Post-Polypectomy Surveillance: Are We Over-Scoping Patients (and Under-Scoping the High-Risk Ones)?

Key Takeaways
Post polypectomy surveillance is a critical component of colorectal cancer prevention strategies, aimed at reducing the incidence of interval colorectal cancers through timely follow up colonoscopy after adenoma removal. However, current surveillance practices frequently diverge from evidence based recommendations, resulting in both overutilization among low risk individuals and inadequate follow up among patients at high risk for advanced neoplasia. This misalignment not only increases healthcare system burden and procedural costs but also creates substantial gaps in cancer prevention efforts.
Colorectal cancer remains one of the leading causes of cancer related morbidity and mortality worldwide, yet it is also among the most preventable malignancies when premalignant lesions are identified and appropriately managed. Colonoscopy with polypectomy significantly reduces colorectal cancer incidence and mortality by interrupting the adenoma carcinoma sequence. Evidence demonstrates that a normal colonoscopy is associated with a markedly reduced risk of incident colorectal cancer, with studies reporting hazard ratios as low as 0.44 compared with unscreened populations. Despite these benefits, determining the optimal surveillance interval following polyp detection remains a persistent clinical challenge.
Current guideline recommendations stratify surveillance intervals according to polyp number, size, histologic characteristics, and completeness of examination. Patients considered low risk, particularly those with one to two small tubular adenomas measuring less than 10 mm without advanced histologic features, are generally recommended to undergo repeat colonoscopy within seven to ten years. Nevertheless, many of these patients continue to receive surveillance at substantially shorter intervals, commonly within three to five years. This pattern of over surveillance exposes patients to unnecessary procedural risks, increases healthcare expenditures, and contributes to limited endoscopic capacity that could otherwise be directed toward higher risk populations.
Conversely, patients classified as high risk often fail to receive appropriate surveillance despite having substantially elevated risk for advanced neoplasia and colorectal cancer. High risk findings include adenomas measuring 10 mm or greater, lesions with villous or tubulovillous histology, high grade dysplasia, or the presence of three or more adenomas. Current guidelines recommend repeat colonoscopy at approximately three years for these individuals. However, studies indicate that only about 30 percent of high risk patients receive surveillance within the recommended timeframe. This underutilization represents a major missed opportunity for secondary cancer prevention and may contribute to the development of interval colorectal cancers.
Several factors contribute to these discrepancies in surveillance practice. Physician uncertainty regarding evolving guideline recommendations remains a significant barrier, particularly given periodic updates and variations between professional society recommendations. Concerns related to medicolegal liability also influence clinical decision making, with many clinicians preferring earlier surveillance intervals to minimize perceived risk of missed malignancy. Additionally, fragmented healthcare systems and poor interoperability of electronic medical records frequently result in incomplete access to prior colonoscopy findings, pathology reports, and recommended follow up plans. These systemic limitations impair continuity of care and complicate risk stratification.
The quality of the initial colonoscopy procedure itself plays a central role in determining appropriate surveillance intervals. High quality colonoscopy is essential for accurate risk assessment and effective colorectal cancer prevention. Key quality indicators include adequate bowel preparation, complete cecal intubation, sufficient withdrawal time, and high adenoma detection rates. Current evidence supports a minimum withdrawal time of at least eight minutes and adenoma detection rates exceeding 35 percent as benchmarks associated with improved outcomes and reduced interval cancer risk. Inadequate examination quality may necessitate shorter surveillance intervals, further complicating adherence to standardized recommendations.
Accurate documentation of colonoscopic findings is equally critical. Detailed reporting of polyp size, morphology, location, histology, completeness of resection, and bowel preparation quality directly informs subsequent surveillance recommendations. Colon polyp size charts and standardized classification systems assist clinicians in translating endoscopic findings into evidence based surveillance intervals. Even relatively small differences in lesion size or histopathologic features can substantially alter recommended follow up timing, emphasizing the importance of precise reporting and pathology correlation.
Technological innovations offer promising solutions to many of these challenges. Electronic clinical decision support systems integrated into electronic health records have demonstrated significant improvements in guideline adherence. Studies show that automated surveillance recommendation tools can increase compliance with evidence based intervals from approximately 57 percent to 81 percent. These systems reduce variability in physician decision making by automatically incorporating colonoscopy findings, pathology results, and guideline algorithms into follow up recommendations.
Automated patient tracking and recall systems also represent an important advancement in surveillance management. These systems can identify patients due for follow up colonoscopy, generate reminders for both patients and providers, and reduce the likelihood of delayed surveillance in high risk individuals. In addition to improving patient outcomes, such approaches may help optimize healthcare resource allocation by minimizing unnecessary repeat procedures in low risk populations while prioritizing timely follow up for those at greatest risk.
The surveillance gap therefore reflects not only individual physician behavior but also broader systemic issues involving healthcare infrastructure, communication failures, and quality assurance limitations. Addressing these barriers requires a multifaceted strategy that combines evidence based guideline implementation, high quality colonoscopy standards, improved documentation practices, and robust digital health infrastructure.
