What a Closed Loop System for Insulin Delivery Contains: Breaking Down Artificial Pancreas Technology in 2026

Key Takeaways
Closed-loop insulin delivery systems represent a revolutionary advancement in diabetes management, combining three essential components to automate blood sugar control and significantly improve patient outcomes.
- Three core components work together: Continuous glucose monitors measure blood sugar every 5 minutes, insulin pumps deliver precise doses, and control algorithms make automated dosing decisions.
- Hybrid systems dominate 2026 market: Five FDA-approved systems (MiniMed 780G, Tandem Control-IQ, Omnipod 5, iLet, CamAPS FX) achieve 70%+ time in target range with under 2% hypoglycemia.
- ADA now recommends as standard care: The 2026 American Diabetes Association Standards designated automated insulin delivery as the preferred method for all type 1 diabetes patients and insulin-requiring type 2 diabetes adults.
- Advanced algorithms enable precision control: PID controllers, model predictive control, and AI-enhanced systems process glucose data to predict needs and adjust insulin delivery every 5 minutes automatically.
- Real-world results prove effectiveness: Clinical studies show users achieve HbA1c around 7% with significant reductions in dangerous blood sugar episodes compared to traditional insulin management methods.
This technology has evolved from experimental treatment to standard diabetes care, fundamentally transforming how patients manage their condition with minimal daily intervention required.
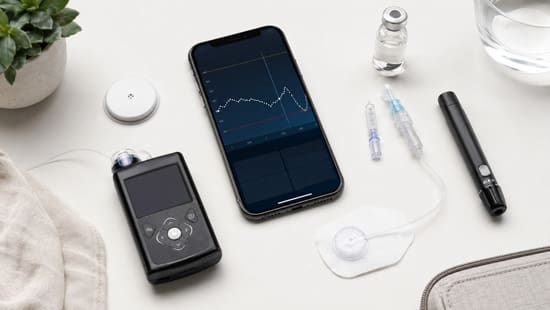
Type 1 diabetes affects 1.1 million children and young adults under age 20 globally. A closed-loop system for insulin delivery contains three essential components: a continuous glucose monitor, an insulin pump, and a control algorithm that automates insulin dosing decisions. Current-generation sensors, such as Dexcom G7 and Abbott’s FreeStyle Libre 3, achieve error rates below 9% with no calibration required. Hybrid closed-loop systems represent the most widely used artificial pancreas configuration in clinical practice. This article examines the technical components, control algorithms, FDA-approved devices, and performance metrics that define closed-loop technology in 2026.
Core Components of Closed Loop Insulin Delivery Systems
A closed-loop system for insulin delivery comprises three integrated components that operate in continuous coordination: the continuous glucose monitor measures interstitial glucose levels, the insulin pump delivers precise subcutaneous doses, and the control algorithm processes sensor data to determine appropriate insulin delivery rates.
Continuous Glucose Monitor (CGM): Real-Time Glucose Sensing
The continuous glucose monitor serves as the sensory input for closed-loop systems. A subcutaneous sensor with a small filament measures glucose concentration in interstitial fluid approximately every 5 minutes. This fluid surrounds cells beneath the skin, and glucose moves from blood vessels into this space, creating a temporal lag between blood glucose and interstitial measurements.
CGM systems consist of three elements: a disposable subcutaneous sensor, a transmitter that wirelessly transmits data, and a receiver that displays glucose values. The sensor detects glucose levels in real time, while the transmitter communicates this information to a handheld device, a smartphone application, or directly to an insulin pump. Performance is measured as mean absolute relative difference (MARD) compared to laboratory reference values, with commercially available CGMs achieving a MARD of 10% or lower. This accuracy level is comparable to standard blood glucose meters.
Real-time CGM differs from intermittently viewed systems in that it transmits data automatically without requiring manual scanning. For hybrid closed-loop systems, real-time transmission is essential, as the algorithm requires continuous glucose data to adjust insulin delivery. Some sensors require periodic fingerstick calibrations, whereas factory-calibrated models eliminate this requirement.
Insulin Pump: Automated Delivery Mechanism
Insulin pumps deliver continuous subcutaneous insulin infusion via programmable, battery-powered devices. These pumps deliver rapid- or ultra-rapid-acting insulin via Teflon or steel catheters inserted into subcutaneous tissue. Pumps can deliver insulin in increments as small as 0.01 units, enabling precise dosing adjustments that would be impossible with manual injections.
