From NAFLD to MASLD Does the New Nomenclature Actually Change Care

Abstract
The transition from non alcoholic fatty liver disease to metabolic dysfunction associated steatotic liver disease represents a significant conceptual evolution in hepatology rather than a purely semantic change. Endorsed by major international liver societies in 2023, this updated terminology seeks to more accurately reflect the underlying pathophysiology of the disease while addressing longstanding concerns regarding the limitations and stigma associated with the previous nomenclature. By removing the emphasis on the absence of alcohol use and instead highlighting the central role of metabolic dysfunction, the term MASLD aligns more closely with current scientific understanding of disease mechanisms and risk factors.
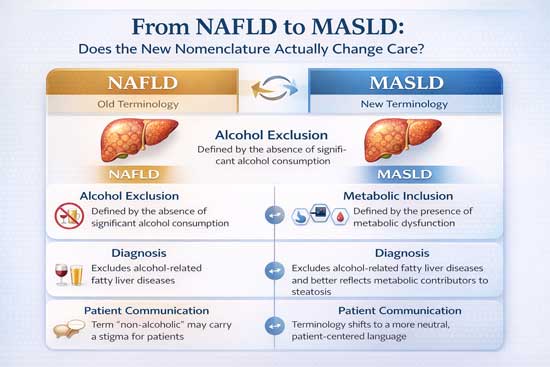
Non alcoholic fatty liver disease has historically been defined by the presence of hepatic steatosis in individuals without significant alcohol consumption or other secondary causes of liver fat accumulation. While this definition has been widely used, it has been criticized for being exclusionary and insufficiently descriptive of the metabolic drivers that underlie disease development. In contrast, MASLD introduces a more inclusive and pathophysiology based framework, requiring evidence of hepatic steatosis in the presence of at least one cardiometabolic risk factor, such as obesity, type 2 diabetes, dyslipidemia, or hypertension. This shift reflects the recognition that steatotic liver disease exists along a spectrum of metabolic dysfunction and is closely linked to systemic metabolic health.
This review examines whether the adoption of MASLD terminology translates into meaningful changes in clinical practice, patient engagement, and overall disease management. From a diagnostic perspective, the revised criteria may facilitate earlier identification of at risk individuals by emphasizing metabolic risk profiling rather than relying primarily on exclusion of alcohol intake. This approach has the potential to improve case finding in primary care and endocrinology settings, where metabolic comorbidities are frequently managed. In addition, the new nomenclature may enhance interdisciplinary collaboration by reinforcing the connection between liver disease and broader metabolic conditions.
Patient communication represents another important domain in which this transition may have clinical relevance. The term non alcoholic has often been associated with confusion or unintended stigma, particularly in populations where alcohol use carries cultural or social implications. By focusing on metabolic dysfunction, MASLD may improve patient understanding of disease etiology, promote acceptance of diagnosis, and encourage engagement with lifestyle and pharmacologic interventions targeting metabolic health.
Despite these conceptual advantages, the translation of nomenclature changes into improved clinical outcomes remains uncertain. Current therapeutic strategies for steatotic liver disease continue to rely heavily on lifestyle modification, including weight loss, dietary changes, and physical activity, alongside management of associated metabolic conditions. While several pharmacologic agents are under investigation, no universally approved disease specific therapy has yet demonstrated consistent benefit across all stages of disease. As such, the renaming alone does not directly alter the available treatment landscape.
Implementation of the MASLD framework also presents practical challenges. Clinicians must adapt to updated diagnostic criteria, integrate new terminology into clinical documentation, and ensure consistency across electronic health records, coding systems, and research protocols. There may also be transitional confusion among healthcare providers, patients, and researchers as both terms coexist in the literature and clinical practice during the adoption phase. Furthermore, epidemiological data collected under the NAFLD definition may not be directly comparable to studies using MASLD criteria, complicating longitudinal analyses and public health surveillance.
From a research perspective, the shift to MASLD has important implications for study design and patient selection. By incorporating metabolic risk factors into the definition, future clinical trials may enroll more homogeneous populations that better reflect disease biology. This could enhance the ability to detect treatment effects and improve the development of targeted therapies. At the same time, researchers must carefully consider how changes in diagnostic thresholds influence prevalence estimates and outcome measures.
In conclusion, the transition from NAFLD to MASLD represents a meaningful step toward aligning disease nomenclature with contemporary understanding of metabolic liver disease. It offers potential benefits in terms of conceptual clarity, patient communication, and integration of care across specialties. However, its impact on clinical outcomes will depend on how effectively this new framework is implemented and whether it catalyzes advances in risk stratification, therapeutic development, and multidisciplinary management. Continued evaluation is needed to determine whether this shift ultimately translates into improved patient care and disease burden reduction.
Introduction
The liver plays a central role in systemic metabolic regulation and is uniquely vulnerable to the consequences of metabolic dysfunction. Over the past several decades, clinicians have increasingly encountered a condition affecting nearly one quarter of the global population, historically termed non alcohol related fatty liver disease. This disorder is defined by the presence of hepatic steatosis in individuals without significant alcohol intake and encompasses a spectrum ranging from simple steatosis to steatohepatitis, progressive fibrosis, cirrhosis, and hepatocellular carcinoma. Despite its high prevalence and clinical significance, the condition has long posed diagnostic and therapeutic challenges due to its heterogeneous pathophysiology, variable clinical course, and strong association with extrahepatic metabolic disease.
