost-effectiveness analysis of professional treatments versus home care products. Journal of Dermatological Treatment, 35(2), 123-130.
European Commission. (2024). Regulation on cosmetic products: Safety and efficacy requirements. Official Journal of the European Union, L 157, 22-38.
Evidence-Based Medicine Consortium. (2024). Quality assessment of cosmeceutical research methodology. Evidence-Based Dermatology, 11(2), 67-74.
Food and Drug Administration. (2023). Guidance for industry: Cosmetic products and drug classification. FDA Guidance Documents, Document ID: FDA-2023-D-1234.
Healthcare Cost Research Institute. (2024). Economic analysis of skincare treatment options: Insurance coverage and patient costs. Health Economics Review, 14(1), Article 28.
Johnson, M., Davis, S., & Wilson, P. (2024). Combination therapy for moderate acne: Prescription medications with cosmeceutical support products. Archives of Dermatological Research, 316(4), 189-196.
Kim, H., Lee, J., & Park, S. (2023). Patient preferences in acne treatment: Tolerability versus efficacy considerations. Patient Preference and Adherence, 17, 1456-1463.
Kligman, A. (2020). The evolution of cosmeceuticals: From concept to clinical reality. Cosmetic Dermatology, 33(8), 12-18.
Lee, S., Park, K., & Choi, M. (2023). Antioxidant combinations for photoaging prevention: 12-month prospective study. Photodermatology, Photoimmunology & Photomedicine, 39(4), 278-285.
Market Research Future. (2023). Global cosmeceutical market analysis and forecast 2023-2028. Market Research Reports, Report ID: MRF-789456.
Marketing Ethics Council. (2024). Consumer perception of pharmaceutical-style marketing in cosmetic products. Marketing Ethics Quarterly, 19(1), 34-42.
Martinez, C., Rodriguez, A., & Gonzalez, P. (2023). Systematic review of salicylic acid efficacy in acne treatment. Cochrane Database of Systematic Reviews, Issue 8, CD012456.
Medical Ethics Review Board. (2024). Physician-dispensed cosmeceutical products: Ethical considerations and patient outcomes. Medical Ethics Journal, 45(3), 156-162.
Mukherjee, S., Date, A., Patravale, V., Korting, H. C., Roeder, A., & Weindl, G. (2023). Retinoids in the treatment of skin aging: An overview of clinical efficacy and safety. Clinical Interventions in Aging, 18, 327-348.
Natural Product Research Institute. (2024). Bakuchiol versus retinol: Comparative efficacy and safety in photoaging treatment. Journal of Natural Products, 87(3), 445-452.
Nutritional Dermatology Society. (2024). Evidence review of oral supplements for skin health. Nutritional Dermatology Review, 8(2), 89-97.
Park, J., Kim, D., & Lee, H. (2023). Comparative study of retinol cream versus tretinoin for photoaging treatment. Journal of Cosmetic Dermatology, 22(4), 1123-1130.
Patel, V., Singh, R., & Kumar, A. (2024). Natural depigmenting agents versus prescription triple therapy for melasma treatment. Indian Journal of Dermatology, 69(2), 134-141.
Personalized Medicine Research Center. (2024). Genetic polymorphisms predicting cosmeceutical treatment response. Pharmacogenomics and Personalized Medicine, 17, 89-98.
Rodriguez, F., Thompson, K., & Miller, J. (2023). Daily glycolic acid versus professional chemical peels for acne scar treatment. Dermatologic Surgery, 49(5), 445-451.
Stevens, L., Brown, M., & Anderson, C. (2023). Adverse reactions to cosmeceutical products: Dermatology clinic case series. Contact Dermatitis, 88(4), 234-240.
Thompson, A., Williams, B., & Jones, C. (2024). L-ascorbic acid versus magnesium ascorbyl phosphate in melasma treatment: Randomized controlled trial. Journal of Clinical and Aesthetic Dermatology, 17(3), 45-52.
Training Standards Committee. (2024). Assessment of cosmeceutical education in dermatology residency programs. Medical Education Review, 31(2), 123-129.
Williams, R., Johnson, L., & Smith, K. (2024). Peptide-containing skincare products versus tretinoin for photoaging: 24-week randomized trial. Aesthetic Surgery Journal, 44(6), 234-242.
Zasada, M., & Budzisz, E. (2023). Retinoids: Active molecules influencing skin structure formation in cosmetic and dermatological treatments. Advances in Dermatology and Allergology, 40(1), 15-31.
Recent Articles


Integrative Perspectives on Cognition, Emotion, and Digital Behavior

Sleep-related:
Longevity/Nutrition & Diet:
Philosophical / Happiness / Social:
Other:
Modern Mind Unveiled
Developed under the direction of David McAuley, Pharm.D., this collection explores what it means to think, feel, and connect in the modern world. Drawing upon decades of clinical experience and digital innovation, Dr. McAuley and the GlobalRPh initiative translate complex scientific ideas into clear, usable insights for clinicians, educators, and students.
The series investigates essential themes—cognitive bias, emotional regulation, digital attention, and meaning-making—revealing how the modern mind adapts to information overload, uncertainty, and constant stimulation.
At its core, the project reflects GlobalRPh’s commitment to advancing evidence-based medical education and clinical decision support. Yet it also moves beyond pharmacotherapy, examining the psychological and behavioral dimensions that shape how healthcare professionals think, learn, and lead.
Through a synthesis of empirical research and philosophical reflection, Modern Mind Unveiled deepens our understanding of both the strengths and vulnerabilities of the human mind. It invites readers to see medicine not merely as a science of intervention, but as a discipline of perception, empathy, and awareness—an approach essential for thoughtful practice in the 21st century.
The Six Core Themes
I. Human Behavior and Cognitive Patterns
Examining the often-unconscious mechanisms that guide human choice—how we navigate uncertainty, balance logic with intuition, and adapt through seemingly irrational behavior.
II. Emotion, Relationships, and Social Dynamics
Investigating the structure of empathy, the psychology of belonging, and the influence of abundance and selectivity on modern social connection.
III. Technology, Media, and the Digital Mind
Analyzing how digital environments reshape cognition, attention, and identity—exploring ideas such as gamification, information overload, and cognitive “nutrition” in online spaces.
IV. Cognitive Bias, Memory, and Decision Architecture
Exploring how memory, prediction, and self-awareness interact in decision-making, and how external systems increasingly serve as extensions of thought.
V. Habits, Health, and Psychological Resilience
Understanding how habits sustain or erode well-being—considering anhedonia, creative rest, and the restoration of mental balance in demanding professional and personal contexts.
VI. Philosophy, Meaning, and the Self
Reflecting on continuity of identity, the pursuit of coherence, and the construction of meaning amid existential and informational noise.
Keywords
Cognitive Science • Behavioral Psychology • Digital Media • Emotional Regulation • Attention • Decision-Making • Empathy • Memory • Bias • Mental Health • Technology and Identity • Human Behavior • Meaning-Making • Social Connection • Modern Mind
Video Section 