The pump provides basal insulin delivery throughout the day at pre-programmed rates, while also allowing user-initiated boluses for meals. In manual mode, basal delivery follows fixed rate profiles. In hybrid closed-loop systems, basal insulin adjusts adaptively based on current glucose demand. The algorithm converts rtCGM values into insulin doses to achieve target glucose levels.
Control Algorithm: The Decision-Making Brain
The control algorithm functions as the computational core, translating glucose measurements into insulin dosing instructions. Three primary algorithm types operate in commercial systems: proportional-integral-derivative (PID) controllers, model predictive control (MPC) algorithms, and fuzzy logic controllers.
PID controllers calculate insulin doses based on three components: deviation from target glucose (proportional), area under the curve between measured and target levels (integral), and rate of glucose change (derivative). Every 5 minutes, the rtCGM transmits a glucose value, and the algorithm calculates a microbolus based on these parameters.
MPC algorithms predict glucose levels 2 to 3 hours ahead and adjust insulin delivery at regular intervals. These predictions account for insulin absorption delays, residual active insulin from previous doses, and diurnal variability in glucose levels. The underlying models use differential mathematical equations that calculate insulin infusion rates as functions of time and changing glucose concentrations.
Fuzzy logic approaches apply rules that mimic clinical reasoning, modulating insulin delivery based on glucose rate of change, acceleration patterns, and residual active insulin[22]. These systems incorporate safety modules to prevent both hypoglycemia and hyperglycemia.
Communication Between Components
Wireless connectivity links the three components via Bluetooth or a proprietary radio frequency protocol. The CGM transmits glucose readings to the controller approximately every 5 minutes. The algorithm then processes this data and sends dosing instructions to the insulin pump, which adjusts delivery accordingly. This closed loop operates continuously, with the controller making autonomous decisions for basal insulin while users manually administer meal boluses in hybrid systems.
Types of Closed-Loop Systems Available in 2026
Closed-loop systems in 2026 fall into three distinct categories based on automation level and hormonal composition. Each configuration addresses different clinical needs and user preferences.
Hybrid Closed Loop Systems
Hybrid closed-loop systems automate basal insulin adjustments while requiring users to announce meals and deliver bolus doses. This classification represents the majority of commercially available devices. The algorithm modulates basal rates in response to CGM data, but cannot anticipate meal-related glucose excursions without user input.
Current FDA-approved hybrid systems include Medtronic MiniMed 780G, Tandem t:slim X2 with Control-IQ, Omnipod 5, iLet Bionic Pancreas, and Sequel twiist. The MiniMed 780G incorporates Meal Detection technology that uses glucose trend analysis to identify missed boluses. Real-world data from 14,899 European users showed 81.4% time in Auto Mode, achieving 72% time in range. Studies demonstrate that time spent in automated delivery correlates directly with TIR and HbA1c reduction.
The 2026 ADA Standards of Care designated automated insulin delivery as the preferred insulin administration method for all individuals with type 1 diabetes and adults with type 2 diabetes. This marks the first time AID received the strongest recommendation level for type 2 diabetes populations.
Fully Automated Closed Loop Systems
Fully closed-loop systems eliminate all user input at mealtimes, delivering therapy without carbohydrate counting or bolus announcements. Unlike hybrid configurations, these systems rely on algorithm-detected glucose rises to trigger insulin delivery. The EVOLUTION 2C feasibility study evaluated an Omnipod fully closed-loop algorithm in adults with type 2 diabetes, achieving 68% time in range with zero meal interactions. The time below range remained at 0.14%, well below the ADA threshold of 4%.
Postprandial hyperglycemia represents the primary challenge for fully automated systems. Without advance notification of meals, algorithms must react to glucose elevation rather than prevent it. The CLOSE IT study compared outcomes in type 1 diabetes: hybrid mode achieved 69% TIR, compared with 66% for fully closed-loop operation. By the same token, the FCL@Home study using neural network algorithms demonstrated 62% TIR, compared with 49% for hybrid systems.
CamAPS HX employs aggressive algorithms initially designed for hospital-based management of type 2 diabetes. More than 90% of EVOLUTION 2 participants continued into extension phases, indicating strong user acceptance.
Dual-Hormone Artificial Pancreas Systems
Dual-hormone systems deliver both insulin and glucagon, mimicking physiological pancreatic function more closely than insulin-only systems. Glucagon mini-boluses prevent hypoglycemia by rapidly releasing hepatic glucose, with onset within 5 minutes. Meta-analyses show that dual-hormone systems achieve greater time in target range and reduced time below range than single-hormone designs.