In June 2023, an international consensus led by major hepatology societies introduced a revised nomenclature, replacing non alcohol related fatty liver disease with metabolic dysfunction associated steatotic liver disease. This change reflects a deliberate shift from a diagnosis of exclusion toward one grounded in positive, pathophysiologically relevant criteria. Rather than defining the condition by the absence of alcohol use, the new terminology emphasizes the presence of metabolic risk factors such as obesity, type 2 diabetes, dyslipidemia, and insulin resistance. This reconceptualization aligns more closely with current understanding of disease mechanisms and aims to improve clinical clarity, patient communication, and research consistency.
The transition to metabolic dysfunction associated steatotic liver disease occurs in the context of a rapidly expanding global burden of metabolic syndrome. Rising rates of obesity, type 2 diabetes, and cardiovascular disease have positioned liver disease as a key component of systemic metabolic dysfunction rather than an isolated hepatic condition. This shift has important implications for clinical practice, as it encourages a more integrated, multidisciplinary approach to patient care. The liver is increasingly recognized as both a target and a contributor to metabolic disease, with hepatic steatosis serving as a marker of broader cardiometabolic risk.
One of the most important practical implications of this nomenclature change lies in diagnostic criteria. The updated framework introduces a more inclusive and clinically actionable approach by requiring evidence of hepatic steatosis in the presence of at least one cardiometabolic risk factor. This approach facilitates earlier identification of at risk individuals and reduces reliance on excluding alcohol consumption as a primary diagnostic step. It also acknowledges that metabolic dysfunction and alcohol use may coexist, allowing for more nuanced classification of patients who would previously have been excluded or misclassified under earlier definitions.
From a therapeutic perspective, the redefinition reinforces the central role of metabolic risk modification in disease management. Lifestyle interventions remain the cornerstone of treatment, including weight reduction, dietary modification, and increased physical activity. However, the emphasis on metabolic dysfunction highlights the importance of addressing comorbid conditions such as diabetes, hypertension, and dyslipidemia as integral components of liver disease management. Pharmacologic strategies are also evolving, with several agents targeting metabolic pathways under investigation for their potential to improve hepatic outcomes.
The nomenclature change also carries implications for patient engagement and communication. The previous terminology, which emphasized the absence of alcohol use, often led to confusion and unintended stigma. By shifting the focus to metabolic health, the new terminology provides a clearer and more constructive framework for discussing disease etiology and management with patients. This may improve adherence to lifestyle and medical interventions by fostering a better understanding of the condition as part of a broader metabolic process.
In research and healthcare policy, the adoption of metabolic dysfunction associated steatotic liver disease is expected to enhance standardization in study design, patient stratification, and outcome measurement. A more precise definition may facilitate the development of targeted therapies and improve the comparability of clinical trial data. In addition, recognizing the condition as part of the metabolic disease spectrum may influence screening recommendations, resource allocation, and public health strategies aimed at reducing the burden of cardiometabolic disease.
Despite these advantages, challenges remain in implementing this new framework across diverse clinical settings. Variability in access to diagnostic tools, differences in healthcare infrastructure, and the need for clinician education may affect the pace and consistency of adoption. Furthermore, long term data evaluating whether this nomenclature shift translates into improved clinical outcomes are still emerging.
In summary, the transition from non alcohol related fatty liver disease to metabolic dysfunction associated steatotic liver disease represents more than a semantic change. It reflects a broader evolution in understanding liver disease as an integral component of systemic metabolic dysfunction. While the full clinical impact of this shift will become clearer over time, the new framework offers a more accurate, inclusive, and patient centered approach that has the potential to improve diagnosis, management, and long term outcomes in this increasingly prevalent condition.
Historical Context and Evolution of Nomenclature
The journey from NAFLD to MASLD reflects decades of evolving understanding about metabolic liver disease. The term “non-alcoholic fatty liver disease” emerged in the 1980s when researchers recognized a pattern of liver pathology similar to alcoholic liver disease but occurring in patients without substantial alcohol consumption. This negative definition – describing what the disease was not rather than what it was – created the foundation for decades of clinical practice.
Early research focused primarily on excluding alcohol as a causative factor. Patients underwent detailed questioning about drinking habits, and arbitrary cutoffs emerged for what constituted “excessive” alcohol consumption. The 20 grams per day threshold for women and 30 grams per day for men became standard, though these limits lacked strong scientific justification and varied across different guidelines.
As metabolic syndrome gained recognition in the 1990s and 2000s, researchers began connecting fatty liver disease with diabetes, obesity, dyslipidemia, and hypertension. The association became so strong that some experts began viewing fatty liver as the hepatic manifestation of metabolic syndrome. This understanding gradually shifted focus from alcohol exclusion toward metabolic inclusion.
The push for nomenclature change gained momentum as clinicians recognized several problems with the NAFLD terminology. Patients often reacted negatively to discussions about alcohol consumption, feeling accused or stigmatized even when they consumed minimal amounts. The negative definition also failed to capture the positive metabolic associations that drive disease pathogenesis.
International efforts to address these concerns culminated in extensive deliberations involving the American Association for the Study of Liver Diseases, the European Association for the Study of the Liver, and the Asian Pacific Association for the Study of the Liver. These organizations worked together to develop new terminology that would better reflect current understanding while maintaining clinical utility.
