Understanding Subclinical Hypothyroidism: The Essential Guide to Borderline Thyroid Test Results

Introduction
Subclinical hypothyroidism is one of the most frequently encountered biochemical thyroid abnormalities in clinical practice, affecting approximately 5 percent of adults in the United States. Its prevalence increases substantially with age and is particularly common in women, reaching nearly 20 percent among women older than 60 years. The condition is defined by an elevated serum thyroid stimulating hormone concentration in the presence of normal circulating free thyroxine levels, reflecting early or mild thyroid dysfunction in which pituitary compensation maintains peripheral thyroid hormone concentrations within the reference range.
Although laboratory findings are central to diagnosis, the clinical significance of subclinical hypothyroidism remains an area of ongoing debate because biochemical abnormalities do not always correlate clearly with symptom burden, long term outcomes, or need for treatment. In many individuals, the condition is identified incidentally during routine laboratory testing, as symptoms may be mild, nonspecific, or entirely absent. When symptoms do occur, they often overlap with those of overt hypothyroidism and may include fatigue, reduced exercise tolerance, weight gain, cold intolerance, constipation, dry skin, cognitive slowing, depressive symptoms, and menstrual irregularities. However, these manifestations are frequently subtle and may also reflect aging, comorbid disease, or other endocrine and metabolic disorders, making clinical recognition particularly challenging.
The underlying causes of subclinical hypothyroidism are diverse, though autoimmune thyroid disease remains the most common etiology in iodine sufficient populations. Chronic autoimmune thyroiditis, particularly Hashimoto thyroiditis, leads to gradual thyroid follicular destruction and impaired hormone synthesis, often accompanied by detectable thyroid peroxidase antibodies or thyroglobulin antibodies. The presence of thyroid autoantibodies is clinically relevant because it increases the likelihood of progression from subclinical to overt hypothyroidism over time. Other causes include partial thyroid failure following thyroid surgery or radioiodine therapy, iodine deficiency or excess, certain medications such as lithium, amiodarone, and immune checkpoint inhibitors, as well as transient thyroid dysfunction during recovery from non thyroidal illness.
Interpretation of thyroid stimulating hormone levels requires careful clinical context because mild TSH elevation may not always represent persistent thyroid disease. Physiological variation, age related shifts in TSH reference ranges, circadian fluctuation, obesity, acute illness, and laboratory variability can all influence results. For this reason, confirmation with repeat testing after an appropriate interval is often recommended before establishing a diagnosis, particularly when TSH elevation is modest. Persistent elevation, especially when accompanied by positive thyroid autoantibodies, strengthens diagnostic confidence and informs long term risk assessment.
From a clinical management perspective, subclinical hypothyroidism presents important decision making challenges because treatment benefits are not uniform across all patient groups. Current evidence suggests that the degree of TSH elevation is a major determinant of therapeutic consideration. Patients with TSH concentrations above 10 mIU/L are more likely to benefit from levothyroxine therapy due to increased risk of progression to overt hypothyroidism, dyslipidemia, and possible cardiovascular effects. In contrast, management of mild TSH elevation, typically between the upper reference limit and 10 mIU/L, requires individualized assessment that considers age, symptoms, cardiovascular risk, pregnancy status, and antibody positivity.
The cardiovascular implications of subclinical hypothyroidism remain an important area of clinical interest. Several studies have linked persistent TSH elevation, particularly at higher levels, to adverse lipid profiles, endothelial dysfunction, and increased risk of coronary artery disease. However, evidence regarding whether levothyroxine treatment consistently improves hard cardiovascular outcomes remains mixed, especially in older adults. Similarly, while some patients report symptomatic improvement with therapy, randomized trials have shown variable benefit in quality of life, fatigue, and cognitive outcomes, particularly in elderly populations with mild biochemical abnormalities.
Special populations warrant particular attention. In pregnancy or in women planning conception, even mild thyroid dysfunction may have implications for maternal and fetal outcomes, including miscarriage risk, preterm birth, and neurodevelopmental considerations. Consequently, lower thresholds for treatment are often recommended in pregnant patients, especially when thyroid autoimmunity is present. Younger adults with symptoms, infertility, goiter, or progressive TSH elevation may also be more appropriate candidates for early intervention.
Treatment, when indicated, typically involves low dose levothyroxine initiated with careful biochemical monitoring. Dose selection should account for age, body weight, cardiovascular status, and severity of TSH elevation. In older adults and those with ischemic heart disease, cautious dose titration is particularly important to avoid overtreatment. Monitoring generally involves reassessment of thyroid stimulating hormone six to eight weeks after initiation or dose adjustment, followed by periodic long term follow up once biochemical stability is achieved.
Equally important is avoidance of unnecessary treatment. Overtreatment may lead to suppressed TSH levels, increasing the risk of atrial fibrillation, reduced bone mineral density, and iatrogenic thyrotoxicosis, especially in older patients. Therefore, treatment decisions should be guided not only by laboratory thresholds but also by a balanced understanding of potential benefits, risks, and patient priorities.