The primary barrier to commercial deployment remains the stability of the glucagon formulation. Early systems required glucagon cartridge replacement every 8 hours. Beta Bionics developed the dual-chamber iLet, which contains insulin and dasiglucagon, a chemically stable synthetic analog. Dual-hormone systems demonstrate particular effectiveness during exercise and after meals, reducing hypoglycemia events without requiring carbohydrate counting.
Control Algorithms: How Artificial Pancreas Systems Make Decisions
Control algorithms represent the computational decision-making component determining insulin delivery rates based on continuous glucose data. Three algorithm families are used in current hybrid closed-loop systems: proportional-integral-derivative controllers, model predictive control, and fuzzy logic approaches.
Proportional-Integral-Derivative (PID) Controllers
PID controllers adjust insulin delivery based on three components: the difference between measured and target glucose levels (proportional component), the area under the curve between measured and target glucose levels (integral component), and the rate of change in measured glucose levels over time (derivative component). The proportional term responds to how far glucose deviates from the target, the integral term accounts for cumulative error duration, and the derivative term reacts to the velocity of glucose change.
An insulin-on-board estimate enhances PID performance by calculating residual active insulin in the body based on clearance rates. This upper constraint prevents overinfusion and reduces the risk of hypoglycemia. Studies comparing traditional PID with IOB-enhanced PID showed 80% of virtual subjects achieved A-zone control with IOB integration versus 30% with standard PID. The control algorithm structure incorporates basal insulin infusion rate, correction factor, and insulin-to-carbohydrate ratio as personalization parameters.
Model Predictive Control (MPC) Algorithms
MPC algorithms use mathematical models of glucoregulatory systems to predict glucose excursions and adjust insulin delivery to achieve target glucose levels, accounting for estimated insulin sensitivity. The algorithm predicts future glucose levels to bring current blood glucose levels into the target range, with prediction horizons typically ranging from 30 to 120 minutes. Studies comparing MPC-based artificial pancreas systems (69% time in range) to sensor-augmented pumps (61% time in range) demonstrated the effectiveness of MPC.
Zone-MPC variants optimize insulin injections to maintain glucose within target zones rather than fixed set points. The University of California implemented zone-MPC, maintaining 80-140 mg/dL during the daytime and 110-220 mg/dL at night, with 2-hour transition periods between the ranges. MPC incorporates constraints on insulin delivery rates and accounts for the effects of meals, exercise, and diurnal variation. The algorithm uses recursive parameter identification to adapt to changes in insulin requirements, while Kalman filters update physiologic model states at each time step for personalized predictions.
Fuzzy Logic and AI-Enhanced Systems
Fuzzy-logic controllers modulate insulin delivery according to rules designed to mimic experienced clinicians’ knowledge and reasoning. These systems do not require differential equations of glucoregulatory systems, allowing clinicians to personalize dosing aggressiveness to individual requirements. Fuzzy logic handles uncertain and imprecise data through fuzzification (transforming precise sensor readings into fuzzy values), rule-based application, inference engine processing, and defuzzification (converting fuzzy outputs to precise delivery rates).
Neural-network artificial pancreas systems encode insulin dosing rules through machine learning trained on saturated datasets. Clinical trials comparing neural-network controllers to University of Virginia MPC algorithms showed identical mean glucose (130 mg/dL) and time-in-range (86.1%) performance, but neural networks demonstrated sixfold lower processing time. Fully closed-loop neural network systems achieved 72% time in range compared to 68% with usual care.
Insulin Feedback Mechanisms
Insulin feedback modifications to PID algorithms account for insulin delivery history and reduce subsequent delivery based on model-predicted subcutaneous, plasma, and interstitial insulin levels. This modification serves as a brake on insulin infusion, mitigating the tendency toward postprandial hypoglycemia. The addition of IFB to PID controllers markedly reduced hypoglycemia occurrence without increasing meal-related glucose excursions. Insulin delivery is restricted to shorter time periods under PID, with IFB providing sufficient meal coverage while achieving a more immediate decline to baseline plasma insulin levels once peak concentrations are reached.
Current Market: FDA-Approved Closed Loop Pumps and Systems
Five FDA-cleared hybrid closed-loop systems dominate the 2026 market, each offering distinct technical implementations for automated insulin delivery.
