Vancomycin for MRSA: Why Clinicians Are Reconsidering the Gold Standard in 2026

Key Takeaways
For decades, vancomycin has served as the cornerstone of treatment for infections caused by methicillin resistant Staphylococcus aureus (MRSA). Its widespread adoption established it as the default empiric therapy across a broad range of clinical settings, particularly for suspected serious gram positive infections. However, evolving resistance patterns, concerns regarding toxicity, and the availability of more effective alternatives for specific infection syndromes have prompted clinicians to reconsider its role in modern antimicrobial practice. Increasingly, MRSA management is shifting from standardized empiric coverage toward diagnostic driven, patient specific therapy that prioritizes both efficacy and safety.
The historical dominance of vancomycin is rooted in its mechanism of action, which involves inhibition of bacterial cell wall synthesis through binding to the D alanyl D alanine terminus of peptidoglycan precursors. While this mechanism initially provided reliable activity against resistant gram positive organisms, adaptive bacterial mechanisms have progressively reduced susceptibility. Since the first documented case of vancomycin resistant Staphylococcus aureus (VRSA) in Michigan in 2002, a total of 52 VRSA cases have been reported worldwide. Resistance is largely mediated by acquisition of the vanA gene cluster, typically transferred from vancomycin resistant enterococci, which alters cell wall targets and produces an estimated thousand fold reduction in vancomycin binding affinity. Although VRSA remains uncommon, the global emergence of reduced susceptibility strains signals an important warning regarding continued reliance on vancomycin as universal first line therapy.
Beyond resistance concerns, vancomycin presents significant pharmacokinetic and safety challenges. The drug possesses a narrow therapeutic window that necessitates careful monitoring to achieve adequate antimicrobial exposure while minimizing toxicity. Historically recommended trough concentrations of 15 to 20 mg/L, intended to optimize treatment of severe MRSA infections, have been associated with substantially increased nephrotoxicity. Observational data demonstrate acute kidney injury occurring in approximately 29.6 percent of patients at higher trough targets compared with 8.9 percent among patients maintained at lower concentrations. These toxicity risks have driven reevaluation of traditional dosing strategies and encouraged adoption of exposure based monitoring approaches.
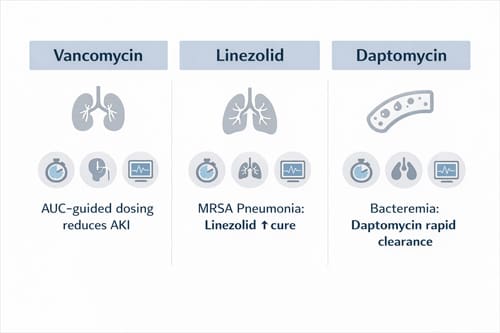
Recent pharmacodynamic research supports area under the concentration time curve (AUC) guided dosing as a safer and more precise alternative to trough based monitoring. AUC guided strategies more accurately reflect drug exposure and have been associated with markedly reduced rates of acute kidney injury, with reported incidences ranging from 0 to 2 percent compared with approximately 8 percent observed under trough only monitoring protocols. This shift represents a broader movement toward individualized antimicrobial dosing aligned with antimicrobial stewardship principles.
Simultaneously, accumulating comparative effectiveness data demonstrate that alternative agents may outperform vancomycin in specific clinical scenarios. Linezolid, an oxazolidinone antibiotic with excellent pulmonary penetration and oral bioavailability, has shown superior outcomes in MRSA pneumonia. Clinical trials report higher clinical cure rates with linezolid compared with vancomycin, approximately 57.6 percent versus 46.6 percent, alongside lower rates of nephrotoxicity at 8.4 percent compared with 18.2 percent. These findings have influenced treatment recommendations, particularly for ventilator associated and hospital acquired pneumonia where tissue penetration plays a critical role in therapeutic success.
Advances in rapid diagnostic technologies have further accelerated the movement away from empiric vancomycin exposure. Rapid MRSA polymerase chain reaction testing allows clinicians to confirm or exclude MRSA colonization or infection within hours rather than days. Negative predictive values ranging from 96 to 98 percent enable safe de escalation of vancomycin therapy within 24 to 48 hours in many clinical settings, reducing unnecessary antibiotic exposure and minimizing toxicity risk. Integration of rapid diagnostics into antimicrobial stewardship programs has therefore become a key component of modern MRSA management.