Understanding MASLD: Diagnostic Criteria and Clinical Features
The transition to MASLD brings with it refined diagnostic criteria that emphasize metabolic dysfunction rather than alcohol exclusion. The new definition requires the presence of hepatic steatosis along with at least one cardiometabolic risk factor. These risk factors include a body mass index of 25 kg/m² or higher in adults, diabetes mellitus, evidence of metabolic dysfunction, or other indicators of metabolic syndrome.
This positive diagnostic approach represents a fundamental shift in clinical thinking. Rather than spending considerable time establishing alcohol consumption patterns, physicians can focus on identifying metabolic risk factors that drive disease pathogenesis. The change may streamline diagnostic workflows while improving patient-physician communication.
The cardiometabolic criteria for MASLD diagnosis encompass several well-established risk factors. Overweight or obesity, defined by BMI thresholds, serves as the most common criterion. However, the guidelines acknowledge ethnic variations in BMI cutoffs, recognizing that Asian populations may develop metabolic dysfunction at lower BMI levels than previously appreciated.
Diabetes mellitus, whether type 1 or type 2, automatically qualifies patients for MASLD diagnosis when hepatic steatosis is present. This connection reflects the strong bidirectional relationship between liver fat accumulation and insulin resistance. Patients with diabetes face increased risks for progressive liver fibrosis and related complications.
Additional metabolic dysfunction criteria include elevated blood pressure, dyslipidemia, and evidence of insulin resistance in non-diabetic individuals. The guidelines also recognize that metabolic dysfunction can manifest in lean individuals, challenging previous assumptions about the universal association with obesity.
Clinical Presentation and Disease Spectrum
MASLD encompasses a broad spectrum of liver pathology, from simple steatosis to advanced fibrosis and cirrhosis. The majority of patients present with simple fatty infiltration of the liver, which may remain stable for years without progression to more severe disease. However, a subset develops metabolic dysfunction-associated steatohepatitis (MASH), previously known as non-alcoholic steatohepatitis (NASH).
MASH represents the inflammatory and potentially progressive form of MASLD. Patients with MASH face increased risks for liver fibrosis, cirrhosis, hepatocellular carcinoma, and liver-related mortality. The transition from MASLD to MASH involves complex interactions between metabolic stress, inflammatory pathways, and genetic predisposition.
Most patients with MASLD remain asymptomatic during early disease stages. When symptoms do occur, they are often non-specific and may include fatigue, right upper quadrant discomfort, or malaise. These vague presentations can delay diagnosis and contribute to disease progression in some individuals.
Physical examination findings in MASLD patients typically relate to underlying metabolic conditions rather than liver disease itself. Hepatomegaly may be present but is not universal. Signs of advanced liver disease, such as spider angiomata, palmar erythema, or ascites, appear only in patients who have progressed to cirrhosis.
Laboratory abnormalities in MASLD commonly include elevated aminotransferase levels, though these may be mild or intermittent. Some patients maintain normal liver enzymes despite having biopsy-proven steatohepatitis. This discordance between biochemical markers and histological findings challenges traditional approaches to disease monitoring.
Diagnostic Approaches and Clinical Assessment
The diagnostic evaluation of suspected MASLD has evolved alongside the nomenclature change, though many core principles remain unchanged. Initial assessment typically begins with a clinical history focusing on metabolic risk factors rather than detailed alcohol consumption patterns. This shift in emphasis may improve diagnostic efficiency while reducing patient discomfort during clinical encounters.
Imaging studies play a central role in the diagnosis and management of MASLD. Ultrasound remains the most commonly used initial imaging modality due to its availability and cost-effectiveness. However, ultrasound has limitations in detecting mild steatosis and cannot reliably distinguish between simple steatosis and steatohepatitis.
Advanced imaging techniques offer improved accuracy in detecting steatosis and assessing fibrosis. Magnetic resonance imaging-based methods, including MRI-PDFF (proton density fat fraction) and magnetic resonance elastography, provide quantitative assessments of liver fat content and stiffness. These tools are becoming more widely available but remain expensive for routine screening.
Transient elastography has gained widespread adoption for non-invasive fibrosis assessment in MASLD patients. The controlled attenuation parameter provides simultaneous evaluation of steatosis severity, while liver stiffness measurements estimate fibrosis stage. This technology enables serial monitoring of disease progression without repeated liver biopsies.
Serum biomarkers for MASLD continue to evolve, with several promising candidates under investigation. The FibroTest, Enhanced Liver Fibrosis panel, and NAFLD fibrosis score remain clinically available options for fibrosis risk stratification. However, none of these markers can reliably distinguish between simple steatosis and steatohepatitis without additional clinical context.
Liver biopsy remains the gold standard for MASLD diagnosis and staging, particularly when treatment decisions depend on histological findings. The procedure remains necessary for research studies and clinical trials requiring definitive disease characterization. However, sampling variability and procedural risks limit its routine clinical application.
Treatment Approaches: Has the Name Change Influenced Therapy?
The transition from NAFLD to MASLD has not yet led to revolutionary changes in treatment approaches, though it may influence how physicians and patients approach therapeutic interventions. The foundation of MASLD management continues to rest on lifestyle modifications targeting weight loss and metabolic improvement. However, the new nomenclature may enhance patient motivation and treatment adherence by providing a clearer understanding of disease mechanisms.