This review examines the current evidence surrounding post polypectomy surveillance, including colon polyp removal findings, treatment protocols, and recommended surveillance intervals across varying risk categories. It explores how polyp number, histologic subtype, and lesion size influence surveillance timing, why three year repeat colonoscopy is critical for certain high risk patients, and how emerging technologies may improve adherence to evidence based care. Ultimately, optimizing surveillance practices represents a crucial opportunity to reduce colorectal cancer incidence, improve patient outcomes, and enhance the efficiency and sustainability of preventive gastrointestinal care.
Understanding Post-Polypectomy Surveillance Guidelines
What the current guidelines recommend
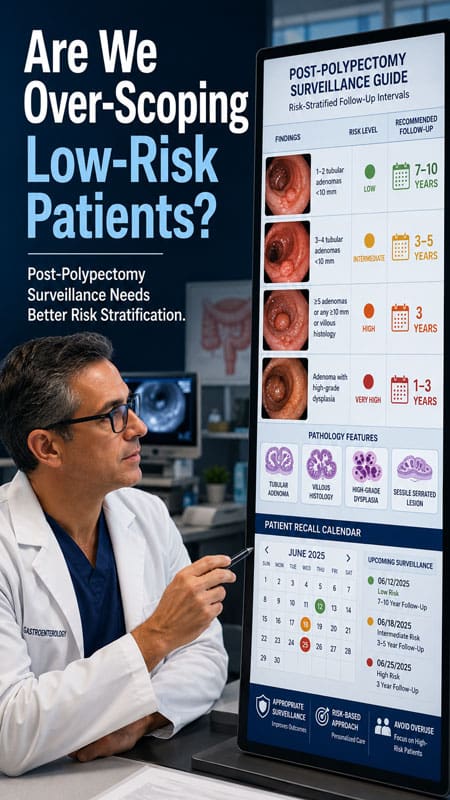
The 2020 U.S. Multi-Society Task Force on Colorectal Cancer issued updated recommendations that shifted surveillance intervals for several patient categories. For patients with normal, high-quality colonoscopy, the recommended interval extends to 10 years. The most notable change affected low-risk patients with 1-2 tubular adenomas smaller than 10 mm. These patients now receive recommendations for repeat colonoscopy in 7-10 years rather than the previous 5-10 year range. This shift reflects new evidence demonstrating that individuals with low-risk adenomas maintain reduced risk for advanced neoplasia and incident colorectal cancer on follow-up.
Patients with 3-4 tubular adenomas less than 10 mm require surveillance in 3-5 years. Those with 5-10 small tubular adenomas need colonoscopy in 3 years. Advanced adenoma findings trigger more intensive surveillance. Any adenoma measuring 10 mm or larger, containing villous histology, or showing high-grade dysplasia warrants repeat examination in 3 years. For patients presenting with more than 10 adenomas, repeat colonoscopy occurs in 1 year. This accelerated interval addresses the possibility of hereditary polyposis syndromes requiring timely clearing colonoscopy to ensure complete adenomatous lesion removal.
Serrated lesion protocols differ based on size, number, and location. Hyperplastic polyps smaller than 10 mm in the rectosigmoid region, numbering 20 or fewer, allow return to standard 10-year screening intervals. Sessile serrated polyps present unique challenges. Patients with 1-2 sessile serrated polyps smaller than 10 mm receive surveillance recommendations between 5-10 years. The interval selection depends on pathologist consistency in distinguishing sessile serrated polyps from hyperplastic polyps, bowel preparation quality, and confidence in complete excision. A 3-year interval applies when concerns exist about pathologic classification, whereas 5 years suits cases with high pathologist reliability and excellent preparation.
Traditional serrated adenomas require 3-year surveillance regardless of size. For 3-4 sessile serrated polyps under 10 mm, intervals range from 3-5 years. Any sessile serrated polyp reaching 10 mm or larger necessitates 3-year follow-up. Sessile serrated polyps containing dysplasia follow the same 3-year protocol. Piecemeal resection of adenomas or sessile serrated polyps exceeding 20 mm represents a distinct scenario requiring 6-month follow-up colonoscopy.
Risk stratification basics
Risk stratification principles established in 2006 and reinforced in subsequent updates categorize patients into two primary groups. Low-risk adenomas consist of 1-2 tubular adenomas smaller than 10 mm. High-risk adenomas include those with villous histology, high-grade dysplasia, size of 10 mm or greater, or presence of 3 or more adenomas. This classification determines surveillance intensity based on the likelihood of developing advanced neoplasia during follow-up periods.
Quality colonoscopy requirements
All surveillance recommendations assume performance of high-quality baseline colonoscopy. Quality metrics encompass colonoscopist performance and examination-specific characteristics. Adenoma detection rate benchmarks require rates exceeding 30% in men and 20% in women. The 2024 updated guidelines raised the minimum acceptable adenoma detection rate to 35% for combined screening, surveillance, and diagnostic examinations in patients aged 45 years or older.
Sessile serrated lesion detection rate emerged as a new priority quality indicator, with a target of 6% or higher. This addition reflects evidence linking low sessile serrated lesion detection to post-colonoscopy colorectal cancer. Cecal intubation rates must reach or exceed 95%[52]. Bowel preparation adequacy thresholds increased from 85% to 90%[52]. The Boston Bowel Preparation Scale provides the preferred assessment method over the Aronchick scale.