Collectively, these developments reflect a broader paradigm shift in infectious disease practice. Rather than applying blanket empiric therapy, clinicians increasingly rely on microbiologic data, pharmacokinetic optimization, infection site considerations, and patient specific risk factors to guide antibiotic selection. Precision antimicrobial therapy aims to balance antimicrobial effectiveness with minimization of adverse events and resistance selection pressure.
Despite these changes, vancomycin retains an important role in MRSA treatment. It remains an appropriate option for bloodstream infections, endocarditis, and other invasive infections when isolates demonstrate susceptibility and when patient specific factors favor its use. Cost considerations, clinician familiarity, and extensive clinical experience also contribute to its continued relevance. The goal is therefore not complete replacement but more selective and evidence based utilization.
This review examines the evolving role of vancomycin in MRSA management by exploring its mechanism of action, the clinical limitations driving the search for alternatives, and emerging comparative evidence, including outcomes in MRSA pneumonia. It also identifies clinical scenarios in which vancomycin remains an appropriate therapeutic choice while outlining how diagnostic innovation and pharmacologic optimization are reshaping antimicrobial decision making. Together, these developments signal a transition toward a more precise, patient centered era of MRSA therapy that aligns antimicrobial stewardship with improved clinical outcomes.
Vancomycin Mechanism of Action: How It Targets MRSA
Glycopeptide antibiotics function through a distinct mechanism that differentiates them from beta-lactam agents. Vancomycin exerts its bactericidal effect by inhibiting peptidoglycan polymerization in bacterial cell walls [1]. The bacterial cell wall contains a rigid peptidoglycan layer with a highly cross-linked structure composed of long polymers of N-acetylmuramic acid (NAM) and N-acetylglucosamine (NAG) [1]. Understanding this vancomycin mechanism of action provides the foundation for recognizing both its therapeutic utility and emerging limitations.
Cell Wall Synthesis Disruption Through Lipid II Binding
Vancomycin binds specifically to the D-alanyl-D-alanine (D-Ala-D-Ala) terminus of the peptidoglycan precursor lipid II [2]. This interaction occurs through the formation of five hydrogen bonds between the glycopeptide and the D-Ala-D-Ala terminus, creating a stable complex that blocks the entire peptidoglycan synthesis cycle [3]. Lipid II represents the central cell wall building block, consisting of the bactoprenol carrier linked to the disaccharide unit N-acetylmuramyl-pentapeptide-N-acetylglucosamine via a pyrophosphate bridge [3].
The binding mechanism involves critical molecular interactions. A rate-limiting step is the formation of backbone-backbone hydrogen bonds between the carbonyl group of residue 4 of vancomycin and the amide group of the terminal D-Ala of lipid II [2]. The positively charged N-terminus of vancomycin interacts with the negatively charged C-terminus of lipid II, a step required to form the final complex [2]. When vancomycin binds to D-alanyl D-alanine, it inhibits glucosyltransferase (peptidoglycan synthase) and the P-phospholipid carrier, thereby preventing the synthesis and polymerization of NAM and NAG within the peptidoglycan layer [1].
This binding sterically shields the cell wall precursor substrate from the penicillin binding proteins (PBPs) that catalyze transglycosylation and transpeptidation [3]. Lipid II sequestration prevents regeneration of the lipid transporter (C55) from lipid II, which requires transglycosylase activity [3]. Since the number of C55 copies in the bacterium is found in small numbers, vancomycin sequestration of lipid II represents a potent means of inhibiting bacterial cell wall biosynthesis [3].
Bactericidal Activity Against Gram-Positive Bacteria
The inhibition weakens bacterial cell walls and causes leakage of intracellular components, resulting in bacterial cell death [1]. When attempting to divide, the lack of a functional cell wall causes the bacterium to flood with fluid from its environment, forcing it to swell and eventually burst [1]. Vancomycin is only active against gram-positive bacteria [1].
The selectivity stems from structural barriers. Unlike penicillin, which can penetrate the lipid bilayer of gram-negative bacteria, vancomycin is nearly three times larger and carries a net positive charge [1]. This size and charge combination prevents vancomycin from entering the gram-negative bacterial cell, rendering the drug ineffective against gram-negative infections [1]. The outer lipopolysaccharide membrane that envelops the gram-negative cell wall peptidoglycan serves as a barrier, preventing penetration of large molecules like vancomycin and obstructing its access to the cell wall target [1].