In summary, subclinical hypothyroidism represents a common but clinically nuanced endocrine disorder that requires thoughtful interpretation of biochemical findings within a broader clinical framework. Accurate diagnosis depends on confirming persistent thyroid stimulating hormone elevation, identifying underlying causes, and assessing progression risk. Management should be individualized, with treatment decisions informed by TSH level, symptom profile, autoimmune status, age, cardiovascular context, and reproductive considerations. As evidence continues to evolve, clinicians must apply current guidelines carefully to ensure that intervention is both evidence based and patient centered.
What is Subclinical Hypothyroidism? 
Defining Borderline Thyroid Function
The laboratory diagnosis of subclinical hypothyroidism requires elevated thyroid-stimulating hormone levels with normal concentrations of free thyroxine and triiodothyronine. This condition, also termed mild thyroid failure or borderline hypothyroidism, represents a biochemical state where the hypothalamic-pituitary axis detects subtle thyroid hormone deficiency despite peripheral hormone levels remaining within population reference ranges.
Healthcare providers diagnose this condition solely through thyroid blood tests. The hallmark finding combines TSH elevation above the upper limit of normal, typically around 4.5 mIU/L, while measured circulating thyroid hormones (free T4 and T3) fall within normal ranges. The individual range for peripheral thyroid hormones is narrower than the population reference laboratory range; a slight reduction within the normal range results in TSH elevation above normal.
Providers may categorize subclinical hypothyroidism by severity:
- Grade 1 (mild): TSH levels between 4.0 and 10.0 mIU/L
- Grade 2 (severe): TSH levels exceeding 10 mIU/L
Approximately 80% of patients with subclinical hypothyroidism have serum TSH less than 10 mIU/L. Most patients with serum TSH below 10 mU/L remain asymptomatic.
How Subclinical Hypothyroidism Differs from Overt Hypothyroidism
Overt hypothyroidism manifests with both elevated TSH levels and decreased thyroid hormone concentrations, whereas subclinical disease presents with abnormal TSH levels only while thyroid hormone levels remain normal. Overt hypothyroidism produces clear clinical manifestations with increased TSH and decreased T4 levels. In contrast, borderline hypothyroidism typically does not cause noticeable symptoms.
The distinction carries clinical relevance. Serum TSH has a log-linear relationship with circulating thyroid hormone levels; a 2-fold change in free thyroxine produces a 100-fold change in TSH[33]. This exponential relationship explains TSH’s sensitivity to minor thyroid hormone fluctuations. The hypothalamic-pituitary axis responds to small decreases in circulating thyroid hormone by elevating TSH above normal when free T4 has fallen only slightly and remains within reference ranges.
Individual patients maintain unique setpoints within the hypothalamic-pituitary axis. TSH levels exhibit wide variability across populations, but intra-individual variation remains minimal secondary to these personal setpoints. Most individuals demonstrate a log-linear TSH-T4 relationship. Be that as it may, some demonstrate nonlinear relationships explaining normal free T4 with TSH exceeding 10 mIU/L, while others may have low T4 levels.
TSH Levels and What They Mean
The upper limit of normal for TSH remains debated. The US Preventive Services Task Force defines subclinical hypothyroidism as TSH exceeding 4.50 mIU/L with normal free T4. Various authorities propose different cutoffs ranging from 2.5 to 5.0 mIU/L as the upper reference limit.
TSH levels fluctuate naturally throughout the day and respond to stress, sleep deprivation, recent infections, intense exercise, and fasting. Testing during or after illness can show temporary changes that normalize on repeat testing. For the purpose of confirming diagnosis, thyroid function should remain stable for several weeks to exclude transient changes from nonthyroidal illness, thyroiditis, or medications.
In patients with TSH below 10 mIU/L and normal free T4, thyroid blood work should be repeated in 3 to 6 months before therapy initiation, as almost half of these patients experience resolution of thyroid abnormalities. Indeed, subclinical hypothyroidism resolved spontaneously in 46% of older participants after 4 years in a prospective study involving 459 individuals aged 65 and older.
Age influences reference ranges. TSH distribution shifts toward higher concentrations with aging. After age 70, values up to 6.0 or even 7.0 mIU/L in the absence of antithyroid antibodies should not result in hypothyroidism diagnosis. In populations 85 years and older, 14% had TSH levels higher than 4.5 mUI/L, particularly among women.
Common Causes of Subclinical Hypothyroidism 
Subclinical hypothyroidism shares the same causative disorders as overt hypothyroidism, differing only in severity. Primary hypothyroidism accounts for 99% of cases, occurring when permanent loss or destruction of the thyroid develops. Central hypothyroidism represents the remainder, arising from pituitary gland or hypothalamus dysfunction.