Medtronic MiniMed 780G Hybrid Closed Loop
The MiniMed 780G received FDA clearance on April 21, 2023, for individuals ages 7 and older with type 1 diabetes. The system features Meal Detection technology that identifies rising glucose trends and automatically delivers correction doses every 5 minutes. Real-world data demonstrated 80% time in range with recommended settings of 100 mg/dL target and 2-hour active insulin time. The system integrates with Guardian 4, Simplera Sync, and Instinct sensors, with the latter providing 15 days of wear. In 2026, FDA expanded clearance for ultra-rapid insulins Fiasp and Lyumjev, and for insulin-requiring type 2 diabetes.
Tandem t:slim X2 with Control-IQ
Control-IQ+ technology predicts glucose levels 30 minutes ahead and delivers AutoBolus corrections up to once hourly when glucose exceeds 180 mg/dL. The system achieved an average time in range of 74% after 12 months in real-world studies. Control-IQ+ became the first commercial AID system FDA-approved for pregnancy in April 2026, following CIRCUIT trial results showing a 12.6 percentage-point higher time in range than standard care. The platform supports Dexcom G6/G7 and Abbott FreeStyle Libre 2 Plus/3 Plus sensors.
Omnipod 5: Tubeless Loopable Pumps
Omnipod 5 is the only tubeless automated insulin delivery system approved for ages 2 and older with type 1 diabetes and for adults with type 2 diabetes. SmartAdjust technology adjusts insulin every 5 minutes, with customizable targets ranging from 110 to 150 mg/dL in 10 mg/dL increments. The waterproof Pods hold 200 units and operate for 72 hours. Smartphone control eliminates the need for a separate controller on compatible devices.
iLet Bionic Pancreas System
FDA-cleared in 2023 for ages 6 and older, the iLet requires only body weight for initialization. The adaptive algorithm eliminates carbohydrate counting by categorizing meals as small, medium, or large. Pivotal trial participants achieved HbA1c reduction from 7.9% to 7.3% over 13 weeks, with 11% more time in the target range. The system pairs with Dexcom G6, G7, and FreeStyle Libre 3 Plus.
CamAPS FX and Diabeloop Systems
CamAPS FX received FDA approval in 2024 for use in people ages 2 and older, including pregnant women. The Liberty feature enables fully closed-loop operation without meal announcements. Diabeloop DBLG2 obtained 510(k) clearance as an interoperable automated glycemic controller, compatible with multiple ACE pumps and iCGM sensors. Real-world DBLG1 studies showed time in range increased from 53% to 69.7% over 6 months.
Performance Metrics and Technical Specifications
Performance metrics quantify the effectiveness of a closed-loop system by standardized measurements of sensor accuracy, glycemic outcomes, temporal dynamics, and safety protocols.
CGM Accuracy: MARD and Sensor Performance
The mean absolute relative difference measures sensor precision relative to laboratory reference values. Dexcom G7 achieves 8.0% MARD in adults and 8.1% in children ages 7-12. Abbott FreeStyle Libre 3 Plus demonstrates 8.2% MARD for ages 6-17. Medtronic Guardian 3 reports 8.68% MARD, whereas Simplera shows 10.2% for adults and 10.9% for pediatric populations. Research indicates that MARD below 10% suffices as a reference for manual insulin dosing, while closed-loop control accepts overall MARDs around 12%. Sensor accuracy decreases during hypoglycemia compared to euglycemic ranges.
Time in Range Improvements
Control-IQ technology increased time in range from 57% to 70% over 13 weeks. Pediatric studies showed improvement from 56.7% at baseline to 69.3% after closed-loop initiation. Automated insulin delivery resulted in a 8.6% increase in TIR compared to sensor-augmented pumps. Commercial hybrid closed-loop users spend more than 70% of their time within the 70-180 mg/dL target range, achieving an HbA1c around 7%. Time below range remains under 2% in most implementations.
System Response Times and Delays
Subcutaneous rapid-acting insulin peaks approximately 90 minutes after delivery, with action persisting for up to 8 hours. Sensor delay comprises the physiological lag between blood and interstitial glucose, plus the inherent processing time of the sensor.
Safety Features and Hypoglycemia Prevention
Predictive low glucose suspend algorithms reduce nocturnal hypoglycemia without increasing hyperglycemia. Automated insulin delivery decreased time below range by 3.7 percentage points.

