Pediatric Immunology Breakthroughs: Can Tolerogenic Therapies Cure Autoimmune Disease?
Key Takeaways
Revolutionary tolerogenic therapies are transforming pediatric autoimmune disease treatment from chronic management to potential cures, offering hope for children with severe conditions.
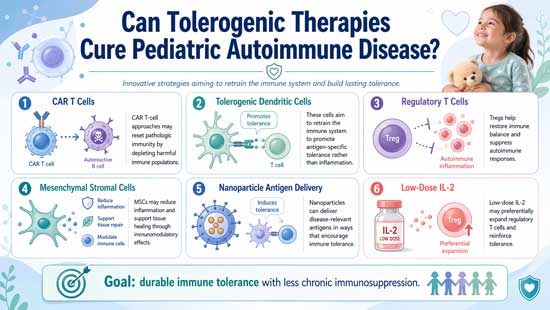
- CAR T-cell therapy achieves remarkable results: 7 of 8 pediatric patients with treatment-resistant autoimmune diseases achieved complete drug-free remission, including lupus and dermatomyositis cases.
- Multiple therapeutic approaches show promise: Tolerogenic dendritic cells, regulatory T cells, and mesenchymal stromal cells demonstrate safety and efficacy in early trials across various pediatric conditions.
- Children require specialized treatment strategies: Pediatric autoimmune diseases manifest differently than adult forms, with distinct symptom patterns and treatment responses requiring age-specific approaches.
- Safety profiles favor tolerogenic therapies: These treatments show lower toxicity than conventional immunosuppression, with treatment-related mortality of 3.3% in high-resource countries versus higher rates with traditional approaches.
- Significant barriers remain for widespread adoption: High manufacturing costs, limited accessibility, and challenges identifying disease-specific antigens hinder broader implementation of these breakthrough therapies.
The convergence of cellular engineering, personalized medicine, and improved understanding of immune tolerance mechanisms positions tolerogenic therapies as the future of pediatric autoimmune disease treatment, potentially eliminating the need for lifelong immunosuppression in favor of one-time curative interventions.
Introduction
Pediatric immunology faces a growing challenge as autoimmune diseases and allergies rise dramatically across populations. Approximately 8% of the world’s population suffers from an autoimmune disease, with children constituting 10-20% of these cases. At least 80 different forms exist, primarily affecting multiple organ systems. Traditional immunosuppressive therapies carry substantial risks for patients. Recent breakthroughs in tolerogenic therapies, specifically CAR T-cell therapy and mesenchymal stromal cell treatments, offer transformative potential for pediatric immunology disorders. This article examines emerging tolerogenic approaches, clinical evidence in pediatric populations, and the future of curative strategies for childhood autoimmune diseases.
Understanding Immune Tolerance in Pediatric Autoimmune Disease
Autoimmune disorders manifest when immune dysregulation triggers immune-mediated damage to healthy tissues and cells, resulting from breakdown of central or peripheral tolerance [1]. The immune system must maintain a delicate balance between monitoring infections from foreign pathogens while preserving tolerance to self-antigens [2]. When this equilibrium fails in pediatric populations, the consequences often differ markedly from those in adults.
What Causes Autoimmune Disease in Children
The exact pathogenesis of autoimmune disorders remains incompletely understood despite extensive research [1]. Early-onset autoimmune diseases, diagnosed before 5 years of age, are linked closely to genetic determinants, including human leukocyte antigen (HLA) and non-HLA variants, as well as gene variants associated with inborn errors of immunity (IEI) [1]. These genetic disorders may present as increased susceptibility to infections, autoinflammatory conditions, allergic diseases, or malignancies. In reality, few IEIs exist where autoimmune manifestations have never been observed [1].
More than 430 monogenic traits falling under IEI have been reported to date [3]. Genetic variants can alter various cellular functions, though the relative risk of each variant in the disease phenotype’s final effect varies considerably [1]. Mutations in genes like FOXP3 cause immunodysregulation, polyendocrinopathy, enteropathy, X-linked (IPEX) syndrome, representing prototype “Tregopathies” [3]. CTLA-4 insufficiency similarly compromises Treg function, resulting in lymphoproliferation and autoimmunity with manifestations resembling IPEX syndrome [3].
