Understanding VITT: What Vaccine Safety Experts Learned Beyond COVID-19

Key Takeaways
Vaccine induced thrombotic thrombocytopenia, commonly referred to as VITT, has become one of the most important examples of how rapid scientific collaboration can reshape clinical outcomes during a global public health emergency. First recognized during the early rollout of adenoviral vector based COVID 19 vaccines, VITT emerged as a rare but severe immune mediated complication characterized by thrombosis occurring in association with thrombocytopenia after vaccination. Initial reports described an unexpectedly high mortality rate, largely because the syndrome was unfamiliar, frequently misdiagnosed, and biologically distinct from conventional thrombotic disorders. However, within a remarkably short period, international collaboration among hematologists, immunologists, neurologists, and regulatory agencies transformed VITT from a highly fatal complication into a condition that can now be recognized early and managed effectively.
The earliest phase of the VITT experience demonstrated the critical value of rapid clinical pattern recognition. By early April 2021, the European Medicines Agency identified a safety signal involving unusual thrombotic events occurring after administration of adenoviral vector COVID 19 vaccines. Clinicians across multiple countries began reporting clusters of patients presenting with cerebral venous sinus thrombosis, splanchnic vein thrombosis, pulmonary embolism, and arterial thromboses accompanied by unexpectedly low platelet counts. Affected individuals were often younger adults without traditional thrombotic risk factors, and symptoms typically developed between 5 and 30 days after vaccination. This temporal association, combined with the unusual anatomical distribution of thrombosis and concurrent thrombocytopenia, raised concern for a previously unrecognized immune mediated syndrome.
VITT is now understood as an acquired prothrombotic disorder driven by antibodies directed against platelet factor 4, or PF4. These antibodies bind to PF4 and form immune complexes that activate platelets through Fc gamma receptor mediated signaling pathways, leading to massive thrombin generation and widespread thrombosis despite low circulating platelet counts. Mechanistically, this process resembles autoimmune heparin induced thrombocytopenia, although it occurs in the absence of prior heparin exposure. The identification of anti PF4 antibodies as the central pathogenic driver represented a major diagnostic breakthrough and immediately informed treatment strategies.
The discovery of this immunologic mechanism fundamentally changed patient outcomes. During the earliest reported cases, mortality approached 40 percent, particularly in patients with cerebral venous thrombosis and delayed diagnosis. Once clinicians recognized the syndrome and implemented targeted treatment protocols, mortality fell dramatically to approximately 4 to 6 percent. This near 90 percent reduction in mortality illustrates the impact of early diagnosis and protocol driven intervention in rare but high risk adverse events.
One of the most important therapeutic advances in VITT management has been the use of high dose intravenous immunoglobulin. Intravenous immunoglobulin administered at 1 gram per kilogram body weight interrupts pathogenic platelet activation by saturating Fc receptors and preventing further antibody mediated platelet stimulation. Clinical observations consistently demonstrate rapid hematologic improvement following treatment, with platelet counts often rising within 12 to 36 hours. This intervention has become a central component of first line therapy and is now widely regarded as the most critical early treatment in confirmed or strongly suspected cases.
Anticoagulation remains equally important, but the choice of anticoagulant is highly specific. Because VITT shares mechanistic similarities with heparin induced thrombocytopenia, heparin should be avoided whenever possible, as it may exacerbate platelet activation and worsen thrombosis. Instead, non heparin anticoagulants such as argatroban, fondaparinux, bivalirudin, or direct oral anticoagulants are recommended depending on the clinical setting and organ involvement. The simultaneous use of immunomodulation and non heparin anticoagulation has become the cornerstone of effective VITT treatment.
Diagnostic accuracy has also improved substantially through the development of focused clinical criteria. VITT should be suspected in any patient presenting within 5 to 30 days of adenoviral vector vaccination with thrombosis, thrombocytopenia, markedly elevated D dimer levels, and reduced fibrinogen concentrations. Laboratory confirmation typically involves enzyme linked immunosorbent assay testing for anti PF4 antibodies, which remains more reliable than rapid immunoassays in this setting. Imaging studies are guided by symptoms, with special attention to cerebral venous sinus thrombosis, abdominal venous thrombosis, and pulmonary vascular involvement.