Withdrawal time in normal colonoscopies for persons aged 45 years or older increased from 6 to 8 minutes minimum. Adherence to recommended screening and surveillance intervals represents a critical quality measure, with a 90% target for consistency with Multi-Society Task Force recommendations[72]. Studies demonstrate that adenoma detection rates below 20% correlate with higher interval cancer risk in subsequent years. Similarly, cecal intubation rates affect outcomes, with endoscopists achieving 95% cecal intubation producing fewer interval cancers than those at 80%.
The Over-Surveillance Problem in Low-Risk Patients
Who are low-risk patients
Low-risk adenomas consist of 1-2 tubular adenomas smaller than 10 mm with low-grade dysplasia only. Patients with these findings demonstrate substantially reduced risk for subsequent advanced neoplasia. A pooled analysis combining data from 8 prospective studies revealed metachronous neoplasia rates of 6.9% in low-risk adenoma groups compared to 15.5% in those with advanced neoplasia on index examination. Meta-analyzes examining 7 studies found metachronous advanced neoplasia incidence ranging from 2.2% to 6.8% in individuals with low-risk adenomas. Further evidence indicates that patients diagnosed with 1-2 non-advanced adenomas maintain the same long-term colorectal cancer risk and mortality as those with no adenomas.
How often colonoscopy should occur for 1-2 small adenomas
The optimal timing for post-polypectomy surveillance in patients with low-risk adenomas has remained challenging for clinicians and guideline developers over four decades. Patients with only 1-2 small tubular adenomas with low-grade dysplasia should undergo follow-up colonoscopy in 7-10 years. For those who underwent colonoscopy before 2020 and had 1-2 small adenomas detected, subsequent colonoscopy may occur in 5-10 years based on earlier guidelines, with precise timing determined by clinical factors including prior findings, family history, and patient preferences. When initial colonoscopy showed 1-4 adenomas less than 10 mm and follow-up colonoscopy returns normal results, the subsequent examination interval extends to 10 years.
The shift from 5-year to 10-year intervals
Guideline evolution reflects accumulating evidence about low-risk adenoma outcomes. In 2003, the U.S. Multi-Society Task Force recommended surveillance in 5 years for patients with 1-2 small tubular adenomas, based largely on low risk for advanced adenomas on follow-up colonoscopy 3-5 years later. The 2006 USMSTF broadened the recommended interval to 5-10 years, relying on observational cohort studies demonstrating low incidence of subsequent advanced adenomas and colorectal cancer after low-risk adenoma removal on baseline examination. The 2012 update continued supporting the 5-10 year interval. The 2020 recommendations extended this further to 7-10 years, reflecting new studies confirming that individuals with low-risk adenomas maintain reduced risk for advanced neoplasia and incident colorectal cancer.
Why physicians over-scope low-risk cases
Despite decades of evidence and guideline recommendations, studies consistently demonstrate that clinicians bring back patients with low-risk adenomas for surveillance earlier than the recommended minimum of 5 years. This pattern suggests overuse of surveillance colonoscopy, which already accounts for more than 25% of colonoscopies performed annually. One study found that 38.1% of subjects with low-risk findings received 3-year surveillance interval recommendations rather than the guideline-recommended 5 years. In comparisons of outcomes, no differences emerged between the 3-year and 5-year recommendation groups in proportions of subjects found to have one or more adenomas (38.8% versus 41.7%), advanced adenomas (7.7% versus 8.2%), or clinically relevant serrated polyps (10.0% versus 10.3%) at follow-up colonoscopy.
Factors most substantially associated with recommendation of 3-year versus 5-year surveillance intervals included African American race (relative risk 1.41 compared to white patients), Asian/Pacific Islander ethnicity (relative risk 1.7 compared to white patients), detection of 2 adenomas at index examination (relative risk 1.47 versus 1 adenoma), more than 3 serrated polyps at index examination (relative risk 2.16), or index examination with fair or poor quality bowel preparation (relative risk 2.16 versus excellent quality). Subjects receiving 3-year recommendations were substantially less likely to have excellent bowel preparation on index examination (23.2% versus 42.1%). Endoscopists recommending shorter intervals averaged 1.4 years younger than those recommending 5-year intervals and came from specialties other than gastroenterology (10.5% versus 5.9%). Overuse of colonoscopy taxes the healthcare system and may compromise care quality, with long waiting times of several months already occurring in some regions, potentially reducing access for symptomatic patients and those with limited means.
The Under-Surveillance Problem in High-Risk Patients
Defining high-risk polyp findings
Advanced neoplasia encompasses adenomas measuring 10 mm or larger, adenomas with tubulovillous or villous histology, adenomas with high-grade dysplasia, or presence of invasive cancer. An adenoma meeting any of these criteria without invasive colorectal cancer receives classification as an advanced adenoma. Patients with 3 or more adenomas belong to a high-risk category due to elevated risk of metachronous advanced neoplasia. Neoplastic polyps, which include adenomas and serrated adenomas, warrant removal to reduce cancer risk. Adenomas account for approximately two-thirds of colon polyps and carry precancerous potential. Traditional serrated adenomas require 3-year surveillance regardless of size.