Genetics alone does not determine disease onset. Environmental triggers and epigenetic control of gene expression complete the puzzle, leading to the development of autoimmune diseases [1]. Diet and lifestyle, combined with genetics, drive autoimmunity through epigenetic mechanisms that alter gene function rather than gene structure [1]. The gut microbiome plays a prominent role in regulating immune function. When the microbiome changes adversely, the risk of autoimmune diseases increases dramatically [1]. The first 1,000 days of life, from conception to 2 years old, prove particularly critical for establishing a diverse microbiome through breastfeeding, natural childbirth, and a healthy diet [1].
Infections represent primary environmental triggers, though evidence remains often inconclusive [1]. Patients with one autoimmune disease are at increased risk of developing additional autoimmune diseases, necessitating annual screening protocols [1]. Women develop many types of autoimmune diseases more frequently than men [4], though gender distribution shows no difference between childhood-onset and adult-onset disease [5].
Central vs Peripheral Immune Tolerance
Central tolerance selectively prevents autoreactive lymphocytes from entering the periphery by mechanisms that occur in the bone marrow or thymus [6]. T cells undergo positive and negative selection in the thymus before entering the periphery to perform immune functions [7]. During thymic development, immature CD4+CD8+ double-positive thymocytes expressing successfully assembled T cell receptors (TCRs) undergo selection based on TCR affinity for self-peptides presented by medullary epithelial cells and dendritic cells [2].
Thymocytes expressing TCRs failing to recognize any self-peptide-MHC die from neglect, whereas strong recognition leads to thymocyte death or lineage deviation through negative selection [2]. The autoimmune regulator (AIRE) plays a critical role in the expression of self-antigens in the thymus, and studies of AIRE-deficient patients and animals reveal the importance of negative selection in establishing central tolerance [2]. The gene expression regulator Ikaros serves as a key mediator in educating immune cells to tolerate self-cells by regulating medullary thymic epithelial cells that mimic the characteristics of cells throughout the body [8].
Despite rigorous central tolerance mechanisms, autoreactive T and B cells escape into the periphery [6]. Peripheral tolerance mechanisms, including deletion and anergy, inhibit unwanted immune activation [6]. Regulatory T cells (Tregs) are a major immunosuppressive population that maintains peripheral immune tolerance. The transcription factor forkhead box P3 (FOXP3) serves as a master regulator of both Treg development and function [2]. Tregs suppress the activity of autoreactive T cells that escaped central deletion and T cells that cross-react with self-antigens due to molecular similarity with microbial antigens [6].
Fas-mediated cell death proves essential for normal lymphocyte homeostasis. Patients with autoimmune lymphoproliferative syndrome (ALPS) carry genetic mutations in major apoptotic inducers, including Fas and, more rarely, FasL, caspase 8, and caspase 10, leading to proliferating lymphocytes failing to undergo programmed cell death [2]. TGF-β functions as a multifunctional cytokine that regulates immune responses, acting on T cells and antigen-presenting cells to inhibit activation and differentiation while also inducing and maintaining T cell tolerance [6].
Why Children Need Different Treatment Approaches
Pediatric and adult-onset autoimmune diseases differ across multiple clinical dimensions. A comparative analysis of 342 adult patients and 79 children with systemic lupus erythematosus revealed distinct manifestation patterns [5]. Lupus nephritis occurred in 43% of pediatric patients, compared with 26.4% in adults [5]. Hematological disorders appeared in 57% of children compared to 36.4% of adults [5]. Butterfly rash manifested in 61% of pediatric cases versus 35.5% in adults [5], while mucosal ulceration occurred in 11.4% of children versus 4% of adults [5].
