Type 2 Diabetes Mellitus as a Multifactorial Cardiometabolic Disease: Cellular Mechanisms, Reductionist Pitfalls, and Evidence-Based Prevention

Abstract
Type 2 diabetes mellitus (T2DM) is a progressive, heterogeneous, multi-organ metabolic disease characterized by chronic hyperglycemia arising from inadequate insulin secretion relative to metabolic demand, usually in the setting of insulin resistance. It is not adequately explained by a single nutrient, a single organ defect, or a single molecular lesion. Instead, T2DM develops through interacting abnormalities in adipose tissue biology, skeletal muscle glucose uptake, hepatic glucose production, pancreatic beta-cell function, incretin signaling, glucagon regulation, renal glucose handling, and central metabolic control (DeFronzo, 2009; Lu et al., 2024).
At the cellular level, chronic nutrient excess, particularly when combined with central adiposity and limited adipose storage capacity, promotes elevated non-esterified fatty acid flux, ectopic lipid deposition, accumulation of bioactive lipid intermediates such as diacylglycerols and ceramides, activation of stress kinases and protein kinase C isoforms, inhibitory phosphorylation of insulin receptor substrate proteins, impaired phosphatidylinositol 3-kinase/Akt signaling, and reduced GLUT4 translocation. Hyperinsulinemia may compensate temporarily. Over time, however, glucotoxicity, lipotoxicity, inflammation, oxidative stress, and endoplasmic reticulum stress impair glucose-stimulated insulin secretion and reduce functional beta-cell mass (Lu et al., 2024).
This review presents a clinician-level synthesis of T2DM pathophysiology while explicitly rejecting oversimplified claims such as “fat causes diabetes” or “carbohydrates cause diabetes.” Saturated fat can worsen insulin sensitivity in some mechanistic and controlled-feeding contexts, yet the broader evidence indicates that diabetes risk is shaped by dietary pattern, energy balance, body-fat distribution, physical activity, sleep, genetics, and social environment (Gaeini et al., 2022; Imamura et al., 2016).
For prevention, the strongest evidence supports sustained weight reduction, regular physical activity, and durable adherence to a high-quality dietary pattern built around minimally processed foods. Among named patterns, Mediterranean-style and other plant-forward dietary approaches currently have the strongest broad support for many adults at elevated risk (American Diabetes Association Professional Practice Committee, 2025; Talegawkar et al., 2024).
Keywords: Type 2 diabetes mellitus; insulin resistance; beta-cell dysfunction; ectopic fat; lipotoxicity; glucotoxicity; adipose tissue dysfunction; mitochondrial stress; Mediterranean diet; diabetes prevention
Recent articles. Check out our extensive video library.
Introduction
Type 2 diabetes mellitus is often discussed as though it were a straightforward disorder of excess blood sugar caused by a single dietary error. It is not. T2DM is better understood as a complex cardiometabolic disease that emerges when inherited susceptibility intersects with adverse energy balance, adipose tissue dysfunction, ectopic fat accumulation, impaired insulin action, and progressive beta-cell stress. Hyperglycemia is the clinical hallmark, but it is not the whole disease. It is the downstream expression of broader metabolic dysregulation involving the liver, skeletal muscle, adipose tissue, pancreas, kidney, gastrointestinal tract, and central nervous system (DeFronzo, 2009; Lu et al., 2024).
This distinction matters clinically. Reductionist explanations may be rhetorically powerful, but they can distort prevention and treatment by implying that a single nutrient or biologic pathway explains everything. In reality, T2DM is heterogeneous. Some patients present with central adiposity and severe hepatic insulin resistance. Others develop diabetes at relatively modest body weight, suggesting limited beta-cell reserve, altered adipose storage capacity, or both. Still others are shaped heavily by inactivity, sleep disruption, medication exposure, psychosocial stress, or broader social determinants of health (American Diabetes Association Professional Practice Committee, 2025; World Health Organization, 2024).
A modern review must therefore do two things at once. It must explain the disease down to the cellular level. It must also resist oversimplification. The sections that follow adopt that approach.