Pharmacokinetic Properties: Tissue Penetration and Distribution
Oral vancomycin has a bioavailability of less than 10% [1]. Following intravenous administration, vancomycin exhibits a rapid onset of action with a serum peak concentration immediately after completion of the infusion [1]. The drug is distributed widely in body tissues and fluids, with a volume of distribution ranging from 0.4 to 1.0 L/kg, excluding cerebrospinal fluid with non-inflamed meninges [1][2]. The drug exhibits protein binding of approximately 55% [1].
Vancomycin does not undergo metabolic transformation and is primarily excreted unchanged [1]. The elimination half-life shows a biphasic pattern, with a relatively quick initial phase and a terminal half-life of 4 to 6 hours in healthy adults with normal renal function [1]. Intravenous vancomycin injection is primarily eliminated by glomerular filtration in the kidney, with 75% excreted via urine [1]. Vancomycin clearance ranges from 0.71 to 1.31 mL/min/kg in adults with normal renal function [1].
Tissue penetration varies considerably by site. Vancomycin penetrates epithelial lining fluid at approximately 50% of plasma levels [2]. The ratio of lung tissue to serum concentration is about 0.25 [2]. Limited cerebrospinal fluid penetration may be enhanced by the presence of inflamed meninges [2]. Penetration into soft tissue may be impaired in patients with diabetes [2]. These pharmacokinetic limitations have clinical implications for mrsa treatment vancomycin efficacy in specific infection sites.

The Rise of Vancomycin Resistant Staphylococcus Aureus
In the mid-1990s, reports indicated that the susceptibility of S. aureus to vancomycin was changing [3]. The emergence of vancomycin resistant staphylococcus aureus represents one of the most concerning developments in antimicrobial resistance. Clinical isolates with reduced susceptibility fall into distinct categories based on minimum inhibitory concentration thresholds and resistance mechanisms.
VISA: Vancomycin-Intermediate S. aureus (MIC 4-8 μg/ml)
Vancomycin-intermediate S. aureus strains exhibit MIC values of 4-8 μg/ml by reference broth microdilution [3][1]. These isolates are associated with hospitalization, persistent infection, and prolongation or failure of vancomycin therapy [1]. The resistance mechanism in VISA strains is not transferable to susceptible strains and is usually associated with vancomycin exposure [3]. Accordingly, the likelihood of transmission to contacts is low, and case investigations for VISA cases are not routinely recommended unless there is suspicion that transmission has occurred [3].
VISA strains generally evolve from heterogeneous vancomycin-intermediate S. aureus (hVISA), defined as an S. aureus strain with a vancomycin MIC within the susceptible range (≤2 μg/ml) determined by conventional methods, while a cell subpopulation exists in the vancomycin-intermediate range (≥4 μg/ml) [1]. The full VISA phenotype leads to common phenotypic changes, including excess peptidoglycan production, thickened cell wall, reduced autolytic activity, reduced biofilm formation, and loss of fitness [1]. Vancomycin becomes trapped in the thickened cell wall and titrated to below lethal concentration [1].
Clinical treatment failures have occurred with vancomycin in infections with isolates showing MIC >4 μg/ml, based on the apparent heterogeneity of strains and subpopulations with higher MICs [3]. The prolonged usage of vancomycin can lead to changes in cell wall patterning or reduced expression of penicillin-binding proteins [4].
VRSA: Complete Resistance via vanA Gene Cluster Transfer
In contrast, vancomycin-resistant S. aureus strains demonstrate MIC values >16 μg/ml [3][1]. VRSA strains have properties that allow them to transfer resistance to susceptible strains or other organisms; therefore, contact investigations and follow-up for VRSA cases are recommended [3]. In 2002, the first VRSA strain was recovered in Michigan, USA [1][5]. In the same year, the second VRSA strain was isolated in Pennsylvania, USA [1].
All identified isolates of S. aureus with reduced susceptibility to vancomycin have been methicillin-resistant S. aureus [3]. Transfer of the vanA gene cluster from Enterococcus species to S. aureus was demonstrated in vitro and in an in vivo model 15 years prior to the first clinical case [5]. VRSA strains carry transposon Tn1546, acquired from vancomycin-resistant Enterococcus faecalis [4][4].