Why repeat colonoscopy in 3 years for advanced adenomas
Advanced adenomas carry a 5 percent annual risk of developing into cancer, with risk escalating as patients age. By age 55, the annual risk reaches 25 percent, and by age 80, it rises to 40 percent. Studies demonstrate that surveillance colonoscopy after baseline removal of adenoma with high-risk features may reduce risk for incident colorectal cancer. The 10-year cumulative probability of colorectal cancer in patients with advanced adenomas was 2.05% with exposure to subsequent surveillance colonoscopy versus 6.22% without surveillance. A French cohort study of 5,779 patients with adenoma found that baseline advanced adenoma associated with a 2.2-fold increased colorectal cancer risk compared to the general population. The risk fell to that of the general population if patients with advanced adenoma had at least one follow-up colonoscopy, while patients without follow-up colonoscopy faced more than 4 times higher risk.
Furthermore, a cohort of 6,972 patients with adenomas showed a 2.9-fold increased risk for incident colorectal cancer compared to the general population, with annual reported risk of 0.43% per year. Patients with either advanced adenoma or large serrated polyps demonstrate higher likelihood of developing colorectal cancer. Those with advanced adenomas showed a multivariable hazard ratio of 4.07 for cancer development. Patients with adenomas containing tubulovillous histology had a hazard ratio of 3.17, villous adenoma 8.51, and high-grade dysplasia 5.95.
Missing surveillance in patients who need it most
Despite guidelines recommending 3-year surveillance intervals for high-risk adenomas, adherence remains problematic. In a large retrospective cohort within community practice, only 30.7% of patients diagnosed with advanced tubular adenoma and 19.5% of patients with 3 or more non-advanced adenomas completed surveillance by 3 years. Another study examining surveillance uptake found that only 25.4% of patients with high-risk adenomas completed surveillance at 3 years, rising to 42.5% at 3.5 years. A multisite cohort spanning 4 regional health systems showed surveillance uptake at 3.5 years ranging between 18.3% and 59.5% for patients with high-risk features.
The consequences of inadequate surveillance manifest in post-polypectomy cancer development. One study reported that 3% of patients with advanced polyps in a surveillance program developed interval colorectal cancer. Among 4,610 patients who had follow-up colonoscopy within 10 years after advanced adenoma removal, 1.8% developed colorectal cancer at the polypectomy site and 1.2% developed cancer at a distinct site. Notably, one-third of patients developed cancer at the polypectomy site despite following appropriate surveillance intervals. The 1-, 5-, and 10-year cumulative incidences of cancer were 0.3%, 1.1%, and 1.6%, respectively.
Colon polyp size chart and risk correlation
The size of a colon polyp correlates directly with its cancer potential. Polyps measuring at least 10 millimeters in diameter meet criteria for advanced classification. Research shows that 91 percent of all advanced adenomas and 100 percent of cancer cases originated from individuals with lesions of 10 mm or larger. A 2018 study evaluating over 500,000 polyps demonstrated that a 10 mm polyp has over six times the risk of a 6 to 9 mm polyp and more than 22 times the risk of a 1 to 5 mm polyp. Studies revealed that 25% of polyps smaller than 10 mm displayed advanced adenoma features. An adenoma may take approximately 10 years to become cancerous, though this timeline shortens in patients with hereditary syndromes.
Common Polyp Types and Their Surveillance Intervals
Classification systems distinguish epithelial polyps based on histologic architecture, cellular composition, and malignant potential. Tubular adenomas account for 80% of all adenomas, representing the most frequently encountered neoplastic polyp type. Their malignant transformation rate at diagnosis reaches 4.8%, whereas tubulovillous adenomas exhibit 19.0% malignancy potential and villous adenomas 38.4%. These differences in cancer risk directly influence surveillance protocols and necessitate accurate pathologic classification.
Tubular adenomas and colon polyps treatment
Colon polyp removal occurs through polypectomy during colonoscopy, wherein providers employ wire loops or forceps to excise the adenoma from the colonic lining. Small adenomas permit removal in one piece, yet larger lesions require piecemeal resection involving extraction in multiple fragments. Patients undergoing piecemeal resection of adenomas exceeding 20 mm need 6-month follow-up colonoscopy to verify complete polypectomy site clearance. Incomplete resection represents a concern for sessile serrated polyps measuring 5 to 10 mm, nearly half of which undergo removal using cold biopsy despite guidelines recommending en bloc snare polypectomy for 6 to 9 mm colon polyps.
Surveillance intervals after tubular adenoma removal depend on adenoma number and size. Detection of 5-10 tubular adenomas smaller than 10 mm triggers 3-year surveillance. Any tubular adenoma measuring 10 mm or larger necessitates 3-year follow-up regardless of adenoma count. Villous histology presence mandates 3-year surveillance even when adenoma size remains below 10 mm.
Sessile serrated lesions
Sessile serrated polyps comprised 2.3% of screening colonoscopy findings in a cohort of 31,932 participants. Approximately 80% of sessile serrated polyps localize proximal to the sigmoid colon. These lesions exhibit endoscopically subtle characteristics including flat shape, indiscrete edges, color similarity to surrounding mucosa, and mucus coverage. Such morphological features complicate detection and contribute to missed lesions on screening colonoscopy.