T2DM is not caused by one nutrient, one organ, or one molecular defect. It is sustained by interacting abnormalities in insulin secretion, hepatic glucose production, skeletal muscle glucose uptake, adipose tissue biology, incretin signaling, glucagon regulation, renal glucose handling, and central metabolic control (DeFronzo, 2009).
Simplified claims may be memorable. They are not biologically sufficient.
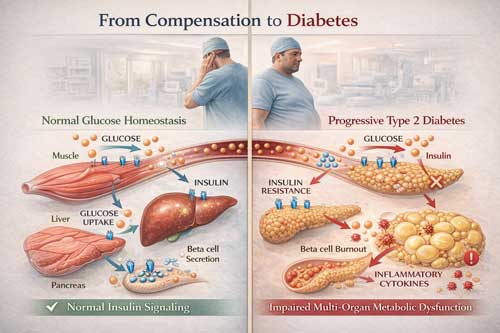
Normal Glucose Homeostasis
To understand how T2DM develops, it is useful to begin with normal physiology. In the healthy state, pancreatic beta-cells sense rising plasma glucose and respond through a tightly regulated stimulus-secretion cascade. Glucose metabolism increases the intracellular ATP/ADP ratio, closes ATP-sensitive potassium channels, depolarizes the beta-cell membrane, opens voltage-gated calcium channels, and triggers calcium-dependent exocytosis of insulin granules. This process links nutrient sensing directly to hormone release (Lu et al., 2024).
Once secreted, insulin binds the insulin receptor, a transmembrane tyrosine kinase receptor. Receptor activation phosphorylates insulin receptor substrate proteins and recruits phosphatidylinositol 3-kinase, leading to Akt activation. In skeletal muscle and adipose tissue, this signaling pathway drives the translocation of GLUT4-containing vesicles to the plasma membrane, thereby increasing glucose uptake. In hepatocytes, insulin signaling suppresses gluconeogenesis, in part through Akt-mediated inhibition of FOXO1, and promotes glycogen synthesis. These actions help maintain fasting euglycemia and facilitate postprandial glucose disposal (Lu et al., 2024).
Glucose homeostasis is not governed solely by insulin. Glucagon supports hepatic glucose output during fasting. Incretin hormones amplify meal-stimulated insulin secretion. The kidney modulates glucose handling through filtration and reabsorption. The brain influences appetite, energy expenditure, hepatic metabolism, and peripheral insulin sensitivity. Even in health, therefore, glycemic regulation is a network phenomenon. In disease, that network becomes progressively destabilized (DeFronzo, 2009).
From Compensation to Insulin Resistance
The transition from metabolic health to overt T2DM usually begins long before fasting glucose rises above diagnostic thresholds. For years, the body may compensate for worsening insulin resistance through increased insulin secretion. During that interval, conventional laboratory markers can look deceptively reassuring. The underlying biology is not reassuring. It reflects a system working harder to preserve normal glucose levels (DeFronzo, 2009; Lu et al., 2024).
Chronic nutrient excess, reduced energy expenditure, and genetically constrained adipose expandability all contribute to this transition. As adipose tissue becomes stressed, circulating fatty acid flux rises, ectopic lipid deposition increases, inflammatory signaling intensifies, and insulin action begins to falter in skeletal muscle and liver. Hyperinsulinemia compensates for a time. Eventually, compensation becomes strain, and strain becomes failure (Lu et al., 2024).
Adipose Tissue Dysfunction as an Upstream Driver
Adipose tissue is not a passive reservoir for excess calories. It is an endocrine and immunometabolic organ with central relevance to insulin sensitivity. In healthy states, adipose tissue stores surplus energy, limits ectopic fat exposure, and secretes adipokines that help preserve insulin responsiveness. When adipose tissue loses the capacity to store energy safely, that protective function begins to fail (Lu et al., 2024).