Conversely, neurological symptoms affected 30% of adult-onset patients but only 6% of children [5]. Polyarthritis occurred more frequently in adults at 86% versus 68% in pediatric patients [5]. Children received high-dose intravenous immunoglobulin treatment more commonly (6.3%) than adults (0.6%) [5]. Mycophenolate mofetil was prescribed to 15.2% of pediatric patients compared to 5.3% of adults [5]. These differences underscore the need to recognize distinct treatment requirements and prognostic considerations for optimal pediatric allergy and immunology care.

Ex Vivo Tolerogenic Cell Therapies: Current State
Cell-based tolerogenic therapies represent a paradigm shift from conventional immunosuppression by targeting autoreactive immune cells while preserving protective immunity [9]. These approaches manipulate cells outside the body before reinfusion, providing disease-specific immunomodulation with reduced systemic toxicity.
Tolerogenic Dendritic Cell (tolDC) Therapy
Dendritic cells function as professional antigen-presenting cells that can induce T cell tolerance in their immature state [10]. Multiple protocols generate tolDC from autologous peripheral blood monocytes using pharmacological agents, including vitamin D3, dexamethasone, rapamycin, or NF-κB inhibitors, as well as anti-inflammatory cytokines IL-10 and TGF-β [11]. Manufacturing processes incorporate maturation steps using compounds such as lipopolysaccharide or monophosphoryl lipid A to induce activation-resistant states [11].
tolDCs derived from rheumatoid arthritis patients exhibit reduced expression of costimulatory molecules, reduced proinflammatory cytokine production, and impaired stimulation of autologous antigen-specific T cells [10]. Toll-like receptor 2 expression serves as an ideal quality control marker, showing exclusive high-level expression by tolDCs [10]. These cells maintain phenotypic stability when challenged with proinflammatory mediators [10].
Clinical experience demonstrates safety across multiple conditions. Eight patients with relapsing-remitting or progressive MS received dexamethasone-tolDCs loaded with myelin peptides via intravenous injection at escalating doses of 50, 150, and 300 million cells administered three times every two weeks [11]. Treatment proved safe and well tolerated, with suggested immunoregulatory mechanisms including increased IL-10 production and reduced memory CD8+ T cells [11]. Two coordinated phase I trials delivered VitD3-tolDCs loaded with myelin peptides via intradermal or intranodal routes at doses of 5, 10, and 15 million cells, administered 6 times [11].
Phase I trials in type 1 diabetes demonstrated that tolDC therapy was well tolerated, with no serious adverse events [12]. Another study demonstrated safety and tolerability with induced immune tolerance responses lasting up to three years post-therapy [12].
Regulatory T Cell (Treg) Expansion Therapy
Regulatory T cells expressing CD4+CD25+FOXP3+ represent the principal orchestrators of peripheral tolerance [13]. Preclinical models demonstrate efficacy: Treg infusion combined with temporary CD8+ T cell depletion and rapamycin induced indefinite graft survival in heart transplantation models [14]. In non-human primate kidney transplantation, monkeys receiving bone marrow transplant with polyclonal Tregs accepted kidney grafts without immunosuppression for more than 294 days, compared to 21-28 days in controls [14].
Peripheral blood is the conventional Treg source for adult trials, yet this approach has limitations in pediatric populations due to limited blood volumes available for extraction [14]. In 2020, investigators initiated the first Treg cell therapy in transplanted children using autologous thymic tissue-derived Tregs (thyTregs) to prevent heart transplant rejection (NCT04924491) [14]. This pioneering phase I-IIa clinical trial represents the first worldwide use of thyTregs from thymic tissue removed during pediatric cardiac surgeries [14].
Long-term follow-up data from pediatric patients with type 1 diabetes who were monitored for 7.5 to 12 years after receiving autologous Treg lymphocytes demonstrated remarkable outcomes [15]. Analysis of 51 participants showed no serious adverse events, tumors, or safety concerns across more than 700 indicators [15]. Clinical benefits included higher C-peptide levels, longer remission, and insulin independence compared to standard therapy [15].