Beyond immediate clinical care, the VITT experience has had profound implications for vaccine safety monitoring systems. It demonstrated that rare adverse events may only become visible after mass deployment, even when pre authorization trials are extensive. More importantly, it showed that international pharmacovigilance systems can function effectively when clinical observations, laboratory findings, and regulatory communication are rapidly integrated. The speed with which clinicians in different countries identified similar cases, shared findings, and established treatment recommendations has become a model for future adverse event investigation.
Scientific investigation has also advanced understanding of why adenoviral vector vaccines triggered this syndrome in rare individuals. Research increasingly points to interactions between vaccine components and platelet factor 4, with particular attention to adenoviral proteins capable of initiating abnormal immune responses. Among these, the adenoviral protein pVII has emerged as a likely contributing trigger. Identification of this molecular component has significant implications for future vaccine engineering, as it creates the possibility of designing safer vector based vaccines that retain immunogenicity while eliminating components associated with pathogenic PF4 binding.
This knowledge extends beyond COVID 19 vaccines alone. The VITT experience has become a broader case study in how modern medicine can respond to unexpected immune mediated complications associated with biologic therapies. It highlights the need for adaptable regulatory frameworks, rapid translational laboratory support, and clinician education capable of responding in real time to newly emerging syndromes.
In retrospect, VITT represents not only a rare vaccine complication but also a major success in global medical response. A syndrome that initially carried high mortality was rapidly defined, mechanistically understood, and effectively treated through coordinated international effort. For hematologists, immunologists, infectious disease specialists, and public health leaders, VITT now serves as a blueprint for managing rare but severe treatment related adverse events in future vaccine development and emergency public health responses.
The Discovery of VITT: Early Warning Signs After Vaccination
Unusual Thrombosis Patterns in Young Vaccine Recipients
Reports of unexpected venous thromboses emerged within weeks of ChAdOx1 nCoV-19 vaccine rollout. Affected individuals were predominantly young or middle-aged adults, with a notable female preponderance. Clinical presentations deviated from typical thrombotic events. Clots formed in cerebral venous sinuses or within portal, splanchnic, and hepatic veins rather than the deep leg veins commonly associated with venous thromboembolism.
The thrombotic events appeared 5 to 29 days after the first vaccine dose. Patients developed acute onset of moderate to severe thrombocytopenia alongside these unusual clots. The disease proved aggressive, with 40% mortality in early case series. Cerebral venous sinus thrombosis emerged as the most frequent manifestation, occurring in 27 of 40 patients with detailed clinical histories. Splanchnic thrombosis, particularly involving the portal vein, represented another common pattern.
Laboratory investigations revealed a striking finding. Nearly all patients had high levels of antibodies reacting to platelet factor 4-heparin complexes, yet none had been previously exposed to heparin. This paradox suggested an autoimmune mechanism resembling spontaneous heparin-induced thrombocytopenia, which occurs without prior heparin administration.
Three Independent Teams Identify the Same Syndrome
By April 2021, three independent research groups published their findings on what would become known as vaccine-induced immune thrombotic thrombocytopenia. These teams documented 39 cases total, establishing VITT as a distinct clinical entity. The convergence of observations from separate investigators lent credibility to the syndrome’s association with adenoviral vector vaccination.
Teams from Norway, Germany, Austria, and the United Kingdom simultaneously reported cases. Correspondingly, each group noted the unusual timing window of 5 to 24 days post-vaccination and the presence of anti-PF4 antibodies in affected patients. The consistency across geographically dispersed populations ruled out localized manufacturing defects or regional confounders.
Timeline from First Cases to Global Recognition
Concerns about thrombosis with thrombocytopenia developed in March 2021 among recipients of the ChAdOx1 nCoV-19 vaccine. By the end of that month, multiple European medical centers had identified patients admitted to hospitals with the characteristic syndrome. The progression from initial clinical suspicion to formal syndrome identification occurred within weeks.