The serrated pathway accounts for 20% to 30% of colorectal cancers. Sessile serrated polyps share molecular features with interval cancers, including microsatellite instability, hypermethylation, and BRAF sequence variations. This association underscores the clinical importance of detecting these lesions during colonoscopy. For 1-2 sessile serrated polyps smaller than 10 mm, repeat colonoscopy occurs in 5-10 years. Sessile serrated polyps measuring 10 mm or larger require 3-year surveillance. Traditional serrated adenomas warrant 3-year follow-up regardless of size.
Hyperplastic polyps
Hyperplastic polyps constitute up to 50% of sigmoid and rectal polyps, typically measuring 1 to 5 mm. These lesions exhibit serrations limited to the upper crypt portion, distinguishing them histologically from sessile serrated polyps. Small hyperplastic polyps smaller than 10 mm in the rectosigmoid region rarely display dysplasia or progress to colorectal cancer. Patients with 20 or fewer hyperplastic polyps smaller than 10 mm in the rectum or sigmoid colon may return to 10-year screening intervals. Conversely, hyperplastic polyps measuring 10 mm or larger necessitate 3-5 year surveillance.
Traditional serrated adenomas
Traditional serrated adenomas account for less than 5% of all serrated polyps, with prevalence reaching 0.4% in screening populations. These lesions frequently occur synchronously with advanced conventional adenomas, including large adenomas, those with villous elements, and adenomas containing high-grade dysplasia. Smoking associates with traditional serrated adenoma development, similarly to its link with sessile serrated polyps and conventional adenomas. Older age correlates with traditional serrated adenoma presence but distinguishes these lesions from sessile serrated polyps, which show no age association. All traditional serrated adenomas require 3-year surveillance colonoscopy.
Why the Surveillance Gap Exists
Lack of access to prior colonoscopy data
Documentation and communication failures impair surveillance adherence across healthcare systems. Electronic health record systems often fail to capture prior colonoscopy findings when patients receive care across multiple institutions. Ransomware actors specifically target gastroenterology practices because complete patient records, including colonoscopy findings and pathology results, cannot be canceled or reissued once stolen. For clinicians making surveillance decisions, the inability to retrieve prior colonoscopy records creates immediate clinical disruption. Office systems frequently lack written policies for colorectal cancer surveillance, and availability of tracking systems remains limited. Only one-third of surveyed physicians reported written colorectal cancer screening policies in their practices. Without adequate tracking mechanisms, identifying patients due for surveillance becomes problematic, and results of screening and diagnostic tests may not be documented properly.
Physician uncertainty and liability concerns
Post-polypectomy surveillance occurs sooner than guideline recommendations, driven by physician intolerance of uncertainty. Physicians might disagree with guidelines, distrust them, remain unfamiliar with recommendations, or consider factors beyond evidence-based intervals when making decisions. In particular, concerns about suboptimal colon preparation, patient preference or worry, and potential legal liability of missed cancers influence surveillance timing. Quality of preparation associates strongly with deviation from guideline recommendations. Endoscopists understandably overuse colonoscopy when bowel preparation quality reaches only fair because they worry that small adenomas might have been missed. Some endoscopists simply distrust research used to support guideline recommendations or express concern about medicolegal issues.
Interval cancer concerns impact physician behavior regarding surveillance intervals and contribute to early repeat examinations in some cases. Studies demonstrate that 19% to 27% of interval cancers occur in the same portion of the colon as the site of prior polypectomy. Up to 17% of lesions measuring 10 mm or larger are missed with optical colonoscopy. Consequently, these missed lesion rates and interval cancer reports heighten physician anxiety about extending surveillance intervals.
Patient preferences and anxiety
Anxiety associated with colonoscopy represents a commonly cited barrier to acceptance of screening recommendations. Studies using the State-Trait Anxiety Inventory reported mean scores ranging between 33.4 and 50.0 out of a total score of 80 on the day of colonoscopy. Among patients who missed or did not attend colonoscopy, 36% to 71% expressed anxiety about the bowel preparation process, 46% to 52% feared anticipated pain from colonoscopy, and 21% to 32% worried about suffering complications. As many as 45% of patients harbored fears about the diagnosis of cancer.
Optimal patient communication in the post-colonoscopy context proves difficult. Sedation with midazolam, fentanyl, and propofol impairs memory retention. Over 98% of colonoscopies in the United States involve sedation, and 79% of providers report giving abnormal results immediately post-procedure, even when the patient has been sedated. Research found that 73% of patients did not recall their post-procedure instructions the next day. Furthermore, 21.1% of individuals left their colonoscopy without knowing how to obtain final results.
Healthcare system factors
Competing health demands make it difficult to address screening during routine office visits. Inadequate use of office systems has been identified as a major barrier for achieving appropriate surveillance. Time constraints during busy clinic days compound communication challenges. Privacy limitations in clinical settings where results are disclosed in areas separated only by curtains create additional barriers. Certain times of the day or week experience patient influx that causes appointments to become backed up, increasing wait times before, during, and after the colonoscopy procedure.