Weight loss remains the most effective intervention for MASLD patients across the disease spectrum. Studies consistently demonstrate that weight reduction of 5-10% can improve hepatic steatosis, while more substantial weight loss may reverse steatohepatitis and reduce fibrosis. The challenge lies in achieving and maintaining meaningful weight loss in patient populations already struggling with metabolic dysfunction.
Dietary interventions for MASLD have become more sophisticated as researchers better understand the relationship between specific nutrients and liver metabolism. The Mediterranean diet has emerged as a preferred approach, offering anti-inflammatory properties alongside weight management benefits. Low-carbohydrate diets also show promise for reducing hepatic steatosis, though long-term adherence remains challenging.
Exercise interventions provide benefits for MASLD patients, independent of weight-loss effects. Both aerobic exercise and resistance training can improve insulin sensitivity and reduce liver fat content. The optimal exercise prescription continues to be refined, but current evidence supports regular moderate-intensity activities over sporadic high-intensity efforts.
Pharmacological interventions for MASLD remain limited, though several promising agents are under investigation. Vitamin E supplementation has shown efficacy in non-diabetic patients with steatohepatitis, though concerns about long-term safety limit its widespread adoption. Pioglitazone, a thiazolidinedione diabetes medication, can improve liver histology but carries risks of weight gain and bone loss.
Newer therapeutic approaches target specific pathways involved in MASLD pathogenesis. GLP-1 receptor agonists, originally developed for diabetes management, show promise for treating liver disease through effects on weight loss, insulin sensitivity, and potentially direct hepatic actions. These medications may become cornerstone therapies as evidence continues to accumulate.
Table 1: Comparison of NAFLD vs MASLD Diagnostic Criteria
| Aspect | NAFLD | MASLD |
| Primary Focus | Alcohol exclusion | Metabolic dysfunction inclusion |
| Alcohol Threshold | <20g/day (women), <30g/day (men) | Not specifically defined |
| Required Criteria | Hepatic steatosis + alcohol exclusion | Hepatic steatosis + ≥1 cardiometabolic risk factor |
| BMI Consideration | Supportive but not required | ≥25 kg/m² (≥23 kg/m² for Asians) |
| Diabetes | Supportive evidence | Automatic qualification |
| Metabolic Syndrome | Associated condition | Core diagnostic feature |
| Clinical Emphasis | What disease is not | What disease is |
| Patient Communication | Often defensive discussions | More collaborative approach |
Impact on Clinical Practice Patterns
The nomenclature change from NAFLD to MASLD has begun influencing clinical practice patterns, though the full impact may take years to materialize. Early observations suggest that physicians are spending less time documenting alcohol consumption patterns and more time assessing metabolic risk factors. This shift in focus aligns better with evidence-based understanding of disease pathogenesis.
Patient education has become more straightforward with the new terminology. Many physicians report that patients respond more positively to discussions about “metabolic liver disease” compared to previous conversations about “non-alcoholic” conditions. The removal of alcohol-related terminology appears to reduce defensive reactions and improve therapeutic relationships.
Referral patterns between primary care physicians and hepatologists may be evolving as the metabolic focus becomes more prominent. Some primary care providers feel more comfortable managing MASLD patients when the emphasis lies on familiar metabolic conditions rather than liver-specific pathology. This comfort level could improve early intervention and prevent disease progression.
However, the implementation of new diagnostic criteria has created some confusion in clinical practice. Electronic health record systems, billing codes, and clinical protocols require updates to reflect the nomenclature change. These administrative adjustments take time and resources, potentially creating temporary inefficiencies in healthcare delivery.
The transition period has also highlighted gaps in medical education regarding metabolic liver disease. Many practicing physicians received training when NAFLD was primarily viewed as a benign condition. The MASLD framework emphasizes the progressive potential and systemic implications of metabolic liver disease, underscoring the need for ongoing education efforts.
Research and Clinical Trial Implications
The shift to MASLD nomenclature has immediate implications for clinical research and drug development efforts. Ongoing clinical trials that began using NAFLD terminology must address how findings translate to the new diagnostic framework. Regulatory agencies are working to establish guidance for this transition period.
Patient recruitment for clinical trials may be affected by the nomenclature change. Some researchers report improved enrollment rates when studies are described using MASLD terminology rather than NAFLD language. Patients appear more willing to participate in research about metabolic liver disease compared to studies focused on alcohol-related exclusions.
The new diagnostic criteria may influence trial design and endpoint selection. Studies can now focus more directly on metabolic parameters and cardiometabolic outcomes rather than alcohol-related confounders. This shift could lead to more clinically relevant research questions and outcomes.
Biomarker development efforts may benefit from the clearer pathophysiological focus of MASLD. Researchers can concentrate on metabolic pathways and inflammatory markers without the conceptual confusion created by negative definitions. This clarity may accelerate the development of non-invasive diagnostic tools.
International research collaboration may improve as global hepatology societies align around common terminology. Previous discrepancies in diagnostic criteria and nomenclature created challenges for meta-analyses and multicenter studies. The MASLD framework provides a foundation for more consistent research approaches.
Patient Perspectives and Communication
An interesting anecdote emerged during one of the first international conferences following the nomenclature change. A hepatologist presenting research findings accidentally referred to “NAFLD” throughout his entire presentation, only to be gently corrected by an audience member who noted that even experts were struggling with the transition. The presenter’s response – that changing decades of habit was like “teaching an old dog new tricks” – generated laughter but highlighted the very real challenges of implementing new terminology across the medical community.