Mesenchymal Stromal Cell (MSC) Treatment
Mesenchymal stromal cells possess immunomodulatory properties enabling modulation of innate and adaptive immune responses [16]. These cells demonstrate ease of isolation from accessible tissues, scale-up in culture, and low immunogenicity, permitting allogeneic off-the-shelf applications [16]. More than one thousand clinical trials have established MSC safety and feasibility [16].
MSCs exert therapeutic effects through paracrine factors produced during microenvironment cross-talk rather than transdifferentiation [16]. They suppress T cell proliferation, B cell activation, and the production of inflammatory cytokines [17]. Allogeneic MSC transplantation may exhibit greater immunosuppressive capacity than autologous MSC transplantation [17].
Clinical applications span multiple autoimmune conditions, including graft-versus-host disease, Crohn’s disease, multiple sclerosis, refractory systemic lupus erythematosus, and systemic sclerosis [12]. In pediatric rheumatic diseases, MSC therapy addresses patients with systemic lupus erythematosus, mixed connective tissue disease, and juvenile idiopathic arthritis experiencing disease activity despite conventional agents [17].
Clinical Trial Results in Pediatric Patients
Clinical experience in children with recent-onset type 1 diabetes demonstrated the safety of tolDC infusions at two dosing schedules [14]. In pediatric rheumatic disease cases, variability exists in MSC source, cell type, and administration route [17]. Two patients received allogeneic umbilical tissue-derived MSCs, while one received autologous adipose tissue-derived MSCs [17]. Despite mild adverse effects, patients reported beneficial MSC treatment, achieving disease remission and a drastic improvement in global assessment [17].
CAR T-Cell Therapy: A Revolutionary Approach for Pediatric Immunology Disorders
Chimeric antigen receptor T-cell therapy has emerged as a potent cellular immunotherapy, initially developed for B-cell malignancies, and is now demonstrating remarkable efficacy in pediatric immunology disorders resistant to conventional treatment regimens. This approach represents a fundamental departure from suppressing immune function to precisely eliminating pathogenic B-cell populations responsible for autoimmune pathology.
How CD19-Targeted CAR T-Cells Work
The therapeutic mechanism involves extracting autologous peripheral blood T cells and genetically modifying them with lentiviral or retroviral vectors to express chimeric antigen receptors targeting CD19, a surface marker expressed across the entire B-cell lineage, including plasmablasts [18]. Unlike traditional T cell receptors, which require interaction with the major histocompatibility complex, CAR-engineered cells recognize and bind directly to CD19 antigens on target cells. Following reinfusion, the modified cells expand rapidly within the patient’s body, systematically eliminating pathological B cells through perforin/granzyme and cytokine-mediated cytotoxicity. This targeted depletion leads to reduced inflammation and restoration of immune system balance, creating conditions favorable for long-term remission without chronic immunosuppressive therapy [18].
The manufacturing process involves transducing isolated T cells with CAR cassettes that integrate into cellular DNA, followed by ex vivo expansion and purification before administration. In essence, each infused CAR T-cell functions as a “therapeutic machine,” producing numerous daughter cells that retain CAR expression, with individual cells eliminating up to 1,000 target cells through serial cytotoxic activity. These engineered lymphocytes possess unlimited tissue access, including privileged sites such as the central nervous system, enabling comprehensive B-cell depletion across all anatomical compartments.
Early Results in Pediatric Lupus and Other Conditions
A landmark case series involving eight pediatric patients aged 5 to 17 years demonstrated unprecedented outcomes in treatment-refractory autoimmune diseases [18][19]. The cohort included four children with systemic lupus erythematosus, three with dermatomyositis, and one with juvenile systemic sclerosis. Following single CAR T-cell infusions, seven patients achieved complete clinical remission, while the eighth patient with systemic sclerosis showed continuous reduction in disease severity without progression [18]. All patients discontinued immunosuppressive medications, with clinical benefits maintained throughout follow-up periods exceeding 24 months [18].