Real-World Adherence to Guidelines
What studies show about actual practice
Studies measuring post-polypectomy guideline adherence reveal persistent gaps between recommendations and clinical practice. A meta-analysis pooling 3 studies totaling 2,147 cases found overall adherence to the 2020 USMSTF guidelines reached 66.9%. This falls substantially below the 95% target standard proposed by the European Society of Gastrointestinal Endoscopy. Adherence to earlier 2012 guidelines demonstrated even greater nonadherence, with rates exceeding 50% in multiple studies.
Recent institutional data following the 2020 guideline release shows more encouraging results at some centers. One institution reported 85% overall adherence among 1,041 procedures, with nonadherence attributable to shorter-than-recommended surveillance intervals in nearly all instances. For procedures with fewer than 3 low-risk polyps, adherence reached 82.6%, while procedures involving 3 to 4 low-risk tubular adenomas achieved 95.8% adherence. However, larger polyp size negatively predicted guideline adherence, with each 1-mm increase in the largest polyp reducing the odds of adherent recommendations.
Physician characteristics influence adherence patterns considerably. Colonoscopists performing fewer than 20 colonoscopies monthly demonstrated lower guideline adherence compared to those performing more than 100 procedures. Academic physicians achieved higher adherence rates, possibly due to increased peer review opportunities and educational access. Approximately 40% of surveyed gastroenterologists scored at a failing level on guideline knowledge assessments. Geographic variation exists as well, with the Western United States demonstrating higher adherence compared to other regions.
The challenge of implementing 10-year intervals
Bayesian changepoint analysis identified a rapid shift in practice following the 2020 guideline publication. Adherence increases began 38.9 days after January 1, 2020, coinciding with guideline release and suggesting quick adoption of interval changes. Despite this rapid uptake, providers consistently recommend inappropriately short intervals. One safety net system review found 24.5% of initial surveillance recommendations fell shorter than guideline recommendations, while only 3.6% extended too long.
Educational interventions show promise but require sustained effort. When endoscopists received wallet cards, posted guidelines near workstations, and participated in monthly guideline discussions, adherence improved from 57% to 81%. However, reminder letters alone proved insufficient to produce meaningful adherence changes.
Documentation and communication failures
Primary care providers report substantial difficulties receiving colonoscopy reports and integrating pathology results. Communications arrive through multiple routes including electronic medical records, mail, fax, or mixed approaches, with some reports never received. Pathology information frequently appears incomplete or separated from endoscopic findings, creating confusion about final surveillance recommendations. Providers suggest consolidating results before transmission and prioritizing impression statements at report tops. Current guidance emphasizes comprehensive documentation as a quality measure but neglects practical information presentation or reliable communication methods to referring providers.
Getting Surveillance Right: A Practical Approach
When to extend surveillance intervals
Clinicians may extend surveillance intervals when specific quality and pathology criteria align favorably. For patients with 1-2 tubular adenomas smaller than 10 mm completely removed at high-quality examination, repeat colonoscopy occurs in 7-10 years. Normal colonoscopy findings permit return to 10-year screening intervals. Sessile serrated polyps measuring less than 10 mm, numbering 1-2, allow surveillance extension to 5-10 years. The decision favoring a 5-year interval over 3 years applies when low concerns exist regarding pathologist consistency in distinguishing sessile serrated polyps from hyperplastic polyps, adequate bowel preparation quality, and confident complete polyp excision.
When to shorten surveillance intervals
Advanced adenoma features necessitate accelerated surveillance protocols. Any adenoma measuring 10 mm or larger requires 3-year follow-up. Adenomas containing villous histology or high-grade dysplasia mandate 3-year intervals regardless of size. Patients with 3-4 tubular adenomas smaller than 10 mm need surveillance in 3-5 years. Detection of 5-10 adenomas triggers 3-year colonoscopy. More than 10 adenomas warrants 1-year surveillance and genetic counseling. Traditional serrated adenomas require 3-year intervals. A 3-year follow-up interval receives preference when concern exists about pathologist consistency, bowel preparation quality, or complete polyp excision.
The role of piecemeal colon polyp removal
Piecemeal resection of adenomas or sessile serrated polyps exceeding 20 mm requires 6-month follow-up colonoscopy. Recent evidence suggests 12-month initial surveillance may prove acceptable for selected low-risk lesions, potentially saving cost, risk, and inconvenience. Lesions assigned to 12-month surveillance averaged smaller size (25.9 mm versus 37.0 mm), demonstrated more serrated histology (63.4% versus 9.6%), and underwent cold piecemeal resection more frequently (74.6% versus 20.4%). Recurrence rates at 6 months and 12 months reached 10.0% in both intervals. Reasonable criteria defining candidates for 12-month initial surveillance include size 20 to 30 mm and absence of high-grade dysplasia.
Balancing benefits and harms of colonoscopy
Modeling studies demonstrate that colonoscopy screening every 10 years from ages 50 through 75 years yields 270 life-years gained per 1000 persons. Harm from screening reaches at most 23 complications per 1000 persons screened. Surveillance colonoscopy after baseline removal of adenoma with high-risk features may reduce incident colorectal cancer risk. The incremental impact of surveillance colonoscopy after baseline removal of low-risk adenomas on incident and fatal colorectal cancer remains uncertain. Provided that quality examination standards are met, extension of surveillance intervals in low-risk patients balances population benefit against procedural burden.