As of April 4, 2021, the European Union Drug Regulating Authorities’ Pharmacovigilance system (EudraVigilance) had received reports of 169 cases of cerebral venous sinus thrombosis and 53 cases of splanchnic vein thrombosis among approximately 34 million people vaccinated in the European Economic Area and United Kingdom. Given that VITT was only formally identified in April 2021, diagnosis prior to this date proved impossible, raising concerns about underreporting.
The syndrome received various designations initially, including vaccine-induced prothrombotic immune thrombocytopenia and thrombosis with thrombocytopenia syndrome. The term VITT gained acceptance for its acknowledgment of pathogenic similarities with heparin-induced thrombocytopenia. Early case series documented that most patients were younger than 60 years, with few having traditional risk factors for thrombosis.
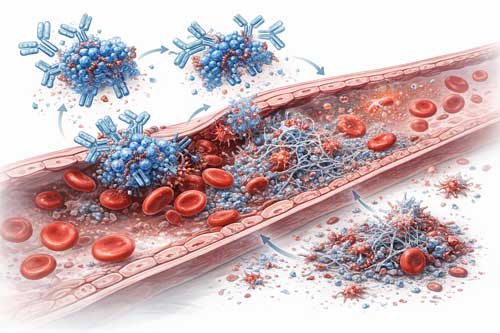
What Is VITT: The Science Behind Vaccine-Induced Thrombocytopenia 
Anti-PF4 Antibodies and Platelet Activation Mechanism
The pathophysiology centers on IgG antibodies targeting platelet factor 4, a positively charged chemokine stored in platelet α-granules. These antibodies, predominantly of the IgG1 subclass, bind to a compact eight-amino acid epitope within PF4’s heparin-binding site. This binding site was identified through alanine-scanning mutagenesis and represents a distinct region from the thirteen-amino acid area targeted by HIT antibodies.
Once generated, anti-PF4 IgG antibodies form immune complexes that engage FcγRIIa receptors on platelets. Receptor clustering triggers intracellular signaling cascades involving Src family kinases, which phosphorylate immunoreceptor tyrosine-based activation motifs. Spleen tyrosine kinase activation follows, subsequently activating phosphoinositide-3 kinase and phospholipase Cγ. This generates inositol triphosphate and diacylglycerol, inducing calcium mobilization and protein kinase C activation.
The signaling cascade produces powerful platelet activation, leading to aggregation, degranulation, and surface expression of procoagulant phosphatidylserine. Activated platelets release extracellular vesicles rich in tissue factor and negatively charged phospholipids, markedly enhancing thrombin generation and fibrin formation. Released PF4 from activated platelets binds additional anti-PF4 antibodies, creating a self-amplifying cycle.
How VITT Differs from Heparin-Induced Thrombocytopenia
VITT antibodies differ substantially from HIT antibodies in their binding properties. Heparin usually inhibits VITT antibody-mediated platelet activation, whereas low heparin concentrations enhance HIT antibody activity. This occurs because VITT antibodies bind the same PF4 site that heparin occupies, explaining why heparin competitively inhibits platelet activation in most VITT cases.
In HIT, heparin crosslinks several PF4 molecules to create the antigenic complex. In contrast, VITT antibodies alone crosslink PF4 without requiring heparin. Analogous to HIT, both conditions require sufficient circulating anti-PF4 antibody levels to create pathogenic immune complexes on platelet surfaces. However, only around 5% of VITT patients develop cross-reacting anti-PF4/heparin antibodies.
Detection methods reflect these differences. Rapid immunoassays demonstrate high sensitivity for HIT (90-97%) but poor sensitivity for VITT (below 25%). PF4-dependent enzyme immunoassays maintain high sensitivity for both disorders, exceeding 99% for HIT and 95% for VITT.