A particularly compelling case involved a 15-year-old girl with rapidly progressive lupus who required hemodialysis despite escalating treatment regimens including hydroxychloroquine, azathioprine, mycophenolate mofetil, and belimumab [11]. Following CD19 CAR T-cell therapy, her autoantibodies disappeared, complement levels normalized within six weeks, and renal function improved sufficiently to discontinue dialysis by day 17 [11]. Three juvenile patients treated across international centers experienced normalization of laboratory parameters, including dramatic reduction or disappearance of anti-double-stranded DNA antibodies [10]. The FDA has authorized REACT-01, the first CAR T-cell clinical trial in the United States specifically for children under 18 with systemic lupus erythematosus [10][20].
Safety Profile Compared to Traditional Immunosuppression
Comparative analysis reveals CAR T-cell therapy demonstrates a more favorable safety profile in autoimmune conditions than in B-cell non-Hodgkin lymphoma applications [21]. Patients with systemic lupus erythematosus experienced lower incidence and severity of cytokine release syndrome, reduced neurotoxicity consistent with immune effector cell-associated neurotoxicity syndrome, and decreased hematotoxicity despite similar CAR T-cell expansion [21]. Although neutrophil nadirs were lower in lupus patients, platelet counts remained near normal, with shorter durations of cytopenias [21].
In the CASTLE phase 1/2a study, no higher-grade cytokine release syndrome (grade 3 or 4) occurred throughout the entire trial [22]. Among juvenile patients, all three experienced mild grade 1 cytokine release syndrome, with no cases of immune effector cell-associated neurotoxicity syndrome reported [10]. CAR T-cell persistence proved consistently shorter in autoimmune diseases, while reconstitution of the adaptive immune system occurred faster with earlier recovery of conventional T and B cells [21]. This enhanced safety profile, combined with the potential for durable drug-free remission, positions CAR T-cell therapy as a transformative option for severe pediatric allergy and immunology conditions.

In Vivo Tolerogenic Strategies: Targeting Without Cell Manipulation
In vivo tolerogenic strategies circumvent the complexity and cost of ex vivo cell manipulation by directly modulating immune responses within the patient’s body. These approaches target dendritic cells and regulatory T cells via antigen-delivery systems, receptor-specific antibodies, and engineered cytokines, offering scalable alternatives for pediatric allergy and immunology applications.
Nanoparticle-Based Antigen Delivery
Phosphatidylserine-liposomes encapsulating disease-specific autoantigens demonstrate therapeutic efficacy across multiple autoimmune conditions [14]. Following intravenous administration, these apoptotic-mimicking vesicles distribute to target organs where antigen-presenting cells capture them and mount tolerogenic responses [14]. Clinical translation shows promise in rheumatoid arthritis and myasthenia gravis models [14]. The DART vaccine platform utilizes a tetrahedral framework of nucleic acids, combining antigenic peptides with RNA modifications and targeting aptamers to generate tolerogenic dendritic cells in situ [15]. In MRL/lpr lupus mice, DART effectively normalized plasma cytokine levels, glomerulonephritis, and joint lesions [15]. Tolerogenic nanoparticles co-loaded with allergens and rapamycin reduce treatment risks while enhancing antigen-presenting cell uptake, decreasing costimulatory molecule expression, and inducing antigen-specific tolerance [23].
Direct DC Receptor Targeting (DEC205, SiglecH, DCIR2)
Selective autoantigen delivery to dendritic cell receptors promotes tolerance through distinct mechanisms, depending on the targeted receptor and DC subset [24]. Myelin peptides fused to anti-DEC205 antibodies target CD8+ conventional dendritic cells and CD103+ migratory dendritic cells, suppressing experimental autoimmune encephalomyelitis, type 1 diabetes, and arthritis models [24]. Anti-DEC205 vaccines induce antigen-specific regulatory T cells and autoreactive T cell anergy dependent on TGF-β [24]. Conversely, targeting DCIR2 on CD8- dendritic cells results in antigen-specific activation and proliferative expansion of natural Foxp3+ regulatory T cells rather than extrathymic Treg induction [25]. SiglecH-targeted approaches direct antigens to plasmacytoid dendritic cells before disease immunization to establish tolerance [22]. Intravenous vaccination proves more effective than subcutaneous or intraperitoneal administration [24].