Closed-loop insulin delivery systems integrate three components that work in continuous coordination: continuous glucose monitors, insulin pumps, and control algorithms. Five FDA-approved hybrid closed-loop configurations dominate clinical practice in 2026, utilizing PID, model predictive control, or fuzzy logic algorithms. Real-world evidence demonstrates these systems achieve 70% or greater time in range while maintaining hypoglycemia below 2%. The 2026 ADA Standards of Care designated automated insulin delivery as the preferred method for all individuals with type 1 diabetes and adults requiring insulin for type 2 diabetes. As a result, artificial pancreas technology has transitioned from experimental intervention to standard therapeutic approach, fundamentally transforming diabetes management protocols for pediatric and adult populations.
FAQs
Q1. How does a closed-loop insulin delivery system work? A closed-loop insulin delivery system connects an insulin pump with a continuous glucose monitor (CGM) to automatically adjust insulin delivery based on real-time blood sugar readings. The CGM measures glucose levels approximately every 5 minutes, and a control algorithm processes this data to determine the appropriate insulin dose, which the pump then delivers subcutaneously. This automated coordination mimics the body’s natural glucose regulation process.
Q2. What are the main limitations of closed-loop insulin systems? Closed-loop systems are more complex than traditional insulin pumps, requiring additional setup, calibration, and sometimes programming. If a CGM sensor malfunctions or provides inaccurate readings, the system cannot properly adjust insulin delivery, potentially affecting glucose control. Additionally, most current systems are hybrid closed-loop, meaning users still need to manually announce meals and deliver bolus doses for food intake.
Q3. What is the difference between hybrid and fully automated closed-loop systems? Hybrid closed-loop systems automate basal insulin adjustments but require users to manually announce meals and deliver bolus doses for carbohydrate intake. Fully automated closed-loop systems eliminate all user input at mealtimes, relying entirely on algorithm-detected glucose rises to trigger insulin delivery without requiring carbohydrate counting or meal announcements.
Q4. How accurate are the continuous glucose monitors used in closed-loop systems? Modern CGM sensors achieve high accuracy with mean absolute relative difference (MARD) rates below 10%. For example, Dexcom G7 demonstrates 8.0% MARD in adults, while Abbott FreeStyle Libre 3 Plus shows 8.2% MARD for ages 6-17. This level of precision is comparable to standard blood glucose meters and sufficient for automated insulin dosing decisions.
Q5. What improvements in blood sugar control do closed-loop systems provide? Closed-loop systems significantly improve time in range, with users typically achieving 70% or more of their time within the target glucose range of 70-180 mg/dL. Studies show these systems increase time in range by approximately 8-13 percentage points compared to traditional insulin delivery methods, while keeping time below range under 2% and helping users achieve HbA1c levels around 7%.
References
[1] – https://pmc.ncbi.nlm.nih.gov/articles/PMC10448576/
[2] – https://my.clevelandclinic.org/health/articles/continuous-glucose-monitoring-cgm
[4] – https://www.omnipod.com/diabetes-hub/learning-center/diabetes-technology/cgms-explained
[6] – https://diatribe.org/diabetes-technology/tandems-control-iq-cleared-united-states
[7] – https://pmc.ncbi.nlm.nih.gov/articles/PMC3570847/
[9] – https://pmc.ncbi.nlm.nih.gov/articles/PMC5093196/
[12] – https://diatribe.org/diabetes-technology/mylife-camaps-fx-system-aid-all-ages
[13] – https://pmc.ncbi.nlm.nih.gov/articles/PMC9207329/
[14] – https://pmc.ncbi.nlm.nih.gov/articles/PMC4609789/
[15] – https://pmc.ncbi.nlm.nih.gov/articles/PMC9733359/
[16] – https://www.frontiersin.org/journals/endocrinology/articles/10.3389/
fendo.2021.768639/full
[17] – https://www.nature.com/articles/s41746-023-00783-1
[18] – https://pubmed.ncbi.nlm.nih.gov/23829285/
[20] – https://www.fda.gov/medical-devices/recently-approved-devices/minimed-780g-system-p160017s091
[22] – https://www.tandemdiabetes.com/products/automated-insulin-delivery/control-iq
[24] – https://www.omnipod.com/what-is-omnipod/omnipod-5