Why Only Adenoviral Vector Vaccines Trigger This Response
Adenoviral vectors possess structural features distinguishing them from mRNA vaccines. Adenoviral capsids and associated nucleic acids carry strong negative charges, functioning as polyanions capable of binding PF4 with high affinity. In vitro studies demonstrated that adenoviral particles readily form stable multimolecular complexes with PF4, whereas lipid nanoparticles and mRNA show no comparable binding properties.
Components of adenoviral vector vaccines act as functional polyanions. Experimental studies revealed that adenoviral capsid proteins, residual free DNA, and other vaccine-associated polyanionic constituents associate with PF4, generating stable complexes with enhanced immunogenicity. These complexes expose cryptic neoepitopes on PF4 not accessible in native conformation, rendering PF4 a danger-associated self-antigen.
The VITT Factor: Immune Complex Formation and FcγRIIa Binding
Immune complex formation requires precise stoichiometric ratios between PF4 and anti-PF4 antibodies for platelet activation. These complexes bind FcγRIIa receptors on both platelets and neutrophils, triggering thromboinflammatory responses. Neutrophil activation leads to neutrophil extracellular trap release, detected in circulating blood and platelet-rich cerebral venous sinus thrombi.
The activation extends beyond platelets. Anti-PF4 immune complexes trigger monocyte activation, promoting tissue factor expression and thrombin generation. They also activate endothelial cells by binding to PF4 linked to endothelial glycosaminoglycans, amplifying the thromboinflammatory response. High-dose intravenous immunoglobulin blocks this FcγRIIa-mediated activation, explaining its therapeutic efficacy.
Clinical Recognition and Diagnostic Breakthroughs 
Key Symptoms Appearing 5-30 Days Post-Vaccination
Recognition depends on identifying the characteristic temporal window. A total of 97% of patients presented between 5 and 30 days after vaccination, with a median onset at 14 days. The 3% who presented between 30 and 48 days had isolated venous thromboembolism without the typical multiorgan involvement. Peak symptom onset occurred during days 6 to 14 post-vaccination.
Persistent and severe headache emerged as the most common presenting symptom, occurring with progressive worsening and resistance to symptomatic treatment. Focal neurological symptoms including blurred or double vision suggested cerebral venous sinus thrombosis or arterial stroke. Severe chest, back, or abdominal pain indicated pulmonary embolism, acute coronary syndrome, or splanchnic vein thrombosis. Unusual bleeding, bruising, petechiae, or blood blisters reflected thrombocytopenia or disseminated intravascular coagulation. Limb swelling with redness, pallor, or coldness suggested deep vein thrombosis or acute limb ischemia.
Laboratory Testing: D-Dimer Elevation and Platelet Count Monitoring
Platelet count and D-dimer represent the most important screening tests. Thrombocytopenia below 150,000 per cubic millimeter occurred in conjunction with markedly elevated D-dimer levels exceeding 4000 fibrinogen-equivalent units. The median initial platelet count in the largest series measured 47 x 10^9/L with wide variability. D-dimer levels below 2000 ng/mL made VITT unlikely. Conversely, values above 4000 FEU, particularly when exceeding eight times the upper limit of normal, strongly suggested VITT even without confirmed thrombosis.
Blood film examination ruled out pseudothrombocytopenia from platelet clumping. Fibrinogen levels warranted measurement, as some patients demonstrated low values indicating consumptive coagulopathy.
ELISA Testing for Anti-PF4 Antibodies
Anti-PF4 antibodies were present in 198 of 220 patients with definite or probable VITT, representing 90% sensitivity. However, only 75% tested positive on the Stago ELISA immunoassay at diagnosis. Different commercial platforms demonstrated varying sensitivity, with the Immucor ELISA showing 100% sensitivity for confirmed cases. Functional assays, including modified serotonin release assays, demonstrated high specificity at 100% but poor sensitivity ranging from 16.7% to 46.2%.
Six patients meeting all other diagnostic criteria had negative ELISA results yet demonstrated profound coagulation activation and received VITT treatment. Moreover, up to one-third of patients showing platelet activation tested ELISA-negative, suggesting functional assays detected cases missed by immunoassays. Rapid tests including chemiluminescent immunoassays proved insufficiently sensitive for VITT detection.