Future Directions and System Improvements
Technological solutions offer pathways to address surveillance adherence challenges through automated systems and enhanced data infrastructure. Health systems increasingly implement multicomponent strategies at individual, provider, and system levels to improve tracking and completion of follow-up colonoscopy.
Better data sharing systems
Optimizing electronic health records with centralized information enables providers to identify patients requiring colorectal cancer screening more efficiently. Health systems develop pursuit lists within EHRs that flag patients who can benefit from screening or surveillance. Automated patient identification raised screening adherence by 24.5% in controlled studies. Interoperable electronic registries prove essential for accurately tracking screening status and targeting outreach effectively. An electronic tool embedded in patient result letters that automatically updated the EHR improved follow-up interval accuracy compared to manual entry.
Decision support tools for physicians
Clinical decision support tools embedded in workflows reduce noncompliance substantially. A web-based EMR tool requiring providers to input polyp characteristics decreased noncompliance from 19% to 9%. Automated surveillance systems using natural language processing achieved 98.7% accuracy in generating surveillance recommendations. These tools reduce inappropriate colonoscopy demand by 2.9% annually. Real-time clinical decision support alerts remind providers about screening care gaps and facilitate test ordering.
Patient education strategies
Patient navigation programs provide phone support, reminder calls, texts, and emails to engage individuals with colonoscopy orders. Health systems use patient portal messaging capabilities to send reminders, provide information about screening options, and follow up about results.
Post-polypectomy surveillance requires precise risk stratification to optimize patient outcomes. Low-risk patients receive excessive surveillance, while high-risk individuals miss critical follow-up examinations. As a matter of fact, adherence to evidence-based intervals remains below acceptable thresholds across healthcare systems. Clinicians must distinguish patients requiring 3-year surveillance from those safely managed with 7-10 year intervals. Electronic decision support tools and interoperable registries offer practical solutions to bridge this adherence gap. When implemented systematically, these interventions reduce inappropriate procedures while ensuring timely surveillance for patients at elevated cancer risk. Quality colonoscopy paired with guideline-concordant intervals represents the optimal strategy for colorectal cancer prevention.
FAQs
Q1. How long should I wait for my next colonoscopy if only 1-2 small polyps were found? If you had 1-2 small tubular adenomas (less than 10 mm) removed during a high-quality colonoscopy, current guidelines recommend waiting 7-10 years before your next colonoscopy. This extended interval is based on evidence showing that patients with these low-risk findings have a very low chance of developing advanced polyps or cancer during this timeframe.
Q2. What makes a polyp “high-risk” and how does that affect follow-up timing? A polyp is considered high-risk if it measures 10 mm or larger, contains villous features, shows high-grade dysplasia, or if you have 3 or more adenomas. These characteristics significantly increase cancer risk, requiring closer monitoring with repeat colonoscopy in 3 years rather than the longer intervals recommended for low-risk findings.
Q3. Why do some doctors recommend colonoscopies more frequently than guidelines suggest? Physicians may recommend earlier surveillance due to concerns about suboptimal bowel preparation quality, worry about missing lesions, liability concerns, patient anxiety, or unfamiliarity with current guidelines. Studies show that nearly 40% of low-risk patients receive 3-year recommendations when 5-10 years would be appropriate, contributing to overuse of colonoscopy resources.
Q4. What happens if a large polyp had to be removed in pieces? When a polyp larger than 20 mm requires piecemeal removal (removal in multiple fragments), you’ll typically need a follow-up colonoscopy in 6 months to ensure the entire polyp was removed and the site has healed properly. Some recent evidence suggests 12 months may be acceptable for certain lower-risk lesions.
Q5. Do all types of polyps require the same surveillance schedule? No, different polyp types have different surveillance intervals. Hyperplastic polyps smaller than 10 mm in the rectum or sigmoid typically allow return to 10-year screening. Sessile serrated polyps require 5-10 year follow-up if small and few in number, but 3 years if 10 mm or larger. Traditional serrated adenomas always require 3-year surveillance regardless of size.