Patient perspectives on the nomenclature change have been largely positive based on early feedback and informal surveys. Many individuals express relief that discussions about their liver condition no longer begin with questions about alcohol consumption. This shift appears to reduce stigma and defensive reactions that previously complicated clinical encounters.
The metabolic framing of MASLD helps patients better understand connections between their liver condition and other health problems. Many report that the new terminology makes more sense given their diabetes, obesity, or cardiovascular risk factors. This improved understanding may translate into better treatment adherence and lifestyle modifications.
However, some patients express confusion about whether their underlying condition has changed or only the name. Healthcare providers report spending additional time during visits to explain that MASLD and NAFLD represent the same disease process, using updated terminology. This educational burden may temporarily increase consultation times.
Family members and caregivers have also responded positively to the nomenclature change. The metabolic focus helps relatives understand that MASLD represents a systemic condition requiring comprehensive lifestyle approaches rather than simply a liver problem. This understanding can improve family support for dietary and exercise interventions.
Healthcare System and Policy Considerations
The transition to MASLD nomenclature has implications for healthcare systems and policy development that extend beyond individual patient care. Insurance coverage decisions, quality metrics, and population health initiatives must all adapt to the new terminology and diagnostic criteria.
Electronic health record systems across healthcare institutions are implementing ICD-11 codes to reflect the new nomenclature. This transition requires notable technical resources and staff training to ensure accurate documentation and billing. Some systems are maintaining dual coding during the transition period to avoid disruption.
Quality improvement initiatives focused on metabolic liver disease may benefit from the clearer pathophysiological framework provided by MASLD terminology. Healthcare systems can develop metrics that better capture the relationship between metabolic syndrome management and liver outcomes. This integration may improve care coordination across specialties.
Population health screening programs face decisions about whether and how to incorporate MASLD assessments. The metabolic focus of the new criteria may facilitate integration with existing diabetes and obesity screening efforts. However, resource allocation and cost-effectiveness analyses are still needed to guide implementation.
Professional society guidelines are being updated to reflect the nomenclature change, but this process requires careful consideration of evidence quality and clinical applicability. The challenge lies in distinguishing between changes that reflect improved understanding versus those that simply represent rebranding of existing concepts.
Challenges and Limitations of the Nomenclature Change
Despite the potential benefits of transitioning from NAFLD to MASLD, several challenges and limitations warrant careful consideration. The implementation process has revealed gaps between conceptual improvements and practical clinical benefits that may take years to address.
One of the primary challenges involves the transition period itself, during which both terminologies coexist in clinical practice and research literature. This dual nomenclature creates confusion for healthcare providers, patients, and researchers who must navigate between old and new systems. Medical literature searches become more complex when both terms are in active use.
The new diagnostic criteria, while conceptually clearer, may not substantially change the patient population identified for treatment. Studies suggest that the vast majority of patients previously diagnosed with NAFLD also meet MASLD criteria. This overlap raises questions about whether the nomenclature change provides meaningful clinical distinctions or primarily represents semantic improvement.
Some critics argue that the focus on metabolic dysfunction may inadvertently exclude patients who develop fatty liver disease through other mechanisms. Genetic variants, medications, and environmental factors can contribute to hepatic steatosis independent of classic metabolic syndrome components. The MASLD criteria may not adequately capture this pathophysiological diversity.
Training and education efforts required for the nomenclature change represent substantial resource investments for healthcare systems. Continuing medical education programs, clinical protocols, and patient educational materials all require updates. These costs may be difficult to justify if clinical outcomes do not demonstrably improve.
The international adoption of MASLD terminology is not universal, creating potential inconsistencies in global research and clinical practice. Some regions may continue to use NAFLD terminology for regulatory or practical reasons, complicating international collaboration and data-sharing efforts.
Comparative Analysis: MASLD vs Related Liver Conditions
Understanding MASLD requires distinguishing it from other liver conditions that may present with similar clinical features. The new nomenclature provides opportunities to clarify these distinctions while highlighting unique pathophysiological mechanisms.
Alcoholic liver disease shares many histological features with MASLD but results from different underlying mechanisms. While both conditions can progress to cirrhosis and hepatocellular carcinoma, the metabolic focus of MASLD distinguishes it from alcohol-induced liver injury. However, patients with metabolic dysfunction who also consume alcohol present diagnostic challenges that require careful clinical judgment.
Drug-induced liver injury can mimic MASLD, particularly when caused by medications that affect metabolic pathways. Certain diabetes medications, immunosuppressants, and other drugs can cause hepatic steatosis or steatohepatitis. The challenge lies in distinguishing medication effects from underlying metabolic liver disease.
Genetic forms of fatty liver disease, such as those caused by PNPLA3 or TM6SF2 variants, may not fit neatly into the MASLD framework. These patients can develop severe liver disease without typical metabolic risk factors, highlighting the limitations of purely metabolic definitions.
Viral hepatitis, particularly hepatitis C, can coexist with metabolic liver disease and complicate diagnostic and treatment approaches. The interaction between viral infection and metabolic dysfunction may accelerate liver disease progression, requiring integrated management strategies.
Wilson disease, hemochromatosis, and other genetic liver conditions can present with fatty infiltration that mimics MASLD. Careful clinical evaluation and appropriate testing remain necessary to exclude these conditions, particularly in younger patients or those without typical metabolic risk factors.