Imaging Requirements for Cerebral and Splanchnic Thrombosis
Cerebral veins represented the most common thrombotic site, occurring in 110 of 220 patients. Unenhanced computed tomography provided a reasonable first test for suspected cerebral venous sinus thrombosis, ruling out alternative diagnoses. However, definitive exclusion required both parenchymal and vascular imaging through CT head with CT venogram or MR head with MR venogram. In 40 patients with cerebral venous thrombosis, secondary intracranial hemorrhage complicated the presentation.
Splanchnic vein thrombosis affected 41 patients, most frequently involving the portal circulation with 30 cases of portal vein thrombosis. Imaging protocols for abdominal thrombosis utilized contrast-enhanced CT or MRI to visualize hepatic, splenic, and mesenteric vessels.
Distinguishing VITT from Other Thrombotic Disorders
D-dimer elevation distinguishes VITT from immune thrombocytopenic purpura, where values remain orders of magnitude lower and thrombosis rarely occurs. In contrast to heparin-induced thrombocytopenia, VITT occurs without heparin exposure and demonstrates different antibody binding properties. The combination of thrombocytopenia, markedly elevated D-dimer, unusual thrombotic sites, and positive PF4-ELISA within the characteristic timeframe establishes the diagnosis[211].
Treatment Protocols That Reduced Mortality by 90% 
Clinical awareness and rapid treatment initiation decreased mortality from 40% to approximately 4-6% within months of syndrome recognition. Treatment success required simultaneous immune modulation and anticoagulation, addressing both antibody-mediated platelet activation and active thrombosis.
High-Dose IVIG as First-Line Immune Modulation
Intravenous immune globulin was administered to 72% of patients at 1 g per kilogram of actual body weight on admission day. Guidelines recommend dosing based on actual rather than ideal body weight, given the dose-dependent inhibition of antibody-induced platelet activation. For cerebral venous sinus thrombosis and splanchnic thrombosis, a second 1 g/kg dose should be given the following day. In other cases, the second dose can be reserved for patients whose platelet count and D-dimer fail to respond by day 3 or 4.
IVIG competitively blocks FcγRIIa receptor binding, preventing immune complex engagement with platelets. Platelet counts typically rise within 12 to 36 hours after the first dose, accompanied by declining D-dimer levels. Systemic glucocorticoids were used in 26% of patients and in 50% of those with platelet counts below 30,000 per cubic millimeter.
Non-Heparin Anticoagulation Strategies
Anticoagulation was administered in 91% of patients, with non-heparin-based agents used in 68%. Safe options include argatroban, fondaparinux, apixaban, and dabigatran. Heparin was administered at some point during admission in 23% of patients, primarily those presenting before mid-March when VITT remained unrecognized. Mortality among heparin-treated patients reached 20%, compared with 16% among those receiving non-heparin anticoagulants.
Why Traditional Treatments Like Aspirin Must Be Avoided
Aspirin does not prevent platelet activation in VITT and increases bleeding risk. Vitamin K antagonists must be avoided in acute VITT, as they cause protein C deficiency and increase microcirculatory thrombosis risk in hypercoagulable states. Platelet transfusions should be avoided, as they may fuel the prothrombotic state.
Plasma Exchange for Severe Cases
Seventeen patients with severe disease involving cerebral venous sinus thrombosis or thrombosis at multiple sites underwent plasma exchange, achieving 90% survival. Both patients who died had extensive intracranial hemorrhage at presentation. Plasma exchange removes anti-PF4 antibodies and should be considered for patients showing continued thrombosis despite IVIG and anticoagulation.
Managing Complications: Bleeding Risk with Low Platelet Counts
Thrombocytopenia does not contraindicate therapeutic-dose anticoagulation in VITT. Patients with the lowest platelet counts may face highest thrombosis risk, making IVIG administration critical to raise platelet counts while maintaining anticoagulation.
Broader Implications for Vaccine Safety Monitoring Systems 
The VITT experience transformed vaccine safety monitoring and future vaccine development strategies globally.