Adipocyte hypertrophy is one of the earliest turning points. As adipocytes enlarge, local hypoxia, extracellular matrix remodeling, and immune-cell recruitment become more prominent. Macrophages accumulate. Cytokine signaling intensifies. Adiponectin levels tend to fall, while lipolysis becomes less effectively suppressed. The result is increased release of non-esterified fatty acids and glycerol into the circulation, with greater substrate delivery to the liver and skeletal muscle (Lu et al., 2024).
At the molecular level, inflammatory pathways such as nuclear factor kappa B and c-Jun N-terminal kinase impair insulin action by promoting inhibitory serine phosphorylation of insulin receptor substrate proteins. Downstream PI3K-Akt signaling weakens. Insulin becomes less effective at suppressing lipolysis. The system then destabilizes further. Once adipose tissue can no longer safely buffer excess energy, metabolic stress is exported to other organs. That shift is a major organizing event in the pathogenesis of T2DM (Lu et al., 2024).
Adipose tissue is where many oversimplified diabetes narratives begin to collapse. The more clinically useful question is not simply how much fat a person carries. It is whether adipose tissue can still store energy safely. When it cannot, the burden shifts to the liver, muscle, and pancreas. That is where the disease becomes metabolically expensive.
Skeletal Muscle Insulin Resistance
Skeletal muscle is the principal site of insulin-stimulated glucose disposal after meals. For that reason, muscle insulin resistance is central to postprandial hyperglycemia. In states of chronic overnutrition, elevated fatty acid flux and impaired metabolic flexibility promote intramyocellular lipid accumulation. The key pathogenic issue is not neutral triglyceride alone, but the associated generation of bioactive lipid intermediates, particularly diacylglycerols and ceramides, which interfere with insulin signaling (Lu et al., 2024).
Diacylglycerols can activate novel protein kinase C isoforms that disrupt receptor-proximal insulin signaling. Ceramides may reduce Akt activation directly or through phosphatase-mediated pathways, impairing GLUT4 translocation and glucose uptake. As muscle becomes less able to dispose of postprandial glucose, beta-cells are forced to increase insulin output. The effect is compensatory at first. It is maladaptive over time (Lu et al., 2024).
This is also an area where interpretation must remain nuanced. Intramyocellular lipid content does not map perfectly onto insulin resistance in every setting. Endurance-trained athletes may have high muscle lipid stores while remaining insulin sensitive, a phenomenon often attributed to greater oxidative capacity and preserved metabolic flexibility. Thus, the relevant pathologic state is not simply “fat in muscle.” It is lipid overload in a context of impaired adaptation, inflammatory signaling, and defective substrate handling (Lu et al., 2024).

Hepatic Insulin Resistance and Steatotic Liver Disease
As insulin resistance progresses, the liver becomes a major driver of dysglycemia. Under normal conditions, insulin suppresses hepatic gluconeogenesis and glycogenolysis after meals. In hepatic insulin resistance, this suppression is incomplete. Endogenous glucose production remains inappropriately high, and fasting plasma glucose begins to rise (Lu et al., 2024).
Hepatic steatosis commonly accompanies this process. Excess non-esterified fatty acids from adipose tissue, increased de novo lipogenesis, and impaired disposal of hepatic lipid all contribute to fat accumulation within hepatocytes. At the intracellular level, diacylglycerol-mediated activation of protein kinase C epsilon and related signaling abnormalities impair insulin receptor activity and weaken suppression of gluconeogenic gene transcription. At the same time, VLDL production may remain elevated, contributing to the hypertriglyceridemia and atherogenic dyslipidemia frequently seen in T2DM (Lu et al., 2024).
Clinically, this matters because fasting hyperglycemia often reflects disordered hepatic glucose output rather than only impaired peripheral glucose disposal. It also helps explain the close relationship between T2DM and metabolic dysfunction–associated steatotic liver disease. Even so, fatty liver should be viewed as a major contributor, not a universal single cause. The pathophysiology extends beyond a single organ and lesion (Lu et al., 2024).