Future Directions and Research Opportunities
The transition to MASLD nomenclature opens new avenues for research and clinical development that could transform our understanding and treatment of metabolic liver disease. These opportunities extend beyond terminology to encompass fundamental questions about disease mechanisms and therapeutic approaches.
Precision medicine approaches to MASLD may benefit from the clearer pathophysiological framework provided by the new nomenclature. Researchers can focus on identifying metabolic subtypes that predict treatment responses or disease progression. Genetic, metabolomic, and proteomic studies may reveal distinct disease phenotypes within the MASLD spectrum.
Artificial intelligence and machine learning applications for MASLD diagnosis and management represent rapidly evolving research areas. These technologies could integrate clinical data, imaging findings, and laboratory results to predict disease outcomes or optimize treatment selection. The metabolic focus of MASLD provides clearer targets for algorithmic development.
Novel therapeutic approaches targeting specific metabolic pathways continue to emerge from laboratory research. Understanding MASLD as a metabolic disease may accelerate the development of combination therapies that address multiple pathogenic mechanisms simultaneously. These approaches could improve treatment efficacy beyond what single-agent therapies have achieved.
The relationship between MASLD and cardiovascular disease deserves continued investigation given shared metabolic underpinnings. Research examining integrated approaches to cardiometabolic risk may reveal opportunities for preventing both liver and cardiovascular complications through unified therapeutic strategies.
Population-based studies examining MASLD epidemiology and natural history could benefit from the new diagnostic criteria. These investigations may provide clearer insights into disease burden, progression rates, and risk factors when alcohol-related confounders are minimized. Such data could inform public health policies and screening recommendations.
Implementation Strategies for Clinical Practice
Successfully implementing the transition from NAFLD to MASLD requires strategic approaches that address the practical challenges facing healthcare providers and institutions. These strategies must balance the benefits of improved nomenclature with the realities of clinical workflow and resource constraints.
Education and training programs represent critical components of successful implementation. Healthcare institutions should develop continuing education initiatives that explain the rationale for nomenclature change while providing practical guidance for applying new diagnostic criteria. These programs should target not only physicians but also nurses, dietitians, and other healthcare team members.
Electronic health record modifications require careful planning and phased implementation to avoid disruption of clinical care. Institutions may benefit from maintaining dual documentation systems during transition periods while gradually shifting to MASLD-focused templates and order sets. Decision support tools can help providers apply new diagnostic criteria consistently.
Patient communication strategies should address common questions and concerns about the nomenclature change. Standardized educational materials can help providers explain that MASLD and NAFLD represent the same condition with updated terminology. These resources should emphasize the benefits of metabolic focus while maintaining continuity of care relationships.
Quality improvement initiatives can leverage the nomenclature change to enhance care coordination and outcomes measurement. Healthcare systems may develop new metrics that capture the integration of metabolic syndrome management with liver disease care. These efforts could identify opportunities for improving patient outcomes through systematic approaches.
Collaboration between primary care and specialty providers may improve as the metabolic focus of MASLD creates common ground for shared care models. Training programs should emphasize how primary care providers can effectively manage early-stage MASLD while identifying patients who require hepatology consultation for advanced disease.
Economic and Healthcare Resource Implications
The economic implications of transitioning from NAFLD to MASLD extend beyond immediate implementation costs to encompass potential long-term benefits from improved disease management and outcomes. Understanding these economic factors is essential for healthcare leaders making resource allocation decisions.
Direct costs associated with nomenclature implementation include electronic health record modifications, staff training, patient education materials, and updated clinical protocols. These one-time expenses may be substantial for large healthcare systems but are generally manageable when spread over appropriate implementation timelines.
Potential cost savings may emerge from more efficient diagnostic approaches that focus on metabolic risk factors rather than extensive alcohol history documentation. Reduced consultation times and improved patient satisfaction could translate into measurable economic benefits, though quantifying these effects requires systematic study.
The impact on pharmaceutical development costs deserves consideration as drug companies navigate regulatory requirements for both NAFLD and MASLD terminology. Clinical trial expenses may increase during transition periods when studies must address both nomenclatures, though long-term benefits may emerge from clearer regulatory pathways.
Healthcare utilization patterns may change as patients become more engaged with metabolic liver disease management compared to previous approaches focused on alcohol exclusion. Improved patient understanding could lead to better adherence to lifestyle interventions and preventive care, potentially reducing long-term healthcare costs.
Insurance coverage decisions for MASLD-related services will likely continue to align with previous NAFLD coverage policies, though some modifications may occur as the metabolic focus becomes more prominent. Coverage for lifestyle intervention programs and metabolic monitoring may expand as evidence supports their effectiveness for liver disease management.
Global Perspectives and Cultural Considerations
The international adoption of MASLD nomenclature highlights important cultural and regional variations in how metabolic liver disease is perceived and managed. These differences influence implementation strategies and may affect the global impact of the nomenclature change.
Cultural attitudes toward alcohol consumption vary widely across different regions and may influence how patients and providers respond to the shift away from alcohol-focused terminology. In cultures where alcohol use carries particular stigma, the MASLD nomenclature may be especially welcomed. Conversely, regions with more liberal attitudes toward alcohol may see less dramatic improvements in patient acceptance.
Healthcare system structures and resources affect the feasibility of implementing new diagnostic approaches across different countries. Regions with limited access to advanced imaging or laboratory testing may struggle to adopt metabolic-focused diagnostic criteria that require extensive cardiometabolic assessment.