Rapid Surveillance Networks Detect Rare Adverse Events
Active surveillance systems proved essential for detecting VITT. The Vaccine Safety Datalink monitored over 10 million vaccinated individuals through electronic health records, enabling near-real-time adverse event detection. Among 16 million vaccinated individuals across three commercial insurance databases, rapid surveillance identified myocarditis and anaphylaxis signals within weeks. In effect, passive reporting through VAERS complemented active monitoring, with FDA and CDC analyzing report trends to identify safety concerns. Disproportionality analysis detected patterns suggesting associations between vaccines and specific adverse events, though these results required confirmation through controlled studies.
Manufacturing Changes to Reduce VITT Risk in Future Vaccines
Scientists identified the adenoviral protein pVII as triggering anti-PF4 antibody production. Modifying or removing this specific protein allows future vaccines to avoid VITT while preserving efficacy. Engineering strategies including insect-specific flaviviruses, chimeric capsids evading neutralizing antibodies, and integrase-defective lentiviral vectors enhance safety profiles.
Heterologous Prime-Boost Vaccination Strategies
Heterologous vaccination emerged as a response to VITT concerns. Priming with adenoviral vector followed by mRNA boost induced 20- to 60-fold higher neutralizing antibody titers compared to homologous regimens. Real-world data confirmed ChAd-BNT heterologous schedules produced antibody responses similar to two BNT doses while avoiding second adenoviral exposure.
Long-Term Follow-Up: Anti-PF4 Antibody Persistence Patterns
Anti-PF4 antibodies persisted for months to years in subsets of patients. ELISA positivity continued beyond functional platelet activation in most cases, indicating that serological persistence does not equate to sustained thrombotic risk. Platelet-activating antibodies became negative within 15.5 weeks in 74% of patients. Remarkably, one patient demonstrated platelet-activating antibodies four years post-vaccination with chronic low-grade thrombocytopenia.
Global Education Programs and Clinical Preparedness
The UK Expert Hematology Panel advised on over 300 potential VITT cases, producing living guidance that reduced mortality through standardized diagnostic pathways. Members conducted multi-media interviews balancing public education without jeopardizing vaccination confidence.
Application to Other Adenoviral Vector Vaccine Development
VITT insights extend beyond COVID-19 to Ebola and other adenoviral vaccines under development. Platform-based approaches incorporating VITT prevention mechanisms position future vaccines for rapid deployment during public health emergencies.
Conclusion 

The rapid identification and characterization of VITT represents a remarkable achievement in global medical collaboration. Three independent research teams converged on the same findings within weeks, establishing diagnostic criteria that transformed a 40% mortality rate into outcomes below 6%. This success emerged through coordinated efforts spanning laboratory investigations, clinical observations, and treatment innovation. Most important, the lessons learned extend beyond COVID-19 vaccination. Modified adenoviral vectors, heterologous vaccination strategies, and enhanced surveillance networks now provide frameworks for future vaccine development. The medical community demonstrated that rare complications, once recognized and understood through rigorous investigation, can be effectively managed through targeted therapeutic interventions.

FAQs 
Q1. What symptoms should prompt immediate medical attention after receiving an adenoviral vector COVID-19 vaccine? Severe and persistent headache, visual disturbances (blurred or double vision), severe abdominal or back pain, unusual bleeding or bruising, shortness of breath, or limb swelling occurring between 5 to 30 days after vaccination warrant urgent medical evaluation. These symptoms may indicate VITT, particularly if they appear during the peak risk window of 6 to 14 days post-vaccination.
Q2. How does VITT differ from heparin-induced thrombocytopenia (HIT)? While both conditions involve anti-PF4 antibodies causing platelet activation, VITT occurs without any heparin exposure and the antibodies bind directly to PF4 without requiring heparin. Additionally, heparin typically inhibits platelet activation in VITT cases, whereas low heparin concentrations enhance antibody activity in HIT. Only about 5% of VITT patients develop antibodies that cross-react with heparin.