The Ominous Octet and the Multi-Organ Nature of T2DM
A narrower muscle-liver-pancreas model is clinically useful, but incomplete. DeFronzo (2009) described the “ominous octet” to emphasize that hyperglycemia in T2DM is sustained by multiple interacting defects: reduced insulin secretion, increased glucagon secretion, increased hepatic glucose production, neurotransmitter dysfunction, increased lipolysis, decreased incretin effect, increased renal glucose reabsorption, and decreased peripheral glucose uptake.
This framework remains valuable because it aligns with contemporary treatment. Metformin primarily reduces hepatic glucose production. Sodium-glucose cotransporter 2 inhibitors act on renal glucose reabsorption. Incretin-based therapies influence gut-pancreas-brain signaling, appetite, and body weight. Insulin sensitizers affect adipose and peripheral metabolism. The therapeutic landscape itself reflects the plural biology of the disease (American Diabetes Association Professional Practice Committee, 2025).
T2DM is not merely a problem of insulin deficiency or insulin resistance. It is a network disease in which multiple defects reinforce one another over time. The “ominous octet” remains clinically useful because it links pathophysiology to therapy (DeFronzo, 2009).
Beta-Cell Compensation, Stress, and Failure
T2DM becomes clinically manifest when pancreatic beta-cells can no longer secrete sufficient insulin to match the degree of insulin resistance. Early in disease development, beta-cells may compensate through hypersecretion and, in some individuals, expansion of functional mass. This adaptive phase can preserve near-normal glucose levels for years. It is not benign. It exposes beta-cells to sustained secretory pressure and cumulative metabolic stress (Lu et al., 2024).
Chronic hyperglycemia contributes to glucotoxicity, while elevated fatty acids and bioactive lipid intermediates contribute to lipotoxicity. Together, these processes impair mitochondrial ATP generation, reduce glucose-stimulated insulin secretion, alter gene expression, and increase oxidative injury. Endoplasmic reticulum stress compounds the problem by activating unfolded protein response pathways that may become maladaptive when secretory demand remains chronically high. Inflammatory signaling, amyloid-related toxicity, oxidative stress, and apoptosis all contribute to loss of functional beta-cell mass over time (Lu et al., 2024).
This progression explains an important clinical truth: insulin resistance alone does not fully determine who develops overt diabetes. The disease emerges when beta-cell reserve becomes inadequate for the imposed metabolic burden. That is why family history, ancestry, developmental exposures, and individual beta-cell fragility matter so much in clinical practice (Lu et al., 2024).
Mitochondrial Dysfunction, Oxidative Stress, and Endoplasmic Reticulum Stress
Mitochondrial biology plays a meaningful, but not exclusive, role in T2DM. In beta-cells, mitochondrial ATP production is essential for glucose-stimulated insulin secretion. In muscle and liver, mitochondrial handling of nutrient flux influences metabolic flexibility, redox balance, and substrate oxidation. Under chronic nutrient excess, mitochondrial overload may increase reactive oxygen species generation, impair oxidative phosphorylation, and contribute to both insulin resistance and beta-cell dysfunction (Lu et al., 2024).
Oxidative stress is especially relevant because beta-cells possess limited antioxidant defense capacity. Excess reactive oxygen species can damage mitochondrial DNA, proteins, and membranes while also activating stress kinases that interfere with insulin signaling. Endoplasmic reticulum stress intersects with these pathways, particularly in the setting of chronic insulin hypersecretion, and may promote misfolded protein accumulation, translational arrest, and apoptotic signaling (Lu et al., 2024).
Yet it would be inaccurate to treat mitochondrial dysfunction as the sole explanation for T2DM. The current evidence supports it as one node within a broader system that includes adipose inflammation, ectopic lipid deposition, glucagon dysregulation, incretin abnormalities, renal glucose retention, and central metabolic control. In a disease this heterogeneous, single-pathway explanations almost always overreach (DeFronzo, 2009; Lu et al., 2024).