References
[2] – https://pmc.ncbi.nlm.nih.gov/articles/PMC4409458/
[3] – https://pmc.ncbi.nlm.nih.gov/articles/PMC13077560/
[4] – https://www.giejournal.org/article/S0016-5107(19)32012-7/fulltext
[5] – https://www.irjournal.org/journal/view.php?number=327
[6] – https://gastro.org/clinical-guidance/follow-up-after-colonoscopy-and-polypectomy-a-consensus-update-by-the-u-s-multi-society-task-force-on-colorectal-cancer/
[7] – https://pmc.ncbi.nlm.nih.gov/articles/PMC6295812/
[8] – https://www.cdc.gov/pcd/issues/2011/mar/10_0081.htm
[9] – https://www.giejournal.org/article/S0016-5107(24)03194-8/fulltext
[10] – https://www.ghadvances.org/article/S2772-5723(24)00126-2/fulltext
[11] – https://gastro.org/news/does-polyp-size-matter/
[12] – https://www.aafp.org/pubs/afp/issues/2015/0115/p93.html
[13] – https://www.mayoclinic.org/diseases-conditions/tubular-adenoma-of-the-colon/diagnosis-treatment/drc-20592128
[14] – https://www.mayoclinicproceedings.org/article/S0025-6196(13)00362-5/fulltext
[15] – https://pmc.ncbi.nlm.nih.gov/articles/PMC7672705/
[16] – https://meriplex.com/cybersecurity-for-gastroenterology-practices-why-gi-data-is-a-target/
[17] – https://pubmed.ncbi.nlm.nih.gov/17686069/
[18] – https://pmc.ncbi.nlm.nih.gov/articles/PMC3199324/
[19] – https://pmc.ncbi.nlm.nih.gov/articles/PMC4114302/
[20] – https://gi.org/guideline/guidelines-for-colonoscopy-surveillance-after-screening-and-polypectomy-a-consensus-update-by-the-us-multi-society-task-force-on-colorectal-cancer/
[21] – https://pmc.ncbi.nlm.nih.gov/articles/PMC6768596/
[22] – https://journals.lww.com/ajg/fulltext/2025/10002/s557_post_polypectomy_
guideline_adherence_in_the.558.aspx
[23] – https://pmc.ncbi.nlm.nih.gov/articles/PMC7648690/
[24] – https://pmc.ncbi.nlm.nih.gov/articles/PMC10954260/
[25] – https://screeningforlife.ca/wp-content/uploads/ACRCSP-Post-Polypectomy-Surveillance-Guideline-2022_final-2.pdf
[26] – https://pmc.ncbi.nlm.nih.gov/articles/PMC5493310/
[27] – https://www.asge.org/docs/default-source/guidelines/recommendations-for-follow-up-after-colonoscopy-and-polypectomy-a-consensus-update-by-the-us-multi-society-task-force-on-colorectal-cancer-2020-march-gie.pdf
[28] – https://pmc.ncbi.nlm.nih.gov/articles/PMC11670815/
[29] – https://www.healthcareexecutive.org/archives/march-april-2023/using-technology-to-increase-colorectal-cancer-screening-rates
[30] – https://pmc.ncbi.nlm.nih.gov/articles/PMC12674911/
[31] – https://pubmed.ncbi.nlm.nih.gov/40267265/
[32] – https://journals.lww.com/ajg/fulltext/2013/10001/implementation_of_a_clinical_
decision_support.2135.aspx
[33] – https://pmc.ncbi.nlm.nih.gov/articles/PMC12868931/
[34] – https://www.gastrojournal.org/article/S0016-5085(24)05304-6/fulltext
Recent Articles


Integrative Perspectives on Cognition, Emotion, and Digital Behavior

Sleep-related:
Longevity/Nutrition & Diet:
Philosophical / Happiness / Social:
Other:
Modern Mind Unveiled
Developed under the direction of David McAuley, Pharm.D., this collection explores what it means to think, feel, and connect in the modern world. Drawing upon decades of clinical experience and digital innovation, Dr. McAuley and the GlobalRPh initiative translate complex scientific ideas into clear, usable insights for clinicians, educators, and students.
The series investigates essential themes—cognitive bias, emotional regulation, digital attention, and meaning-making—revealing how the modern mind adapts to information overload, uncertainty, and constant stimulation.
At its core, the project reflects GlobalRPh’s commitment to advancing evidence-based medical education and clinical decision support. Yet it also moves beyond pharmacotherapy, examining the psychological and behavioral dimensions that shape how healthcare professionals think, learn, and lead.
Through a synthesis of empirical research and philosophical reflection, Modern Mind Unveiled deepens our understanding of both the strengths and vulnerabilities of the human mind. It invites readers to see medicine not merely as a science of intervention, but as a discipline of perception, empathy, and awareness—an approach essential for thoughtful practice in the 21st century.
The Six Core Themes
I. Human Behavior and Cognitive Patterns
Examining the often-unconscious mechanisms that guide human choice—how we navigate uncertainty, balance logic with intuition, and adapt through seemingly irrational behavior.
II. Emotion, Relationships, and Social Dynamics
Investigating the structure of empathy, the psychology of belonging, and the influence of abundance and selectivity on modern social connection.
III. Technology, Media, and the Digital Mind
Analyzing how digital environments reshape cognition, attention, and identity—exploring ideas such as gamification, information overload, and cognitive “nutrition” in online spaces.
IV. Cognitive Bias, Memory, and Decision Architecture
Exploring how memory, prediction, and self-awareness interact in decision-making, and how external systems increasingly serve as extensions of thought.
V. Habits, Health, and Psychological Resilience
Understanding how habits sustain or erode well-being—considering anhedonia, creative rest, and the restoration of mental balance in demanding professional and personal contexts.
VI. Philosophy, Meaning, and the Self
Reflecting on continuity of identity, the pursuit of coherence, and the construction of meaning amid existential and informational noise.
Keywords
Cognitive Science • Behavioral Psychology • Digital Media • Emotional Regulation • Attention • Decision-Making • Empathy • Memory • Bias • Mental Health • Technology and Identity • Human Behavior • Meaning-Making • Social Connection • Modern Mind
Video Section 