Low-Dose IL-2 Therapy for Treg Activation
Daily low-dose IL-2 administration induces selective regulatory T cell expansion and clinical improvement in chronic graft-versus-host disease, with approximately 50% of patients experiencing benefit [26]. The mechanism involves increased phosphorylated STAT5 in regulatory T cells, enhanced thymic generation, proliferation, and reduced apoptotic susceptibility [26]. In type 1 diabetes patients, doses ranging from 0.33 to 3 million international units daily for five consecutive days produced dose-dependent Treg expansion [27]. At 3 million international units daily, increased Treg levels persisted through day 60 [27]. Regulatory T cells retained 20-fold higher sensitivity to IL-2 than effector T cells and natural killer cells [27].
Engineered Cytokine Approaches (AcTaferons)
AcTaferons represent Activity-on-Target cytokines utilizing mutant interferons with severely reduced receptor affinity fused to targeting antibodies [22]. Unlike conventional immunocytokines, these engineered molecules cannot signal systemically unless cells express surface molecules recognized by the targeting moiety [22]. This design eliminates the toxic side effects associated with standard cytokine therapies while providing cell-specific immunomodulation [22]. Engineered cytokines modulate immune responses to promote tissue repair and regeneration [20].
Clinical Evidence and Real-World Outcomes in Pediatric Patients
Accumulating clinical data reveals that tolerogenic therapies produce measurable outcomes across pediatric immunology disorders, though response rates vary by condition and intervention type.
Success Rates Across Different Autoimmune Conditions
Tolerance-inducing cell-based therapies demonstrated safety with an overall frequency of serious adverse events at 0.018 (95% CI: 0.006–0.051) [28]. TolDC therapy demonstrated safety and tolerability in patients with multiple sclerosis, type 1 diabetes, and rheumatoid arthritis in phase I trials [11]. In rheumatoid arthritis, approximately 30% of patients achieved sustained remission, though 50% of these experienced flare upon treatment discontinuation [29]. For systemic lupus erythematosus, MSC treatment improved disease activity, renal function, proteinuria, and anti-dsDNA titre in cyclophosphamide/glucocorticoid-refractory patients [29]. Immune tolerance in pediatric liver transplantation proved clinically attainable and reduced lifelong immunosuppression burden [30].
Treatment-Related Mortality and Safety Data
Treatment-related mortality varied dramatically by economic setting. In high-resource countries, treatment-related mortality affected 3.3% versus 2.5% of childhood cancer patients between 2001–2010 and 2011–2021 [31]. Conversely, low- and middle-income countries experienced treatment-related mortality of 6.82% (95% CI: 5.99%–7.64%), accounting for 30.9% of overall mortality [32]. Specifically, treatment-related mortality reached 14.19% (95% CI: 9.65%–18.73%) in low-income countries, 9.21% (7.93%–10.49%) in lower-middle-income countries, and 4.47% (3.42%–5.53%) in upper-middle-income countries [33].
Long-Term Follow-Up: Remission vs Relapse Rates
Pediatric Graves disease treated with antithyroid drugs showed remission rates of 28.8% overall, increasing with treatment duration to 75% after nine years [21]. In relapsed acute lymphoblastic leukemia, five-year overall survival reached 56% and event-free survival 47% on the UK ALL R2 protocol [34]. For relapsed acute myeloid leukemia, five-year overall survival was 42% in BFM cohorts and 35% in COG cohorts [35].
Comparison with Conventional Immunosuppressive Therapy
Conventional immunosuppression rarely achieves cure or drug-free remission, requiring long-term maintenance therapy for disease control [29].
Challenges and Future Directions in Pediatric Allergy and Immunology
Despite promising clinical results, several obstacles impede the widespread adoption of tolerogenic therapies in pediatric allergy and immunology practice.