Autoimmune Thyroid Disease (Hashimoto’s Thyroiditis)
Chronic autoimmune thyroiditis stands as the most common endogenous cause of hypothyroidism in older adults. Hashimoto’s thyroiditis accounts for 60-80% of subclinical hypothyroidism cases. This autoimmune disorder produces chronic inflammation of the thyroid gland through antibodies that damage thyroid tissue. Patients develop antibodies against thyroid peroxidase (TPO) and thyroglobulin (Tg), which attack healthy thyroid cells.
The inflammatory process gradually impairs the gland’s capacity to produce thyroid hormone. Hashimoto’s thyroiditis runs in families and occurs predominantly in women. Patients with positive thyroperoxidase antibodies face increased risk of developing drug-induced hypothyroidism, particularly when preexisting Hashimoto’s thyroiditis exists. More than 90% of patients with autoimmune thyroiditis exhibit anti-TPO positivity.
Previous Thyroid Treatments and Surgery
Thyroid failure commonly develops after treatment for Graves’ disease. Radioactive iodine treatment carries the highest risk, though hypothyroidism may eventually occur in 5 to 25 percent of patients treated with surgery or antithyroid drugs. Total thyroidectomy necessitates lifelong thyroid hormone replacement therapy. Following partial thyroidectomy, approximately 40% of patients require thyroid hormone replacement.
Radiation used to treat head and neck cancers affects thyroid function and leads to hypothyroidism. Surgery produces measurable hormonal changes; studies demonstrate that thyroid manipulation during surgery causes increased T4 and thyroglobulin levels intraoperatively, followed by postoperative decreases in TSH, T4, and T3 levels.
Medications That Affect Thyroid Function
Multiple pharmaceutical agents interfere with thyroid function at various levels. Drug-induced hypothyroidism occurs through several mechanisms:
- Iodine-containing medications: Amiodarone, containing 37.5% iodine by weight, causes thyroid dysfunction in 2 to 24% of patients. Hypothyroidism develops in 6 to 10 percent of patients receiving amiodarone. The drug’s long half-life (40-55 days) means dysfunction can occur up to one year after discontinuation.
- Lithium: Used for bipolar disorder management, lithium inhibits thyroid hormone release by increasing thyroidal iodine content and blocking hormone synthesis.
- Cancer immunotherapies: Interferon-alpha affects thyroid function in up to 10% of patients after three months of treatment. Tyrosine kinase inhibitors influence thyroid function in more than 50% of cases through thyroidal inflammation and upregulated hepatic metabolism. Checkpoint inhibitors like pembrolizumab produce thyroid changes in up to 14% of patients.
- Glucocorticoids: These medications suppress TSH secretion at the hypothalamic level. Dexamethasone doses as low as 0.5 mg can lower serum TSH levels.
- Other agents: Dopamine agonists like bromocriptine, somatostatin analogs, and certain antiepileptic medications including carbamazepine and valproic acid alter thyroid function.
Age-Related Changes in Thyroid Function
While aging itself produces minor effects on the thyroid gland, thyroid disorders become more common with advancing age. TSH levels increase progressively with age in iodine-sufficient populations, becoming apparent from the third decade of life. The National Health and Nutrition Examination Survey demonstrated that the 97.5 centile exceeds 7.0 mIU/L in individuals aged over 80 years.
Risk factors for subclinical hypothyroidism include older age, female sex, genetic susceptibility with family history of hypothyroidism, pregnancy with progression from postpartum thyroiditis, obesity, and Type 1 diabetes. Thyroid disease in close family members such as parents, siblings, or children provides an important diagnostic clue.
Recognizing the Symptoms of Subclinical Hypothyroidism 
Why Symptoms Are Often Mild or Absent
Subclinical hypothyroidism remains asymptomatic in most patients. In fact, approximately 70% of individuals with this condition experience no symptoms whatsoever. The mild elevation in TSH with preserved normal thyroid hormone levels explains this clinical silence. The body maintains adequate peripheral thyroid hormone concentrations despite early hypothalamic-pituitary axis activation, preventing the development of overt clinical manifestations.
When symptoms do occur, their presentation tends toward subtlety. The gradual nature of thyroid dysfunction progression means manifestations develop slowly, often over several years. Patients may barely notice early changes or attribute them to aging, stress, or lifestyle factors rather than thyroid pathology. This diagnostic ambiguity presents challenges for clinicians attempting to distinguish true thyroid-related symptoms from coincidental complaints.
Non-Specific Symptoms to Watch For
The symptoms of subclinical hypothyroidism lack specificity, occurring commonly in numerous other medical conditions and normal physiological states. Fatigue represents the most frequently reported complaint. Studies indicate that up to one-third of patients presenting with fatigue have thyroid disease, with women affected predominantly. Fatigue manifests as physical tiredness and lack of energy, distinct from weakness, and may persist despite adequate rest.