Global Distribution: 52 VRSA Cases Documented Worldwide
Since 2002, a total of 52 VRSA strains carrying van genes have been reported [1][2]. The distribution includes 14 isolated in USA, 16 in India, 11 in Iran, 9 in Pakistan, 1 in Brazil, and 1 in Portugal [1][2]. Among the documented VRSA strains, vanA was present in 69% of cases tested by PCR [4]. The prevalence of vanA, vanB, and vanC1 positive strains were 71%, 26%, and 4% among VRSA isolates [6].
vanA-Mediated Resistance: D-Ala-D-Lac Modification Mechanism
Although 11 van gene clusters have been discovered to confer vancomycin resistance, only the vanA gene cluster is responsible for the isolated VRSA strains [1]. Five proteins encoded by the vanA gene cluster are essential for vancomycin resistance: VanS, VanR, VanH, VanA and VanX [1][2]. VanS and VanR form a two-component system regulating the cluster genes in the presence of vancomycin [2]. VanH, VanA, and VanX modify precursor molecules from D-Ala-D-Ala to the resistant form, D-Ala-D-Lac [2].
The modified d-Ala-d-Lac leads to an almost 1000-fold decrease in affinity with vancomycin [1][2]. The action target of vancomycin is the terminal d-Ala-D-Ala moieties of the precursor lipid II, with which vancomycin forms hydrogen bond interactions and prevents subsequent transglycosylation and transpeptidation [1]. However, modification to d-Ala-D-Lac greatly reduces vancomycin’s affinity, leading to loss of bactericidal effect on strains with modified peptidoglycan precursors [2]. The vanA gene cluster resides within transposon Tn1546 [2].
Clinical Limitations Driving the Shift From Vancomycin 
Clinical challenges extend beyond resistance patterns, affecting daily prescribing decisions across multiple infection types. The therapeutic space between efficacy and toxicity has contracted substantially over recent decades, forcing clinicians to reconsider vancomycin for mrsa in numerous clinical scenarios.
Narrow Therapeutic Index and Toxicity Concerns
Target trough levels have climbed from 5-10 mg/L in the 1980s to 15-20 mg/L in 2009 guidelines as bacteria evolved increased resistance [3]. This elevation creates a precarious balance. Troughs below 10 mg/L may promote resistant strain emergence, yet concentrations above 15 mg/L carry nephrotoxicity risks [3]. Nephrotoxicity occurred in 29.6% of patients with trough concentrations exceeding 15 mg/L compared to 8.9% with lower levels [7]. Multivariate analysis revealed vancomycin trough concentrations above 15 mg/L associated with a 3-fold increased risk of nephrotoxicity [7]. In another cohort, vancomycin-induced nephrotoxicity affected 31.7% of patients, with 25.2% experiencing mortality during hospitalization [3]. Failure to achieve recommended therapeutic levels between 15-20 mg/L occurred in 16.8% of patients, while 47.5% had excessive levels exceeding 20 mg/L [3].
Acute Kidney Injury Risk in Surgical Prophylaxis
Combination prophylaxis with vancomycin plus beta-lactams demonstrates particular concern in orthopedic procedures. Patients receiving dual antibiotics developed acute kidney injury at rates of 13% versus 8% with cefazolin alone in primary hip and knee arthroplasty [1]. The dual-antibiotic group experienced higher severity, with 3% developing Grade II and III acute kidney injury compared to 0% in the cefazolin-only group [1]. Across broader surgical populations, combination prophylaxis produced acute kidney injury in 23.8% of cases versus 13.9% receiving beta-lactam alone [1]. The number needed to harm for severe AKI following cardiac surgery reached 167 [1]. Specifically, the NNH for any postoperative AKI was 22 in cardiac surgery patients, 76 following orthopedic procedures, and 25 after vascular surgical procedures [1].
Therapeutic Drug Monitoring Challenges: AUC/MIC Ratios
Trough-only monitoring fails to accurately reflect total drug exposure. A multinational observational study found target drug levels achieved in only half of patients using conventional strategies among critically ill populations [3]. The 2020 revised guidelines recommend AUC/MIC monitoring over trough-based approaches, targeting an AUC between 400-600 mg*hour/L [8]. Trough monitoring associated with higher nephrotoxicity rates in spite of widespread implementation [8]. AUC-guided dosing demonstrated reduced nephrotoxicity, with hospitalized patients using trough-only dosing experiencing 8% AKI incidence compared to 0-2% with AUC dosing [3]. Calculating AUC requires additional resources, specialized software, and further training compared to simple trough measurements.