Why “Fat Causes Diabetes” Is an Oversimplification
The statement that “fat causes diabetes” has intuitive appeal because it contains a mechanistic core. Elevated fatty acid flux, ectopic lipid deposition, and accumulation of diacylglycerols and ceramides do contribute to insulin resistance. Controlled feeding studies also suggest that replacing saturated fat with polyunsaturated fat can improve aspects of glucose-insulin homeostasis under certain conditions (Imamura et al., 2016).
But a mechanistic contribution is not the same as a complete causal explanation. Fat is not a biologically uniform exposure. Its metabolic effects vary according to fatty acid subtype, food matrix, replacement nutrient, total dietary pattern, energy balance, adiposity, and duration of exposure. A diet high in saturated fat within a hypercaloric, ultra-processed pattern is not metabolically equivalent to saturated fat consumed in a calorie-appropriate, fiber-rich, minimally processed pattern. The epidemiologic literature reflects that complexity. A recent systematic review and dose-response meta-analysis did not find a statistically significant overall association between total dietary saturated fat intake and incident T2DM across cohort studies, underscoring the limits of sweeping claims (Gaeini et al., 2022).
A more defensible conclusion is that excess energy intake, adipose dysfunction, ectopic lipid stress, and poor dietary quality are central features of T2DM pathogenesis, and that saturated fat may amplify those processes in some dietary and metabolic contexts. That is a precise statement. It is also a more honest one.
Claim: “Fat causes diabetes.”
Evidence-based correction: Certain fatty acid patterns, especially in hypercaloric and low-quality diets, may worsen insulin sensitivity. However, T2DM risk depends on dietary pattern, energy balance, adipose tissue function, ectopic fat burden, physical activity, and host susceptibility, not fat intake in isolation (Gaeini et al., 2022; Imamura et al., 2016).
Clinical takeaway: Replace slogans with mechanism.
“Fat causes diabetes” sounds decisive. The problem is that it says too little and too much at the same time. It says too little because it ignores energy balance, adipose biology, inactivity, sleep, and beta-cell reserve. It says too much because it treats a mechanistic contributor as a universal explanation.
Why “Carbohydrates Cause Diabetes” Is Also an Oversimplification
The opposing claim, that carbohydrates cause diabetes, is no better. It relies on the same conceptual mistake: treating a broad nutrient category as though it were metabolically uniform. Refined grains, sugar-sweetened beverages, and highly processed carbohydrate-rich foods are associated with greater metabolic risk, but carbohydrate-containing foods differ profoundly in structure, fiber content, energy density, glycemic effect, satiety value, and relation to the overall dietary pattern (Talegawkar et al., 2024).
That distinction matters. Whole grains, legumes, fruits, and other minimally processed carbohydrate-containing foods are repeatedly associated with lower risk of T2DM when consumed as part of high-quality eating patterns. These foods often support satiety, improve fiber intake, reduce dietary energy density, and displace less favorable foods. By contrast, refined and ultra-processed carbohydrate-rich foods can promote excess energy intake and worsen metabolic risk. The clinically meaningful contrast is therefore not carbohydrate versus fat. It is dietary quality versus dietary degradation (Talegawkar et al., 2024).
The American Diabetes Association likewise does not endorse one ideal percentage of calories from carbohydrate, protein, and fat for diabetes prevention. Instead, it emphasizes individualized, evidence-based eating patterns that improve adherence, support healthy weight, and reduce cardiometabolic risk (American Diabetes Association Professional Practice Committee, 2025).
Claim: “Carbohydrates cause diabetes.”
Evidence-based correction: Refined, ultra-processed carbohydrate-rich foods may increase risk, but high-quality carbohydrate-containing foods such as whole grains, legumes, fruits, and vegetables are associated with lower diabetes risk when part of an overall healthy dietary pattern (Talegawkar et al., 2024).
Clinical takeaway: The relevant question is not whether a food contains carbohydrate. It is what kind of carbohydrate, in what form, within what pattern.
Public debate often swings between two caricatures: “fat causes diabetes” and “carbs cause diabetes.” Neither statement survives close reading of the evidence. A stronger interpretation is that ultra-processed, energy-dense, low-fiber dietary patterns, combined with inactivity, adverse fat distribution, and limited metabolic reserve, create the conditions in which diabetes develops.