The prevalence of metabolic risk factors varies globally, potentially affecting the clinical utility of MASLD criteria in different populations. Some regions face predominantly obesity-related metabolic dysfunction, while others deal with diabetes or other metabolic abnormalities. These variations may require tailored implementation approaches.
Language and translation issues pose practical challenges to the global adoption of MASLD terminology. Medical terminology translation often involves complex cultural and linguistic considerations that go beyond literal word conversion. Professional medical translation services may be required to ensure accurate communication across different languages.
Regional medical education systems and continuing education requirements influence how quickly healthcare providers can adopt new nomenclature and diagnostic approaches. Countries with mandatory continuing education programs may achieve faster implementation compared to regions where professional development is voluntary.
Long-term Outcomes and Monitoring
Evaluating the long-term impact of the NAFLD-to-MASLD transition requires establishing metrics and monitoring systems that capture meaningful changes in clinical practice and patient outcomes. These assessment approaches must balance scientific rigor with practical feasibility.
Patient outcome measures should focus on clinically meaningful endpoints rather than simply documentation or terminology changes. Metrics such as disease progression rates, treatment adherence, patient satisfaction, and quality of life indicators may provide more valuable insights than simple adoption rates of new terminology.
Healthcare provider behavior changes represent important intermediate outcomes that may predict longer-term clinical benefits. Surveys and observational studies examining how changes in nomenclature influence diagnostic approaches, treatment decisions, and patient communication could provide valuable implementation feedback.
Research productivity and quality in the field of metabolic liver disease may improve as investigators work within a clearer conceptual framework. Metrics such as publication rates, research funding success, and clinical trial enrollment could indicate whether the nomenclature change facilitates scientific progress.
Population health indicators related to metabolic liver disease awareness and management may show improvements as the MASLD framework gains acceptance. Public health surveillance systems could track screening rates, early detection, and prevention efforts to assess broader societal impacts.
International collaboration and standardization efforts in metabolic liver disease research and clinical care may benefit from unified terminology. Measures of cross-border research cooperation, guideline harmonization, and clinical protocol standardization could indicate progress toward global consensus.

Conclusion
The transition from NAFLD to MASLD represents more than a cosmetic change in medical terminology. This nomenclature shift reflects decades of accumulated evidence about the metabolic underpinnings of fatty liver disease and attempts to align clinical language with pathophysiological understanding. However, the ultimate success of this transition will be measured not by adoption rates of new terminology but by tangible improvements in patient care and outcomes.
Early evidence suggests that the MASLD nomenclature offers several advantages over previous approaches. Patients appear more receptive to discussions about metabolic liver disease compared to conversations focused on alcohol exclusion. Healthcare providers report improved efficiency in diagnostic evaluations when the emphasis shifts to metabolic risk-factor assessment rather than to detailed alcohol-history documentation. These changes may enhance the therapeutic relationship and improve treatment adherence.
Yet the nomenclature change alone cannot address fundamental challenges in managing metabolic liver disease. Effective treatments remain limited, with lifestyle modification continuing to serve as the primary therapeutic intervention. The transition to MASLD does not eliminate the difficulty of achieving and maintaining weight loss in populations already struggling with metabolic dysfunction. Neither does it resolve questions about optimal screening strategies, treatment timing, or long-term monitoring approaches.
The implementation process has revealed both opportunities and challenges that deserve ongoing attention. Healthcare systems must invest resources in training, education, and infrastructure modifications to support the transition. These investments require justification by demonstrated improvements in clinical outcomes rather than by mere conceptual advantages.
Research opportunities emerging from the MASLD framework may ultimately provide the greatest return on investment from this nomenclature change. Clearer pathophysiological focus could accelerate therapeutic development, improve biomarker discovery, and enhance our understanding of disease progression mechanisms. However, realizing these benefits requires sustained commitment to high-quality research that builds upon the conceptual foundation provided by updated terminology.
The global nature of metabolic liver disease demands international cooperation and standardization efforts that extend beyond nomenclature to encompass clinical protocols, research methodologies, and public health approaches. The MASLD framework provides a foundation for this cooperation, but success depends on continued collaboration among healthcare providers, researchers, and policy makers worldwide.
The transition from NAFLD to MASLD brings both promise and challenges that require careful consideration by healthcare providers and healthcare systems. Understanding these implications is essential for the successful implementation and optimization of patient care.
The nomenclature change improves conceptual clarity by focusing on metabolic dysfunction rather than alcohol exclusion. This shift aligns clinical terminology with current understanding of disease pathophysiology and may enhance patient-provider communication. Healthcare providers should expect more efficient diagnostic evaluations and improved patient receptiveness to treatment discussions.
Implementation requires strategic planning and resource allocation to address training needs, system modifications, and patient education requirements. Healthcare institutions should develop phased approaches that minimize disruption while ensuring consistent application of new diagnostic criteria. Quality improvement initiatives can leverage the transition to enhance care coordination and outcomes measurement.
Research opportunities emerging from the MASLD framework may drive future advances in understanding and treating metabolic liver disease. The clearer pathophysiological focus could accelerate therapeutic development and improve clinical trial design. However, realizing these benefits requires sustained investment in high-quality research efforts.