Poor Tissue Penetration in Pneumonia and Endocarditis
Vancomycin concentrations in epithelial lining fluid ranged from 5 to 25% of simultaneous plasma levels, while whole homogenized lung tissue concentrations reached 24-41% [9]. Penetration into skin tissue proves lower for patients with diabetes, with median concentrations of 0.1 mg/L compared to 0.3 mg/L in nondiabetic patients [5]. With uninflamed meninges, cerebral spinal fluid vancomycin concentrations ranged from 0 to approximately 4 mg/L [5]. These distribution limitations challenge linezolid vs vancomycin for mrsa pneumonia debates, as adequate lung tissue concentrations remain difficult to achieve.
Treatment-Emergent Resistance in Bacteremia Cases
Studies of MRSA and hVISA bacteremia revealed higher morbidity rates in patients infected with heterogeneous VISA strains [5]. Patients with vancomycin MIC of 1.5 mg/L experienced a 2.4-fold higher rate of treatment failure compared to those with MIC of 1 mg/L [5]. Vancomycin’s bactericidal rate remains slow compared to beta-lactams, with activity affected by bacterial inoculum [5]. Large bacterial burdens in stationary growth phase or anaerobic environments pose challenges to bactericidal speed and extent [5].
Vancomycin Alternatives: Comparing Efficacy and Safety Profiles
Multiple agents now challenge vancomycin’s historical dominance in MRSA infections, each offering distinct pharmacological advantages across different clinical scenarios.
Daptomycin: Once-Daily Dosing for MRSA Bacteremia
This cyclic lipopeptide disrupts bacterial cell membrane integrity through calcium-dependent insertion, causing rapid depolarization and cell death. A randomized trial established daptomycin 6 mg/kg daily as noninferior to standard therapy for S. aureus bacteremia and right-sided endocarditis [10]. However, higher doses between 8-12 mg/kg demonstrate superior outcomes in invasive infections. High-dose daptomycin produced 60% success rates compared to 35% with vancomycin in MRSA endocarditis, with more rapid bacteremia clearance (1 vs 5 days) [11]. Switching to daptomycin within 3 days of vancomycin initiation reduced 30-day mortality [12]. The once-daily administration eliminates therapeutic drug monitoring requirements, though pulmonary surfactant interaction renders daptomycin ineffective for bronchoalveolar pneumonia.
Linezolid vs Vancomycin for MRSA Pneumonia: Clinical Trial Data
The oxazolidinone linezolid demonstrates superior efficacy in MRSA pneumonia across multiple trials. A meta-analysis of seven randomized controlled trials with 1,239 patients found linezolid increased clinical cure rates (RR 0.81) and microbiological eradication rates (RR 0.71) compared to vancomycin [13]. The ZEPHyR study reported clinical cure in 57.6% of linezolid-treated patients versus 46.6% receiving vancomycin, with nephrotoxicity occurring in 8.4% versus 18.2% respectively [14]. Retrospective analysis showed linezolid reduced antibiotic switches (4.2% vs 17.6%) and all-cause mortality (23.2% vs 41.2%) [2]. Network meta-analysis ranked linezolid superior to vancomycin for MRSA-induced pneumonia and skin infections [6].
Ceftaroline: Beta-Lactam Activity Against MRSA
Ceftaroline fosamil represents the first widely approved beta-lactam retaining MRSA activity through high-affinity binding to PBP2a. This fifth-generation cephalosporin exhibits four-fold greater activity than vancomycin against MRSA isolates, with MIC90 values of 0.25 μg/ml [15]. Egyptian surveillance found 98.79% ceftaroline susceptibility among MRSA isolates [16]. FDA approval covers complicated skin infections and community-acquired pneumonia. Real-world studies reported 54-76% clinical success rates for MRSA pneumonia [17].