Clinical studies comparing symptomatic profiles provide insight into which manifestations correlate with elevated TSH. One investigation of 33 patients with subclinical hypothyroidism versus 20 euthyroid controls found dry skin, cold intolerance, and easy fatigability occurred significantly more frequently in those with raised TSH levels. These symptoms improved following thyroid hormone treatment. Similarly, another study of 69 female patients demonstrated that clinical indices based on symptoms and physical signs became more abnormal as TSH levels increased, even with normal T4 concentrations.
Common manifestations include:
- Fatigue and tiredness
- Unexplained weight gain or difficulty losing weight
- Cold intolerance and sensitivity to cold temperatures
- Dry, coarse skin and hair
- Hair loss and thinning
- Constipation
- Depression and low mood
- Decreased attention span and memory problems
- Muscle weakness, cramps, and aches
- Menorrhagia (heavy menstrual bleeding)
- Bradycardia and diastolic hypertension
- Hoarseness of voice
- Carpal tunnel syndrome
When Symptoms Actually Indicate Thyroid Problems
Determining whether symptoms actually stem from borderline hypothyroidism versus other causes poses substantial clinical difficulty. The vague, non-specific nature of hypothyroid symptoms means they overlap extensively with other common conditions. Fatigue or weight gain, for instance, more likely result from lifestyle factors or alternative medical conditions than thyroid dysfunction.
Nonetheless, some patients with subclinical hypothyroidism experience symptoms that appear disproportionate to their mild biochemical abnormalities. This discordance between laboratory findings and clinical presentation complicates treatment decisions. Residual symptoms can persist even after TSH normalization with levothyroxine therapy. Proposed explanations for persistent symptoms despite normalized thyroid profiles include accompanying comorbidities, incorrect symptom attribution, unrealistic patient expectations, tissue-level hypothyroidism with low T3 concentrations, altered T4 to T3 conversion, or impaired cellular thyroid hormone entry.
The frequency of autoimmune thyroid disease correlates with higher fatigue rates compared to post-thyroidectomy hypothyroidism. Patients with positive thyroperoxidase antibodies demonstrate increased depression risk independent of TSH elevation, suggesting a possible genetic basis for neuropsychiatric associations. Studies examining neuropsychiatric manifestations suggest increased rates of depression and panic disorder in subclinical hypothyroidism, along with poorer antidepressant treatment response.
Assessing hypothyroid symptoms influences whether thyroid replacement therapy becomes indicated. Providers must weigh symptom presence, severity, and impact on quality of life against the costs and potential adverse effects of treatment when making management decisions.
How Doctors Diagnose Borderline Hypothyroidism 
The Role of TSH Testing
Clinicians begin diagnostic testing with a thyroid-stimulating hormone test alone when thyroid dysfunction is suspected. The hypothalamic-pituitary-thyroid axis regulates hormone production, with TSH levels serving as the key indicator of thyroid function in primary hypothyroidism. Since TSH is regulated by thyroid hormones, TSH levels reliably predict thyroid hormone function.
This TSH-centered approach provides clinical efficiency. Unless clinical evidence suggests central thyroid dysfunction at the hypothalamus or pituitary level, which is rare, TSH serves as the principal test to assess primary thyroid gland dysfunction. In British Columbia, clinical laboratories first measure plasma TSH when ‘suspected hypothyroidism or hyperthyroidism’ is selected on standard outpatient requisitions; they proceed to measure free T4 only if TSH is abnormal.
TSH alone suffices to monitor therapy for patients taking levothyroxine monotherapy for primary hypothyroidism. Once TSH normalizes under treatment, annual repeat TSH measurements are appropriate unless clinical changes might impact levothyroxine pharmacokinetics, such as pregnancy, significant weight changes, gastrointestinal disorders, or supplements containing calcium and iron. Following dose adjustments, clinicians should wait at least 6 weeks before re-checking TSH, as TSH values change slowly.
Understanding Free T4 and T3 Measurements
When TSH exceeds normal limits, free T4 measurement distinguishes clinical hypothyroidism (low free T4) from subclinical hypothyroidism (normal free T4). If TSH is high and free T4 is low, the diagnosis is overt hypothyroidism. Conversely, if the second test shows high TSH but T4 and T3 fall within the standard range, the diagnosis is subclinical hypothyroidism.
Free T4 testing provides more accurate thyroid function assessment than total T4 because it is not affected by changes in binding proteins. An elevated TSH with normal free T4 may suggest subclinical hypothyroidism, whereas high TSH with low free T4 may indicate primary hypothyroidism. Routinely evaluating total T3, total T4, or free T3 levels is not indicated. The free T3 test is sometimes performed for suspected overactive thyroid gland.