Long-term success of the nomenclature change depends on demonstrable improvements in clinical outcomes rather than simply adoption of new terminology. Monitoring efforts should focus on meaningful endpoints such as disease progression rates, treatment adherence, and patient satisfaction. Continued evaluation will guide refinements to implementation strategies and clinical approaches.
Healthcare providers should view the MASLD transition as an opportunity to improve patient care while recognizing that fundamental challenges in metabolic liver disease management persist. Success requires commitment to evidence-based practice, ongoing education, and collaborative approaches that address the complex metabolic underpinnings of this increasingly common condition.
Frequently Asked Questions
Q: Do I need to change how I diagnose fatty liver disease with the new MASLD criteria?
A: The diagnostic approach shifts from excluding alcohol consumption to identifying metabolic risk factors. You still need to assess for hepatic steatosis, but the emphasis moves to documenting cardiometabolic conditions like diabetes, obesity, or metabolic syndrome rather than detailed alcohol history.
Q: Will insurance coverage change for patients previously diagnosed with NAFLD?
A: Insurance coverage should remain consistent during the transition period, as MASLD and NAFLD represent the same disease process with updated terminology. However, some insurers may eventually expand coverage for metabolic interventions as the metabolic focus becomes more prominent.
Q: How should I explain the name change to my patients?
A: Emphasize that their condition hasn’t changed, only the name and understanding of it. Explain that the new terminology better reflects that their liver condition is related to metabolic factors like diabetes or obesity rather than alcohol. This often reduces patient anxiety and defensive reactions.
Q: Are there new treatment options available with the MASLD nomenclature?
A: The treatment options remain largely the same, with lifestyle modification continuing as the primary intervention. However, the metabolic focus may lead to better integration with diabetes and obesity management, potentially improving overall care coordination.
Q: Do the laboratory tests or imaging studies change with MASLD?
A: The diagnostic tests remain the same, including blood work, imaging studies, and liver biopsy when indicated. However, you may spend more time assessing metabolic parameters and less time documenting alcohol consumption patterns.
Q: How quickly do I need to adopt the new terminology?
A: Implementation timelines vary by institution and region. Many healthcare systems are using a phased approach over several months to years. Focus on understanding the concepts rather than rushing to change all documentation immediately.
Q: Will medical students and residents need different training for MASLD?
A: Medical education is gradually incorporating the new nomenclature and metabolic focus. Current trainees should learn both terminologies during the transition period, with emphasis on understanding the metabolic pathophysiology that drives disease development.
Q: Does MASLD change when I should refer patients to hepatology?
A: Referral criteria remain similar, typically involving evidence of advanced fibrosis, elevated liver enzymes, or complications of liver disease. However, the metabolic focus may help primary care providers feel more comfortable managing early-stage disease independently.
References
Eslam, M., Newsome, P. N., Sarin, S. K., Anstee, Q. M., Targher, G., Romero-Gomez, M., … & George, J. (2020). A new definition for metabolic dysfunction-associated fatty liver disease: An international expert consensus statement. Journal of Hepatology, 73(1), 202-209.
Rinella, M. E., Lazarus, J. V., Ratziu, V., Francque, S. M., Sanyal, A. J., Kanwal, F., … & Newsome, P. N. (2023). A multisociety Delphi consensus statement on new fatty liver disease nomenclature. Journal of Hepatology, 79(6), 1542-1556.
Younossi, Z. M., Koenig, A. B., Abdelatif, D., Fazel, Y., Henry, L., & Wymer, M. (2016). Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology, 64(1), 73-84.
Powell, E. E., Wong, V. W. S., & Rinella, M. (2021). Non-alcoholic fatty liver disease. The Lancet, 397(10290), 2212-2224.
Chalasani, N., Younossi, Z., Lavine, J. E., Charlton, M., Cusi, K., Rinella, M., … & Sanyal, A. J. (2018). The diagnosis and management of nonalcoholic fatty liver disease: Practice guidance from the American Association for the Study of Liver Diseases. Hepatology, 67(1), 328-357.
Targher, G., Tilg, H., & Byrne, C. D. (2021). NAFLD and increased risk of cardiovascular disease: Clinical associations, pathophysiological mechanisms and pharmacological implications. Gut, 70(4), 844-855.
Romero-Gómez, M., Zelber-Sagi, S., & Trenell, M. (2017). Treatment of NAFLD with diet, physical activity and exercise. Journal of Hepatology, 67(4), 829-846.
Sanyal, A. J., Chalasani, N., Kowdley, K. V., McCullough, A., Diehl, A. M., Bass, N. M., … & NASH CRN. (2010). Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis. New England Journal of Medicine, 362(18), 1675-1685.
Wong, V. W. S., Adams, L. A., de Lédinghen, V., Wong, G. L. H., & Sookoian, S. (2018). Noninvasive biomarkers in NAFLD and NASH—current progress and future promise. Nature Reviews Gastroenterology & Hepatology, 15(8), 461-478.
Vilar-Gomez, E., Martinez-Perez, Y., Calzadilla-Bertot, L., Torres-Gonzalez, A., Gra-Oramas, B., Gonzalez-Fabian, L., … & Romero-Gomez, M. (2015). Weight loss through lifestyle modification significantly reduces features of nonalcoholic steatohepatitis. Gastroenterology, 149(2), 367-378.
Recent Articles


Integrative Perspectives on Cognition, Emotion, and Digital Behavior

Sleep-related:
Longevity/Nutrition & Diet:
Philosophical / Happiness:
Other:
Video Section 