Combination Therapy Strategies: Vancomycin Plus Beta-Lactams
Adding beta-lactams to vancomycin or daptomycin produces mixed results. Meta-analysis of 806 patients demonstrated combination therapy reduced microbiological failure risk (OR 0.54) and persistent bacteremia (OR 0.48) without increasing nephrotoxicity [18]. Duration of bacteremia shortened by 1.06 days with combination regimens [19]. However, mortality benefits remain unproven, with 30-day mortality showing no difference between combination and monotherapy approaches.
Newer Agents: Dalbavancin and Oritavancin for Outpatient Treatment
Long-acting lipoglycopeptides enable weekly administration through extended half-lives. While FDA-approved only for skin infections, 64% of institutional use involves off-label indications including bacteremia and osteomyelitis [20]. Patients received median 7 days standard therapy before transitioning to long-acting agents. These antibiotics provide viable options for therapy completion when daily outpatient antimicrobial administration proves impractical.
When Vancomycin Remains the Appropriate Choice 
Despite emerging vancomycin alternatives, specific clinical scenarios and patient populations warrant its continued use when properly dosed and monitored. Recent guidelines emphasize precision in patient selection rather than blanket empiric coverage.
MRSA Treatment Vancomycin: Guidelines for Initial Empiric Therapy
The 2020 consensus guidelines recommend an AUC/MIC ratio of 400-600 mg*hour/L, assuming a broth microdilution MIC of 1 mg/L, for patients being treated for serious methicillin-resistant staphylococcus aureus infections [8]. These AUC targets should be achieved early in therapy, within 24-48 hours, given the importance of early and appropriate treatment [8]. Loading doses based on actual body weight are suggested for patients who are critically ill, requiring renal replacement therapy, or receiving continuous infusion therapy [8].
Empiric antimicrobial therapy with activity against MRSA consists of vancomycin or daptomycin prior to availability of culture and susceptibility data [21]. Notwithstanding the availability of alternatives, vancomycin for mrsa remains appropriate as initial empiric therapy in suspected invasive infections. Once susceptibility results confirm methicillin-sensitive S. aureus, treatment should be de-escalated to a beta-lactam agent such as nafcillin, oxacillin, or cefazolin [21]. Observational studies demonstrated nafcillin superior to vancomycin for preventing relapse in MSSA bacteremia, with failure rates of 4% versus 20% [21].
Caution applies when extrapolating vancomycin dosing data to mild noninvasive infections, as almost all available pharmacokinetic and toxicodynamic data derive from patients treated for serious MRSA infections, with the majority from complicated bloodstream infections [8]. Logistic regression identified initiation of vancomycin after a positive culture result as a predictor of treatment failure (OR 4.41) [22], underscoring the need for optimized empiric dosing before any modification of monitoring practices [22].
Risk Stratification: MRSA Colonization History
Patients with prior MRSA colonization or infection demonstrate 9-10 times greater odds of MRSA surgical site infection [4]. Nasal colonization with MRSA has a negative predictive value exceeding 94% for lower respiratory tract infection [23]. Polymerase chain reaction testing of nasal swabs achieves 98% NPV for MRSA colonization and can yield results within 2 hours [23]. Implementation of MRSA PCR testing reduces the duration of empirical MRSA-targeted therapy by approximately 2 days without worsening clinical course and lowers acute kidney injury incidence [23].
MRSA screening should be avoided in patients with recent nasal decolonization before screening, MRSA infection within 30 days before admission, or structural lung disease such as cystic fibrosis or bronchiectasis where colonization occurs more frequently in the lower respiratory tract [23].
Hip and Knee Replacement Surgeries: Prophylaxis Considerations
Hip or knee replacement surgery carries 3-4 times greater odds of MRSA surgical site infection [4]. The 2022 SHEA/IDSA/APIC guidance recommends reserving vancomycin prophylaxis to patients who are MRSA colonized [4]. In the event that routine MRSA colonization surveillance is unavailable, hip and knee replacement procedures represent an exception where vancomycin prophylaxis warrants consideration [4]. Healthcare-associated characteristics, including previous hospitalization or hemodialysis, were not associated with MRSA surgical site infection [4].
De-escalation Strategies Based on Culture Results
Approximately 98% of positive blood cultures for Staphylococcus aureus occur within 48 hours [23], creating a practical window for therapy assessment. About 43% of patients in education-based protocols avoided vancomycin initiation based on negative active surveillance cultures without additional negative outcomes [5]. Early vancomycin discontinuation based on negative surveillance and quantitative cultures proves reasonable in clinically suspected infections [5]. In like manner, stable patients in settings with low-to-moderate proportions of MRSA bacteremia (20-40%) can avoid empiric vancomycin therapy through screening protocols [23].