Thyroid Antibody Tests
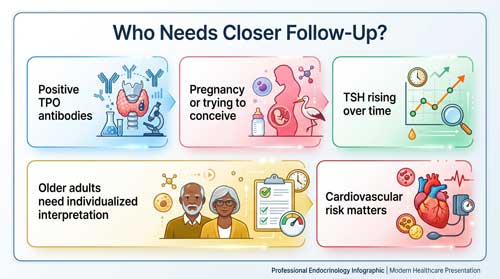
Thyroid peroxidase antibodies can help confirm Hashimoto’s thyroiditis as a cause of primary hypothyroidism. Testing for TPO antibodies does not help diagnose hypothyroidism; be that as it may, a positive test result suggests an autoimmune etiology. In patients with subclinical hypothyroidism, TPO antibodies should be checked. If positive for TPO antibodies, this indicates an autoimmune etiology and correlates with a 2-fold increased risk of progression to overt hypothyroidism.
Being positive for anti-TPO antibodies increases the risk of developing overt hypothyroidism in people with subclinical hypothyroidism, with progression rates of 4.3% per year versus 2.1% per year in antibody-negative individuals. Many clinical endocrinologists use the TPO antibody test as a diagnostic tool in deciding whether to treat a patient with subclinical hypothyroidism[244].
If performed and a person tests anti-TPO antibody positive, there is no reason to repeat anti-TPO antibody testing. The TPO antibody titers decrease with time and do not need repetition. This test seldom changes clinical management.
Confirming the Diagnosis Over Time
Multiple tests over 3 to 6 month intervals should be performed to confirm or rule out abnormal findings. For patients with initial abnormal TSH, repeat testing should occur after 2 to 3 months to confirm diagnosis, as 30% to 62% of elevated TSH values normalize spontaneously without intervention. In patients with TSH below 10 mIU/L and normal free T4, thyroid blood work should be repeated in 3 to 6 months before therapy initiation, as almost half of these patients experience resolution of thyroid abnormalities.
Non-thyroidal illness and some medications can cause TSH elevation; they need to be ruled out before establishing a diagnosis of subclinical hypothyroidism. Professional groups recommend repeating thyroid function tests if results fall above or below specified reference intervals for confirmation of persistent dysfunction in asymptomatic persons before making a diagnosis or considering treatment strategies, unless serum TSH exceeds 10.0 or falls below 0.1 mIU/L.
Among people older than 65 years, TSH levels in the subclinical hypothyroid range commonly normalize over time without treatment. In one study, 60% of patients with one abnormal TSH had normal TSH levels when retested at one year.
Treatment Options for Subclinical Hypothyroidism 
Treatment for subclinical hypothyroidism requires individualized decision-making based on TSH elevation degree, patient age, symptoms, cardiovascular risk factors, and personal preferences. International guidelines differ regarding treatment thresholds owing to conflicting evidence on long-term clinical benefits.
When Treatment Is Recommended
Levothyroxine treatment is generally recommended when TSH exceeds 10.0 mIU/L. This threshold reflects uniform agreement among endocrinologists, justified by high progression rates to overt hypothyroidism and demonstrated cardiovascular benefits. Patients with TSH higher than 8 mIU/L face substantial progression risk, with 70% developing TSH levels exceeding 10 mIU/L within 4 years.
For TSH levels between 4.5 and 10.0 mIU/L, treatment decisions become more nuanced. Current guidelines suggest considering treatment in:
- Adults under 65-70 years with persistent symptoms consistent with hypothyroidism
- Patients with underlying coronary artery disease or heart failure
- Individuals with concomitant dyslipidemia requiring improvement
- Positive thyroid peroxidase antibodies indicating autoimmune thyroiditis
- Women planning pregnancy or currently pregnant
Conversely, treatment is not recommended for adults 70 years and older with mild subclinical hypothyroidism unless TSH exceeds 10 mIU/L. Recent evidence suggests elderly patients may not derive mortality benefits from treatment.
The Watchful Waiting Approach
For most patients with TSH below 10 mIU/L, particularly those 65 years and older, watchful waiting represents the preferred strategy[313]. TSH levels normalize spontaneously in 30% to 62% of patients within months without intervention.
Active surveillance protocols recommend repeating thyroid function tests within 8 to 12 weeks because thyroid function normalizes in 6% to 35% of patients. If subclinical hypothyroidism persists, testing should occur every 6 months for the first 2 years, then annually thereafter.
Levothyroxine Therapy: What to Expect
When treatment is initiated, levothyroxine serves as the drug of choice. Weight-adjusted dosing of 1.5 µg/kg daily is recommended for patients without cardiac disease, whereas lower starting doses of 25 to 50 µg daily are appropriate for elderly patients or those with heart problems[312].
The medication must be taken on an empty stomach because foods and supplements interfere with absorption. Patients should wait 30 to 60 minutes before eating and at least 4 hours before taking calcium, iron, or antacids.
Monitoring and Follow-Up Care
TSH should be rechecked 2 to 3 months after starting levothyroxine, with dose adjustments targeting TSH in the lower half of the reference range (0.4 to 2.5 mIU/L). Once stable, annual monitoring suffices. Patients started on treatment primarily for symptoms should be reassessed 3 to 4 months after TSH normalization; treatment should cease if symptoms fail to improve.