Identifying Disease-Specific Antigens
The optimal treatment strategy would specifically target disease-associated antigens rather than function as a global immunosuppressant, limiting side effects while addressing underlying disease mechanisms [36]. However, identifying autoantigens responsible for initiating autoimmune disease remains challenging [36]. Type 1 diabetes exemplifies this complexity, with several known autoantigens complicating therapeutic approaches [36]. Moreover, most autoimmune conditions manifest only after substantial tissue damage occurs [36]. Type 1 diabetes typically presents when fewer than 10-20% of functional β cells remain in the pancreas [36]. The optimal route of delivery and dosage for antigen-specific immunotherapy requires further investigation across different disease states [36].
Cost and Accessibility Barriers
Gene and cell therapies face profound commercial viability challenges despite demonstrating safety and efficacy in early-stage trials [10]. The average cost to bring a treatment to market reaches USD 2.50 billion, with success rates below 10% [10]. Clinical trial costs are particularly burdensome for rare diseases, due to geographically dispersed patient populations that require multiple international sites [10]. Long-term follow-up requirements spanning 5-15 years result in sustained monitoring expenses far exceeding those of standard small-molecule programs [10]. When companies discontinue development programs, the associated intellectual property typically remains under the original developer’s control, blocking repurposing or continuation by other entities [10].
Need for Multicenter Pediatric Clinical Trials
Coordinated action across private companies, regulatory agencies, and non-profit organizations becomes essential to explore non-traditional business models [10]. Trial discontinuation leaves patients without viable alternatives, potentially diminishing participant recruitment for future studies [10].
Personalized Medicine Approaches
Autoimmune diseases exhibit heterogeneity, with patients experiencing different manifestations based on genetics and environmental exposures [37]. Genome and RNA sequencing, combined with tissue biopsies, enable the identification of the biological underpinnings of patient symptoms [37], facilitating the development of targeted therapies tailored to individuals [37].
Tolerogenic therapies represent a paradigm shift in the management of pediatric autoimmune diseases, moving beyond chronic immunosuppression toward curative strategies. CAR T-cell therapy has demonstrated unprecedented success, with seven of eight pediatric patients achieving complete drug-free remission in treatment-refractory conditions. Similarly, mesenchymal stromal cells, tolerogenic dendritic cells, and regulatory T cell therapies show favorable safety profiles while inducing sustained immunological tolerance. Barriers remain, particularly in antigen identification, manufacturing costs, and limited accessibility across healthcare settings. Multicenter clinical trials specifically designed for pediatric populations become essential to validate these approaches. As personalized medicine advances through genomic profiling and targeted antigen delivery systems, tolerogenic therapies hold genuine promise to cure rather than merely control childhood autoimmune diseases.

FAQs
Q1. What are tolerogenic therapies, and how do they differ from traditional treatments for pediatric autoimmune diseases? Tolerogenic therapies target autoreactive immune cells while preserving protective immunity, rather than broadly suppressing the entire immune system as conventional treatments do. These approaches include CAR T-cell therapy, regulatory T cell expansion, and tolerogenic dendritic cell treatments that aim to restore immune balance and, potentially, cure autoimmune diseases rather than just controlling symptoms with lifelong immunosuppression.
Q2. How successful has CAR T-cell therapy been in treating children with autoimmune diseases? CAR T-cell therapy has shown remarkable success in pediatric autoimmune conditions. In a landmark study of 8 children aged 5-17 with treatment-refractory diseases, including lupus and dermatomyositis, 7 achieved complete clinical remission after a single infusion. All patients were able to discontinue immunosuppressive medications, with benefits maintained for over 24 months in follow-up.
Q3. Why do children with autoimmune diseases need different treatment approaches than adults? Children with autoimmune diseases present with distinct clinical patterns compared to adults. For example, pediatric lupus patients show higher rates of kidney involvement (43% vs 26.4%), blood disorders (57% vs 36.4%), and butterfly rash (61% vs 35.5%) than adults. These differences in disease manifestation, combined with children’s developing immune systems and different medication tolerances, require specialized treatment strategies tailored to pediatric populations.