2026 Recommendations: Optimizing MRSA Treatment Selection
Treatment optimization in 2026 requires integration of rapid diagnostics, institutional protocols, and individualized patient assessment to balance efficacy against toxicity risks.
Diagnostic-Driven Therapy: Rapid MRSA PCR Testing
Nasal PCR assays demonstrate negative predictive values between 96.5% and 98.7% for MRSA pneumonia [3], enabling safe de-escalation of empiric vancomycin for mrsa within 24-48 hours. Implementation reduces anti-MRSA therapy duration by approximately 2 days without compromising clinical outcomes [3]. Pharmacist-driven protocols achieved vancomycin discontinuation within 24 hours of negative results in 88.9% of patients [24]. Cost analysis revealed savings of $40-108 per patient through earlier antibiotic cessation [3]. The median time from PCR order to result averages 4.4 hours [24].
Antimicrobial Stewardship Program Integration
Stewardship interventions decreased vancomycin utilization from 119 to 74 days of therapy per 1,000 patient days, a 37.8% reduction [25]. Accordingly, acute kidney injury incidence declined by over 50% during the same period [25]. Programs should elevate antimicrobial stewardship from supplementary to essential practice [26]. Education addressing proper empiric use and de-escalation strategies based on culture susceptibility remains fundamental [27].
Patient-Specific Factors: Renal Function and Infection Site
Selection of empirical therapy depends on infection type, local resistance patterns, drug availability, adverse-effect profile, and individual patient characteristics [27]. Patients with impaired renal function require dose adjustment based on creatinine clearance and therapeutic drug monitoring [1]. Target AUC/MIC ratios of 400-600 mg*hour/L optimize outcomes while minimizing nephrotoxicity [27]. Alternatives including linezolid require no renal adjustment, whereas daptomycin necessitates extended dosing intervals [1].
OPAT Considerations: Transitioning to Outpatient Alternatives
Daptomycin demonstrates superior performance in outpatient settings, with 94.6% success rates compared to 86.3% for inpatient therapy [28]. Once-daily dosing, reduced monitoring requirements, and lower adverse event rates make daptomycin preferable to vancomycin for longer-term outpatient treatment [28]. Long-acting lipoglycopeptides enable weekly administration when daily infusions prove impractical.

Conclusion 

Vancomycin retains clinical utility despite emerging alternatives and resistance patterns. Clinicians must weigh nephrotoxicity risks against therapeutic benefits. Rapid diagnostics enable precise patient selection, reducing unnecessary exposure. Linezolid demonstrates superior outcomes in pneumonia, while daptomycin excels in bacteremia. AUC-guided dosing optimizes safety margins. MRSA colonization screening identifies appropriate candidates for empiric coverage. Treatment selection demands integration of infection site, renal function, and institutional resistance data. De-escalation protocols preserve vancomycin’s effectiveness while minimizing toxicity. The 52 documented VRSA cases underscore antimicrobial stewardship urgency. Evidence-based algorithms balance traditional approaches with newer agents, ensuring optimal outcomes across diverse clinical scenarios. Personalized therapy replaces blanket empiric coverage as the cornerstone of modern MRSA management.
Frequently Asked Questions: 
FAQs
Q1. Is vancomycin still commonly prescribed for MRSA infections in 2026? Vancomycin continues to be used for MRSA infections, particularly as initial empiric therapy for serious invasive infections before culture results are available. However, clinicians now emphasize precise patient selection through risk stratification and rapid diagnostic testing rather than blanket empiric coverage. Once susceptibility results confirm the infection type, treatment is often de-escalated to more targeted alternatives based on the specific infection site and patient characteristics.
Q2. What are the recommended vancomycin blood levels for treating different MRSA infections? For most MRSA infections, the target AUC/MIC ratio is 400-600 mg*hour/L, which typically corresponds to trough levels of 10-15 mg/L. However, for more serious infections such as MRSA pneumonia, osteomyelitis, endocarditis, and bacteremia, higher trough levels of 15-20 mg/L are targeted to improve drug penetration to infection sites. Current guidelines recommend AUC-guided dosing over trough-only monitoring to optimize efficacy while minimizing toxicity risks.