Special Considerations and Risk Factors 
Certain populations require modified assessment and management strategies when subclinical hypothyroidism is identified.
Subclinical Hypothyroidism in Pregnancy
Treatment becomes indicated during pregnancy when TSH exceeds pregnancy-specific reference ranges, particularly in women with positive thyroid peroxidase antibodies. Subclinical hypothyroidism affects 4-8% of reproductive-age individuals, with treatment associated with lower pregnancy loss risk, especially when TSH ranges between 4.1 and 10 mU/L. Women diagnosed before 21 weeks’ gestation face more than fourfold increased rates of progression to overt hypothyroidism within 5 years postpartum. TPO antibody levels exceeding 50 IU/mL predict higher progression rates.
Considerations for Older Adults
TSH elevation occurs naturally with aging, with the 97.5th percentile exceeding 7.0 mIU/L in individuals over 80 years. Hypothyroidism prevalence reaches 23.78% in adults 65 years and older. Symptoms manifest atypically in elderly patients, often presenting as confusion, depression, falls, or heart failure rather than classic hypothyroid features. Treatment remains controversial in this population, as randomized trials have not demonstrated benefits for quality of life or symptom relief.
Cardiovascular Health and Heart Disease Risk
Subclinical hypothyroidism increases cardiovascular disease risk through multiple mechanisms. TSH levels exceeding 7 mIU/L correlate with elevated heart attack risk, while levels above 10 mIU/L associate with heart failure development. The condition produces endothelial dysfunction, increased systemic vascular resistance, and reduced nitric oxide availability. Cardiovascular disease mediates 14.3% of associations between subclinical hypothyroidism and all-cause mortality.
Impact on Cholesterol and Metabolism
Hypothyroidism elevates total cholesterol and LDL-cholesterol levels through decreased hepatic LDL receptor expression. Treatment with levothyroxine in subclinical hypothyroidism reduces total cholesterol by 12 mg/dL and LDL-cholesterol by 11 mg/dL. Patients with TSH above 10 mIU/L demonstrate particularly pronounced dyslipidemia.
Progression to Overt Hypothyroidism
Progression rates vary based on antibody status and TSH elevation. Annual progression occurs in 4.3% of patients with positive TPO antibodies versus 2.1% in antibody-negative individuals.

Conclusion 

Subclinical hypothyroidism requires nuanced clinical judgment rather than reflexive treatment. Practitioners should base management decisions on several factors:
- TSH elevation degree (above or below 10 mIU/L)
- Patient age and comorbidities
- Symptom presence and severity
- TPO antibody status
Treatment clearly benefits patients with TSH exceeding 10 mIU/L. Conversely, watchful waiting serves most individuals with milder elevations, particularly those over 65 years. Confirmation through repeat testing prevents unnecessary intervention, as up to 62% of cases resolve spontaneously. For the purpose of optimal care, physicians must individualize decisions, weighing progression risk against treatment burden. Recent evidence supports restraint in asymptomatic elderly patients while emphasizing intervention for younger adults with persistent elevation and cardiovascular risk factors.

FAQs 
Q1. What exactly does subclinical hypothyroidism mean? Subclinical hypothyroidism, also called borderline or mild hypothyroidism, is a condition where your TSH (thyroid-stimulating hormone) level is slightly elevated above the normal range but your thyroid hormones (T4 and T3) remain within normal limits. It represents an early stage of thyroid dysfunction where your body is working harder to maintain normal hormone levels.
Q2. Should I take levothyroxine if I have subclinical hypothyroidism? Treatment depends on several factors. Levothyroxine is generally recommended if your TSH exceeds 10 mIU/L. For TSH levels between 4.5 and 10 mIU/L, treatment may be considered if you’re under 65-70 years old with symptoms, have heart disease, are planning pregnancy, or have positive thyroid antibodies. Many cases resolve on their own without medication, especially in older adults.
Q3. Can borderline thyroid levels return to normal without treatment? Yes, thyroid levels frequently normalize on their own. Studies show that 30% to 62% of people with initially elevated TSH levels see their values return to normal within months without any intervention. This is why doctors typically recommend retesting after 2-3 months before starting treatment, as up to half of patients with mild elevation experience spontaneous resolution.
Q4. What lifestyle changes can help manage borderline hypothyroidism? Maintaining a healthy lifestyle supports thyroid function. Key steps include eating a nutrient-rich diet, getting adequate sleep, staying physically active, maintaining a healthy weight, managing stress through techniques like meditation or yoga, limiting alcohol consumption, and avoiding smoking. If you have diabetes, controlling blood sugar levels is also important.
Q5. How often should I get my thyroid levels checked if I have subclinical hypothyroidism? If you’re not on treatment, your doctor should retest your thyroid function within 8-12 weeks initially. If subclinical hypothyroidism persists, testing should occur every 6 months for the first 2 years, then annually. If you start levothyroxine, TSH should be rechecked 2-3 months after starting or adjusting doses, and once stable, annual monitoring is typically sufficient.