Behavioral, Environmental, and Social Drivers
The pathogenesis of T2DM extends beyond cellular biochemistry into lived behavior and environment. Physical inactivity is a major independent risk factor, in part because regular muscle contraction increases glucose uptake and improves insulin sensitivity. In the Diabetes Prevention Program, intensive lifestyle intervention reduced incident diabetes by 58% over approximately three years, and long-term follow-up demonstrated durable, though attenuated, benefit over roughly 21 years (Knowler et al., 2002; Knowler et al., 2025).
These findings remain foundational because they show that sustained weight reduction and physical activity can alter the natural history of dysglycemia. The American Diabetes Association continues to recommend at least 150 minutes per week of moderate-intensity activity and clinically meaningful weight reduction for adults at high risk, with greater benefit often seen when weight loss reaches or exceeds the 7% range originally targeted in the Diabetes Prevention Program (American Diabetes Association Professional Practice Committee, 2025).
Sleep disruption and circadian irregularity also matter. Device-based cohort data have shown that moderate and high sleep irregularity are associated with increased risk of incident T2DM, even among adults who appear to obtain adequate average sleep duration. That observation reinforces an increasingly important point: metabolic risk is shaped not only by what people eat and whether they exercise, but also by how consistently they sleep and recover (Chaput et al., 2024).
More broadly, the World Health Organization emphasizes that diabetes risk is influenced by obesity, physical inactivity, unhealthy diet, and social context. That framing is useful because it resists the temptation to reduce a complex disease to a single behavioral variable. Patients do not develop T2DM in laboratories. They develop it in real environments, under real constraints (World Health Organization, 2024).
Evidence-Based Dietary Prevention: What Pattern Helps Most?
When the prevention question is asked clinically rather than ideologically, the answer becomes clearer. The best diet for preventing T2DM is not the one attached to the strongest slogan. It is the pattern that consistently improves diet quality, supports healthy body composition, reduces visceral and ectopic fat burden, and can be sustained over time. That is why current guidance does not endorse one universal macronutrient formula (American Diabetes Association Professional Practice Committee, 2025).
The American Diabetes Association recommends evidence-based eating patterns tailored to patient needs and preferences and specifically recognizes Mediterranean and low-carbohydrate approaches as reasonable options in appropriate contexts. At the same time, the broader nutrition evidence supports dietary patterns rich in vegetables, fruits, legumes, nuts, whole grains, and fish or seafood, and lower in red and processed meats, refined grains, and sugar-sweetened foods and beverages (American Diabetes Association Professional Practice Committee, 2025; Talegawkar et al., 2024).
Among named patterns, a Mediterranean-style, plant-forward, minimally processed diet has the strongest broad support for general prevention. Its advantage lies not in ideology, but in convergence. It tends to improve overall diet quality, increase fiber and micronutrient intake, reduce reliance on ultra-processed foods, and align well with long-term adherence for many adults. Low-carbohydrate approaches may also work well in selected individuals, especially when they reduce refined food intake and support weight control, but they should be judged by food quality, sustainability, and metabolic outcome rather than carbohydrate reduction alone (American Diabetes Association Professional Practice Committee, 2025; Talegawkar et al., 2024).
There is no single ideal macronutrient ratio for preventing T2DM.
The strongest overall evidence supports a Mediterranean-style, minimally processed, plant-forward dietary pattern that helps maintain or reduce body weight and lowers intake of refined grains, sugar-sweetened beverages, and heavily processed foods (American Diabetes Association Professional Practice Committee, 2025; Talegawkar et al., 2024).
Clinical shorthand: Favor food quality, sustainability, and weight control over dietary absolutism.
Clinical Implications
A modern clinician-level model of T2DM has immediate therapeutic implications. First, treatment should begin from the premise that the disease is heterogeneous. Patients differ in dominant mechanisms, degree of adiposity, liver fat burden, beta-cell reserve, sleep quality, food environment, and capacity for sustained behavior change. A uniform explanatory script will therefore fail many patients (American Diabetes Association Professional Practice Committee, 2025; Lu et al., 2024).