Q3. What are the main side effects and organ risks associated with vancomycin therapy? Vancomycin primarily affects the kidneys, with nephrotoxicity occurring in approximately 30% of patients when trough concentrations exceed 15 mg/L. The risk increases significantly when vancomycin is combined with other antibiotics, particularly in surgical prophylaxis settings. Acute kidney injury rates can reach 24% with combination therapy compared to 14% with single-agent treatment. The drug can also cause hearing damage, and its narrow therapeutic index requires careful monitoring to balance effectiveness against toxicity.
Q4. Why are clinicians increasingly choosing alternatives to vancomycin for MRSA treatment? Several factors drive the shift away from vancomycin: rising resistance patterns (52 VRSA cases documented worldwide), poor tissue penetration in pneumonia and soft tissue infections, high nephrotoxicity rates, complex therapeutic drug monitoring requirements, and slower bactericidal activity compared to alternatives. Newer agents like linezolid demonstrate superior outcomes in MRSA pneumonia, while daptomycin shows better results in bacteremia with once-daily dosing and no monitoring requirements.
Q5. When is vancomycin still considered the most appropriate choice for MRSA treatment? Vancomycin remains appropriate for initial empiric therapy in suspected serious MRSA infections before culture results are available, particularly in patients with documented MRSA colonization history or those undergoing hip and knee replacement surgeries in facilities without routine MRSA screening. It’s also suitable when alternatives are contraindicated due to drug interactions, allergies, or specific infection characteristics. However, treatment should be reassessed within 24-48 hours based on culture results and patient response.
References: 
[2] – https://pubmed.ncbi.nlm.nih.gov/30671324/
[3] – https://pmc.ncbi.nlm.nih.gov/articles/PMC11420041/
[4] – https://pmc.ncbi.nlm.nih.gov/articles/PMC10897724/
[5] – https://journals.lww.com/ccmjournal/fulltext/2013/12001/1096__
early_vancomycin_de_escalation_in_patients.1050.aspx
[6] – https://pmc.ncbi.nlm.nih.gov/articles/PMC11428633/
[7] – https://pmc.ncbi.nlm.nih.gov/articles/PMC3232787/
[8] – https://www.idsociety.org/practice-guideline/vancomycin/
[9] – https://pubmed.ncbi.nlm.nih.gov/20055750/
[10] – https://www.nejm.org/doi/full/10.1056/NEJMoa053783
[11] – https://pmc.ncbi.nlm.nih.gov/articles/PMC8573721/
[12] – https://www.contagionlive.com/view/daptomycin-as-frontline-therapy-for-mrsa-bacteremia-has-the-time-come-
[13] – https://pubmed.ncbi.nlm.nih.gov/33401013/
[14] – https://pubmed.ncbi.nlm.nih.gov/24238896/
[15] – https://pmc.ncbi.nlm.nih.gov/articles/PMC3140339/
[16] – https://onlinelibrary.wiley.com/doi/10.1155/ijm/4558662
[17] – https://publications.ersnet.org/content/errev/32/170/230117
[18] – https://www.sciencedirect.com/science/article/pii/S2213716520302575
[19] – https://pubmed.ncbi.nlm.nih.gov/33728980/
[20] – https://pmc.ncbi.nlm.nih.gov/articles/PMC9751981/
[21] – https://www.uptodate.com/contents/clinical-approach-to-staphylococcus-aureus-bacteremia-in-adults
[22] – https://pmc.ncbi.nlm.nih.gov/articles/PMC8463016/
[23] – https://pmc.ncbi.nlm.nih.gov/articles/PMC10644157/
[24] – https://pmc.ncbi.nlm.nih.gov/articles/PMC8326869/
[25] – https://pmc.ncbi.nlm.nih.gov/articles/PMC6255640/
[26] – https://shea-online.org/updated-guidance-shows-how-hospitals-should-protect-patients-from-resistant-infections/
[27] – https://www.ncbi.nlm.nih.gov/books/NBK482221/
[28] – https://nhia.org/a-review-of-daptomycin-vs-vancomycin-for-susceptible-infections-is-one-superior-for-outpatient-parenteral-antibiotic-treatment-opat/
Video Section
Check out our extensive video library (see channel for our latest videos)
Recent Articles