[25] – https://www.omnipod.com/current-podders/resources/omnipod-5/faqs/about-omnipod-5
[27] – https://beyondtype1.org/ilet-bionic-pancreas-automated-insulin-delivery/
[28] – https://www.drugdeliverybusiness.com/camdiab-unveils-fully-closed-loop-insulin-feature/
[29] – https://www.diabeloop.com/media-press/press-releases/diabeloop-fda-510k-clearance
[30] – https://pubmed.ncbi.nlm.nih.gov/33431420/
[31] – https://www.dexcom.com/en-us/accuracy
[32] – https://diatribe.org/diabetes-technology/study-automated-insulin-delivery-improves-time-range
[34] – https://pmc.ncbi.nlm.nih.gov/articles/PMC10698222/
[35] – https://pmc.ncbi.nlm.nih.gov/articles/PMC10755602/
[36] – https://www.sciencedirect.com/science/article/abs/pii/S2213858719301500
[37] – https://www.sciencedirect.com/science/article/pii/S240584402202936X
[38] – https://pmc.ncbi.nlm.nih.gov/articles/PMC12296855/
[39] – https://pmc.ncbi.nlm.nih.gov/articles/PMC9780196/
[40] – https://pmc.ncbi.nlm.nih.gov/articles/PMC9445345/
[41] – https://www.tandemdiabetes.com/products/automated-insulin-delivery
[42] – https://pharmacally.com/closed-loop-insulin-delivery-systems-the-new-standard/
[43] – https://tcoyd.org/2025/04/automated-insulin-delivery-systems-comparison/
[44] – https://pubmed.ncbi.nlm.nih.gov/38557128/
[45] – https://www.omnipod.com/diabetes-hub/learning-center/diabetes-technology/ada-2026
[48] – https://pubmed.ncbi.nlm.nih.gov/29337384/
[49] – https://blog.profil.com/blog/dual-hormone-artificial-pancreas-systems
Recent Articles


Integrative Perspectives on Cognition, Emotion, and Digital Behavior

Sleep-related:
Longevity/Nutrition & Diet:
Philosophical / Happiness / Social:
Other:
Modern Mind Unveiled
Developed under the direction of David McAuley, Pharm.D., this collection explores what it means to think, feel, and connect in the modern world. Drawing upon decades of clinical experience and digital innovation, Dr. McAuley and the GlobalRPh initiative translate complex scientific ideas into clear, usable insights for clinicians, educators, and students.
The series investigates essential themes—cognitive bias, emotional regulation, digital attention, and meaning-making—revealing how the modern mind adapts to information overload, uncertainty, and constant stimulation.
At its core, the project reflects GlobalRPh’s commitment to advancing evidence-based medical education and clinical decision support. Yet it also moves beyond pharmacotherapy, examining the psychological and behavioral dimensions that shape how healthcare professionals think, learn, and lead.
Through a synthesis of empirical research and philosophical reflection, Modern Mind Unveiled deepens our understanding of both the strengths and vulnerabilities of the human mind. It invites readers to see medicine not merely as a science of intervention, but as a discipline of perception, empathy, and awareness—an approach essential for thoughtful practice in the 21st century.
The Six Core Themes
I. Human Behavior and Cognitive Patterns
Examining the often-unconscious mechanisms that guide human choice—how we navigate uncertainty, balance logic with intuition, and adapt through seemingly irrational behavior.
II. Emotion, Relationships, and Social Dynamics
Investigating the structure of empathy, the psychology of belonging, and the influence of abundance and selectivity on modern social connection.
III. Technology, Media, and the Digital Mind
Analyzing how digital environments reshape cognition, attention, and identity—exploring ideas such as gamification, information overload, and cognitive “nutrition” in online spaces.
IV. Cognitive Bias, Memory, and Decision Architecture
Exploring how memory, prediction, and self-awareness interact in decision-making, and how external systems increasingly serve as extensions of thought.
V. Habits, Health, and Psychological Resilience
Understanding how habits sustain or erode well-being—considering anhedonia, creative rest, and the restoration of mental balance in demanding professional and personal contexts.
VI. Philosophy, Meaning, and the Self
Reflecting on continuity of identity, the pursuit of coherence, and the construction of meaning amid existential and informational noise.
Keywords
Cognitive Science • Behavioral Psychology • Digital Media • Emotional Regulation • Attention • Decision-Making • Empathy • Memory • Bias • Mental Health • Technology and Identity • Human Behavior • Meaning-Making • Social Connection • Modern Mind
Video Section 