References: 
- American Academy of Family Physicians. (n.d.). Subclinical hypothyroidism. https://www.aafp.org/pubs/afp/issues/2015/0601/od1.html
- American Academy of Family Physicians. (n.d.). Subclinical hypothyroidism: Deciding when to treat. https://www.aafp.org/pubs/afp/issues/1998/0215/p776.html
- American Academy of Family Physicians. (n.d.). Hypothyroidism: Diagnosis and treatment. https://www.aafp.org/pubs/afp/issues/2021/0515/p605.html
- American Association of Clinical Endocrinology. (n.d.). Hypothyroid treatment. https://www.aace.com/patient-journey/thyroid/planning-and-treatment/hypothyroid-treatment
- American Thyroid Association. (n.d.). Management of hypothyroidism during pregnancy: When and how to treat?. https://www.thyroid.org/management-hypothyroidism-pregnancy/
- American Thyroid Association. (n.d.). Patient thyroid information. https://www.thyroid.org/patient-thyroid-information/ct-for-patients/march-2024/vol-17-issue-3-p-7-8/
- American Thyroid Association. (n.d.). Patient thyroid information. https://www.thyroid.org/patient-thyroid-information/ct-for-patients/march-2021/vol-14-issue-3-p-7-8/
- American Thyroid Association. (n.d.). Patient thyroid information. https://www.thyroid.org/patient-thyroid-information/ct-for-patients/may-2017/vol-10-issue-5-p-8/
- American Thyroid Association. (n.d.). Patient thyroid information. https://www.thyroid.org/patient-thyroid-information/ct-for-patients/april-2021/vol-14-issue-4-p-6-7/
- Biondi, B., & Cooper, D. S.. (2018). The clinical significance of subclinical thyroid dysfunction. https://www.mayoclinicproceedings.org/article/S0025-6196(11)62389-6/fulltext
- British Thyroid Foundation. (n.d.). Older patients and thyroid disease. https://www.btf-thyroid.org/older-patients-and-thyroid-disease
- Cleveland Clinic. (2025). Subclinical hypothyroidism: Symptoms & treatment. https://my.clevelandclinic.org/health/diseases/23544-subclinical-hypothyroidism
- Cleveland Clinic. (2019). Subclinical hypothyroidism: Not every patient requires treatment. https://www.ccjm.org/content/86/2/101
- Cleveland Clinic. (2025). Individualized management of subclinical hypothyroidism. https://consultqd.clevelandclinic.org/individualized-management-of-subclinical-hypothyroidism
- Cleveland Clinic Journal of Medicine. (2025). Subclinical hypothyroidism: Symptoms to look out for and when to treat. https://www.ccjm.org/content/92/4/221
- Consultant360. (n.d.). Subclinical hypothyroidism. https://www.consultant360.com/content/subclinical-hypothyroidism
- Donangelo, I., & Suh, S. Y.. (2017). Subclinical hypothyroidism in pregnancy: A systematic review and meta-analysis. https://www.ajog.org/article/S0002-9378(16)31409-0/fulltext
- DrOracle. (n.d.). When should a thyroid-stimulating hormone (TSH) test be repeated?. https://www.droracle.ai/articles/709897/when-should-a-thyroid-stimulating-hormone-tsh-test-be-repeated
- Fatourechi, V.. (2001). Subclinical thyroid disease. https://www.mayoclinicproceedings.org/article/S0025-6196(11)62389-6/fulltext
- Gaitonde, D. Y., Rowley, K. D., & Sweeney, L. B.. (2012). Hypothyroidism: An update. https://www.aafp.org/pubs/afp/issues/2012/0801/p244.html
- Georgia Diabetes and Endocrinology Task Force. (n.d.). Diagnosis and treatment of subclinical hypothyroidism. https://gdatf.org/bulletins/diagnosis-and-treatment-of-subclinical-hypothyroidism/
- Healthline. (n.d.). Subclinical hypothyroidism in pregnancy. https://www.healthline.com/health/hypothyroidism/subclinical-hypothyroidism-pregnancy
- Heymann, R., & Brent, G. A.. (2005). Rapid progression from subclinical to symptomatic overt hypothyroidism. https://pubmed.ncbi.nlm.nih.gov/15901527/
- Hindustan Times. (2026). Got borderline thyroid levels? Pathologist explains what it really means and when you truly need medication. https://www.hindustantimes.com/lifestyle/health/got-borderline-thyroid-levels-pathologist-explains-what-it-really-means-and-when-you-truly-need-medication-101768834923141.html
- Inoue, K., Tsujimoto, T., Saito, J., & Sugiyama, T.. (2020). Association of subclinical hypothyroidism and cardiovascular disease with mortality. https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2760440