Second, lifestyle intervention remains foundational, not because medication is unimportant, but because body weight, physical activity, sleep, and diet quality influence many underlying defects simultaneously. Third, pharmacotherapy should be selected with pathophysiology in mind. Metformin remains useful for hepatic glucose production. Incretin-based therapies target gut-pancreas-brain pathways and body weight. Sodium-glucose cotransporter 2 inhibitors address renal glucose retention and cardiorenal risk. Insulin sensitizers influence adipose and peripheral metabolism. A multi-defect disease often requires a multi-mechanism response (American Diabetes Association Professional Practice Committee, 2025; DeFronzo, 2009).
Conclusion
Type 2 diabetes mellitus is not a one-cause disease, and it should not be described as one. It arises through the cumulative interaction of insulin resistance, beta-cell vulnerability, adipose tissue dysfunction, ectopic lipid deposition, inflammatory and oxidative stress, behavioral risk, and social environment. The cellular details matter because they explain the physiology. The clinical details matter because they explain the patients (Lu et al., 2024; World Health Organization, 2024).
The most scientifically defensible stance is therefore anti-reductionist. Neither “fat causes diabetes” nor “carbohydrates cause diabetes” adequately captures the disease. Saturated fat can contribute in some contexts. Refined carbohydrate excess can contribute in others. But the totality of evidence points to a multifactorial process shaped by diet quality, energy balance, body-fat distribution, activity, sleep, genetics, and social environment. For prevention, the strongest overall evidence supports sustained weight management, regular physical activity, and a high-quality dietary pattern centered on minimally processed foods, with Mediterranean-style eating standing as the most broadly supported named pattern for many adults at elevated risk (American Diabetes Association Professional Practice Committee, 2025; Talegawkar et al., 2024; World Health Organization, 2024).
The most useful message for clinicians is not that one side of the nutrition debate has finally won. It is that T2DM is more biologically layered than either side usually admits. If we teach that complexity clearly, prevention advice becomes more credible, treatment becomes more individualized, and the science becomes harder to misuse.
References
- American Diabetes Association Professional Practice Committee. (2025). 3. Prevention or delay of diabetes and associated comorbidities: Standards of Care in Diabetes—2026. Diabetes Care, 49(Supplement 1), S52–S66.
- Chaput, J. P., Dutil, C., Featherstone, R., Ross, R., Giangregorio, L., Saunders, T. J., Janssen, I., Poitras, V. J., & Tremblay, M. S. (2024). Sleep irregularity and the incidence of type 2 diabetes. Diabetes Care, 47(12), 2139–2147.
- DeFronzo, R. A. (2009). From the triumvirate to the ominous octet: A new paradigm for the treatment of type 2 diabetes mellitus. Diabetes, 58(4), 773–795.
- Gaeini, Z., Bahadoran, Z., & Mirmiran, P. (2022). Saturated fatty acid intake and risk of type 2 diabetes: An updated systematic review and dose-response meta-analysis of cohort studies. Advances in Nutrition, 13(6), 2214–2228.
- Imamura, F., Micha, R., Wu, J. H. Y., de Oliveira Otto, M. C., Otite, F. O., Abioye, A. I., Mozaffarian, D., & Forouhi, N. G. (2016). Effects of saturated fat, polyunsaturated fat, monounsaturated fat, and carbohydrate on glucose-insulin homeostasis: A systematic review and meta-analysis of randomised controlled feeding trials. PLoS Medicine, 13(7), e1002087.
- Knowler, W. C., Barrett-Connor, E., Fowler, S. E., Hamman, R. F., Lachin, J. M., Walker, E. A., Nathan, D. M., & Diabetes Prevention Program Research Group. (2002). Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. The New England Journal of Medicine, 346(6), 393–403.