Q4. Are tolerogenic therapies safe for children with autoimmune diseases? Clinical evidence demonstrates that tolerogenic therapies have favorable safety profiles in pediatric patients. CAR T-cell therapy in children with autoimmune diseases showed lower rates of serious side effects compared to cancer applications, with only mild cytokine release syndrome and no severe neurotoxicity. Long-term follow-up of pediatric patients with type 1 diabetes receiving regulatory T cell therapy for up to 12 years showed no serious adverse events, tumors, or safety concerns.
Q5. What are the main challenges preventing widespread use of tolerogenic therapies in children? The primary obstacles include identifying disease-specific antigens for targeted treatment, high manufacturing costs averaging $2.50 billion to bring therapies to market, limited accessibility, especially in low- and middle-income countries, and the need for more multicenter pediatric clinical trials. Additionally, the complexity of ex vivo cell manipulation and long-term follow-up requirements spanning 5-15 years creates significant logistical and financial barriers.
References
[1] – https://healthsciences.arizona.edu/news/stories/pediatric-autoimmune-diseases-finding-hope-research
[2] – https://pmc.ncbi.nlm.nih.gov/articles/PMC2605935/
[3] – https://www.nature.com/articles/s41423-020-00626-z
[4] – https://medlineplus.gov/autoimmunediseases.html
[5] – https://pubmed.ncbi.nlm.nih.gov/25516474/
[6] – https://pmc.ncbi.nlm.nih.gov/articles/PMC10715743/
[7] – https://www.nature.com/articles/s41392-024-01952-8
[8] – https://pediatrics.ucsf.edu/news/uncovering-how-our-immune-system-learns-tolerance
[9] – https://pmc.ncbi.nlm.nih.gov/articles/PMC3628318/
[10] – https://pmc.ncbi.nlm.nih.gov/articles/PMC12628092/
[11] – https://pmc.ncbi.nlm.nih.gov/articles/PMC10403757/
[12] – https://scholar.usuhs.edu/en/publications/cell-based-treatment-of-autoimmune-diseases-in-children/
[14] – https://www.sciencedirect.com/science/article/pii/S1549963422001216
[15] – https://pubmed.ncbi.nlm.nih.gov/39983922/
[16] – https://pmc.ncbi.nlm.nih.gov/articles/PMC8922475/
[17] – https://pmc.ncbi.nlm.nih.gov/articles/PMC8194100/
[18] – https://benzifoundation.org/car-t-therapy-autoimmune-diseases/
[20] – https://advanced.onlinelibrary.wiley.com/doi/10.1002/adtp.202500103
[21] – https://e-emj.org/journal/view.php?doi=10.12771/emj.2023.e31
[22] – https://pmc.ncbi.nlm.nih.gov/articles/PMC7241419/
[23] – https://www.sciencedirect.com/science/article/abs/pii/S0168365924000221
[24] – https://www.frontiersin.org/journals/immunology/articles/10.3389/
fimmu.2021.657768/full
[25] – https://pubmed.ncbi.nlm.nih.gov/30134798/
[26] – https://pmc.ncbi.nlm.nih.gov/articles/PMC3686517/
[27] – https://pmc.ncbi.nlm.nih.gov/articles/PMC8153751/
[28] – https://www.sciencedirect.com/science/article/pii/S1568997221001464
[29] – https://pmc.ncbi.nlm.nih.gov/articles/PMC6390030/
[30] – https://onlinelibrary.wiley.com/doi/10.1111/petr.70260?af=R
[31] – https://pmc.ncbi.nlm.nih.gov/articles/PMC11332546/
[33] – https://pubmed.ncbi.nlm.nih.gov/37517410/
[34] – https://pmc.ncbi.nlm.nih.gov/articles/PMC9259965/
[35] – https://pmc.ncbi.nlm.nih.gov/articles/PMC8151466/
[36] – https://pmc.ncbi.nlm.nih.gov/articles/PMC4918244/
[37] – https://www.autoimmuneinstitute.org/articles/personalized-medicine-for-autoimmune-diseases
Recent Articles


Integrative Perspectives on Cognition, Emotion, and Digital Behavior

Sleep-related:
Longevity/Nutrition & Diet:
Philosophical / Happiness / Social:
Other:
Video Section 