- Javed, Z., Sathyapalan, T., & Razvi, S.. (2016). Levothyroxine treatment of mild subclinical hypothyroidism: A review of potential risks and benefits. https://pmc.ncbi.nlm.nih.gov/articles/PMC4740939/
- Levels. (n.d.). Guide to free T4. https://www.levels.com/blog/guide_to_free_t4
- Mayo Clinic. (n.d.). Hypothyroidism: Diagnosis and treatment. https://www.mayoclinic.org/diseases-conditions/hypothyroidism/diagnosis-treatment/drc-20350289
- Mayo Clinic Laboratories. (n.d.). T4 (thyroxine), free, serum. https://www.mayocliniclabs.com/test-catalog/overview/82041
- Medical News / News-Medical. (2024). Study finds fourfold increase in risk of overt hypothyroidism after diagnosis of gestational subclinical hypothyroidism. https://www.news-medical.net/news/20240802/Study-finds-fourfold-increase-in-risk-of-overt-hypothyroidism-after-diagnosis-of-gestational-subclinical-hypothyroidism.aspx
- MedlinePlus. (n.d.). Thyroid antibodies. https://medlineplus.gov/lab-tests/thyroid-antibodies/
- National Center for Biotechnology Information. (2023). Management of subclinical hypothyroidism: A focus on proven health effects in the 2023 Korean Thyroid Association guidelines. https://pmc.ncbi.nlm.nih.gov/articles/PMC10475969/
- National Center for Biotechnology Information. (2024). Progression of gestational subclinical hypothyroidism and hypothyroxinemia to overt hypothyroidism after pregnancy: Pooled analysis of data from two randomized controlled trials. https://pmc.ncbi.nlm.nih.gov/articles/PMC11563732/
- National Center for Biotechnology Information. (2023). Severity of fatigue and its relationship with TSH before and after levothyroxine replacement therapy in patients with primary hypothyroidism. https://pmc.ncbi.nlm.nih.gov/articles/PMC10045891/
- National Center for Biotechnology Information. (2023). Relationship between subclinical hypothyroidism and the risk of cardiovascular complications. https://pmc.ncbi.nlm.nih.gov/articles/PMC9922092/
- National Center for Biotechnology Information. (2015). Thyroid and the heart. https://pmc.ncbi.nlm.nih.gov/articles/PMC4318631/
- National Center for Biotechnology Information. (2018). Cardiovascular risk in patients with subclinical hypothyroidism. https://pmc.ncbi.nlm.nih.gov/articles/PMC5983087/
- National Center for Biotechnology Information. (2011). Effects of thyroid dysfunction on lipid profile. https://pmc.ncbi.nlm.nih.gov/articles/PMC3109527/
- NHS. (n.d.). Levothyroxine. https://www.nhs.uk/medicines/levothyroxine/
- Patel, Y., Joseph, N., & others. (2019). Subclinical hypothyroidism. https://www.ncbi.nlm.nih.gov/books/NBK536970/
- Pearce, E. N.. (2012). Update in lipid alterations in subclinical hypothyroidism. https://www.ncbi.nlm.nih.gov/books/NBK83492/
- Pharmaceutical Journal. (n.d.). It’s a murky business: The row over levothyroxine use in subclinical disease. https://pharmaceutical-journal.com/article/feature/its-a-murky-business-the-row-over-levothyroxine-use-in-subclinical-disease
- Puskarich, M.. (2024). Subclinical hypothyroidism. https://www.ncbi.nlm.nih.gov/books/NBK615101/
- Ruíz-Pacheco, M. G., Hernández, I., Hernández-Estrella, G., et al.. (2023). Severity of fatigue and its relationship with TSH before and after levothyroxine replacement therapy in patients with primary hypothyroidism. https://pmc.ncbi.nlm.nih.gov/articles/PMC10045891/
- StatPearls Publishing. (2023). Hypothyroidism. https://www.ncbi.nlm.nih.gov/books/NBK279005/
- Test Catalog. (n.d.). Thyroid peroxidase antibodies, serum. https://neurology.testcatalog.org/show/TPO
- Wiley / Frontiers. (2019). Subclinical hypothyroidism: To treat or not to treat, that is the question!. https://www.frontiersin.org/journals/endocrinology/articles/10.3389/
fendo.2019.00177/full - Wiley / Frontiers. (2020). Levothyroxine therapy in elderly patients with hypothyroidism. https://www.frontiersin.org/journals/endocrinology/articles/
10.3389/fendo.2020.591588/full - Wong, B., & others. (2016). Association of thyroid function with arterial stiffness and cardiovascular outcomes. https://www.ahajournals.org/doi/10.1161/JAHA.115.002225
- Wu, J., & others. (2023). Subclinical hypothyroidism and cardiovascular outcomes. https://pmc.ncbi.nlm.nih.gov/articles/PMC10069079/
Video Section
Check out our extensive video library (see channel for our latest videos)
Recent Articles