- Knowler, W. C., Doherty, L., Edelstein, S. L., Hamman, R. F., Lachin, J. M., Nathan, D. M., Temprosa, M., Crandall, J. P., Kitabchi, A. E., & DPP/DPPOS Research Group. (2025). Long-term effects and effect heterogeneity of lifestyle and metformin interventions on type 2 diabetes incidence over 21 years in the US Diabetes Prevention Program randomised clinical trial. The Lancet Diabetes & Endocrinology, 13(6), 469–481.
- Lu, X., Xie, Q., Pan, X., Zhang, R., Zhang, X., Peng, G., Zhang, Y., Shen, S., & Tong, N. (2024). Type 2 diabetes mellitus in adults: Pathogenesis, prevention and therapy. Signal Transduction and Targeted Therapy, 9, 262.
- Talegawkar, S., Tobias, D., Fung, T., Giovannucci, E., Hoelscher, D. M., Anderson, C. A. M., Booth, S., Deierlein, A., Gardner, C., Raynor, H., Stanford, F. C., Taylor, C., English, L. K., Reigh, N., Higgins, M., Butera, G., Callahan, E. H., Huang, S., Raghavan, R., Terry, N., & Obbagy, J. (2024). Dietary patterns and risk of type 2 diabetes: A systematic review. U.S. Department of Agriculture, Food and Nutrition Service, Nutrition Evidence Systematic Review.
- World Health Organization. (2024, November 14). Diabetes.
Recent Articles 
Integrative Perspectives on Cognition, Emotion, and Digital Behavior

Sleep-related:
Longevity/Nutrition & Diet:
Philosophical / Happiness:
Other:
Modern Mind Unveiled
Developed under the direction of David McAuley, Pharm.D., this collection explores what it means to think, feel, and connect in the modern world. Drawing upon decades of clinical experience and digital innovation, Dr. McAuley and the GlobalRPh initiative translate complex scientific ideas into clear, usable insights for clinicians, educators, and students.
The series investigates essential themes—cognitive bias, emotional regulation, digital attention, and meaning-making—revealing how the modern mind adapts to information overload, uncertainty, and constant stimulation.
At its core, the project reflects GlobalRPh’s commitment to advancing evidence-based medical education and clinical decision support. Yet it also moves beyond pharmacotherapy, examining the psychological and behavioral dimensions that shape how healthcare professionals think, learn, and lead.
Through a synthesis of empirical research and philosophical reflection, Modern Mind Unveiled deepens our understanding of both the strengths and vulnerabilities of the human mind. It invites readers to see medicine not merely as a science of intervention, but as a discipline of perception, empathy, and awareness—an approach essential for thoughtful practice in the 21st century.
The Six Core Themes
I. Human Behavior and Cognitive Patterns
Examining the often-unconscious mechanisms that guide human choice—how we navigate uncertainty, balance logic with intuition, and adapt through seemingly irrational behavior.
II. Emotion, Relationships, and Social Dynamics
Investigating the structure of empathy, the psychology of belonging, and the influence of abundance and selectivity on modern social connection.
III. Technology, Media, and the Digital Mind
Analyzing how digital environments reshape cognition, attention, and identity—exploring ideas such as gamification, information overload, and cognitive “nutrition” in online spaces.
IV. Cognitive Bias, Memory, and Decision Architecture
Exploring how memory, prediction, and self-awareness interact in decision-making, and how external systems increasingly serve as extensions of thought.
V. Habits, Health, and Psychological Resilience
Understanding how habits sustain or erode well-being—considering anhedonia, creative rest, and the restoration of mental balance in demanding professional and personal contexts.
VI. Philosophy, Meaning, and the Self
Reflecting on continuity of identity, the pursuit of coherence, and the construction of meaning amid existential and informational noise.
Keywords
Cognitive Science • Behavioral Psychology • Digital Media • Emotional Regulation • Attention • Decision-Making • Empathy • Memory • Bias • Mental Health • Technology and Identity • Human Behavior • Meaning-Making • Social Connection • Modern Mind
Video Section 