Q3. Why do only adenoviral vector vaccines trigger VITT and not mRNA vaccines? Adenoviral vectors contain strongly negatively charged components, including capsid proteins and residual DNA, that bind tightly to platelet factor 4 (PF4). These complexes expose hidden parts of PF4 that trigger antibody production. In contrast, the lipid nanoparticles and mRNA in mRNA vaccines show no comparable binding properties with PF4.
Q4. How did treatment advances reduce VITT mortality rates so dramatically? Once clinicians recognized VITT and implemented proper treatment protocols, mortality decreased from 40% to approximately 4-6%. The key interventions include high-dose intravenous immunoglobulin (IVIG) to block antibody-platelet interactions, non-heparin anticoagulants to prevent clot formation, and plasma exchange for severe cases. Early recognition and avoiding contraindicated treatments like aspirin and platelet transfusions also contributed significantly.
Q5. How long do anti-PF4 antibodies persist after VITT diagnosis? Anti-PF4 antibodies can persist for months to years in some patients, though their presence doesn’t necessarily indicate ongoing thrombotic risk. The platelet-activating antibodies typically become negative within about 15.5 weeks in 74% of patients. However, serological tests may remain positive longer than the functional risk period, meaning a positive antibody test alone doesn’t equate to sustained danger.
References: 
American Society of Hematology. (2022, May 9). Vaccine-induced immune thrombotic thrombocytopenia. https://www.hematology.org/covid-19/vaccine-induced-immune-thrombotic-thrombocytopenia
Bissola, A.-L., Daka, M., Arnold, D. M., Nazi, I., Smith, J. W., Moore, J. C., Nazy, I., et al. (2022). The clinical and laboratory diagnosis of vaccine-induced immune thrombotic thrombocytopenia. Blood Advances, 6(14), 4228–4235. https://doi.org/10.1182/bloodadvances.2022007766
Bourguignon, A., Arnold, D. M., Warkentin, T. E., Smith, J. W., Pannu, T., Shrum, J. M., Al Maqrashi, Z. H., Shroff, A., Lessard, M.-C., Blais, N., et al. (2021). Adjunct immune globulin for vaccine-induced immune thrombotic thrombocytopenia. New England Journal of Medicine, 385(8), 720–728. https://doi.org/10.1056/NEJMoa2107051
Centers for Disease Control and Prevention. (2025, September 12). About the Vaccine Safety Datalink (VSD). https://www.cdc.gov/vaccine-safety-systems/vsd/index.html
Chevassut, T. (2021). VITT, COVID-19 and the Expert Haematology Panel: The story of how the UK responded to emerging cases of vaccine-induced immune thrombocytopenia and thrombosis during the vaccination programme. Clinical Medicine, 21(6), e600–e604. https://doi.org/10.7861/clinmed.2021-0488
Cines, D. B., & Bussel, J. B. (2022). SARS-CoV-2 vaccine-induced immune thrombotic thrombocytopenia. Journal of Infection. https://www.journalofinfection.com/article/S0163-4453(22)00055-X/fulltext
Favaloro, E. J., Clifford, J., Leitinger, E., Parker, M., Sung, P., Chunilal, S., Tran, H., Kershaw, G., Fu, S., Passam, F., et al. (2022). Assessment of immunological anti-platelet factor 4 antibodies for vaccine-induced thrombotic thrombocytopenia (VITT) in a large Australian cohort: A multicenter study comprising 1284 patients. Journal of Thrombosis and Haemostasis, 20(12), 2896–2908. https://doi.org/10.1111/jth.15881
Goodwin, G., Latimore, C., & Baker, A. (2021, November 3). ITP vs. VITT: Rare but distinct conditions associated with the COVID-19 vaccines. Florida College of Emergency Physicians. https://fcep.org/itp-vs-vitt-rare-but-distinct-conditions-associated-with-the-covid-19-vaccines/
Gabarin, N., Arnold, D. M., Nazy, I., & Warkentin, T. E. (2022). Treatment of vaccine-induced immune thrombotic thrombocytopenia (VITT). Seminars in Hematology, 59(2), 89–96. https://doi.org/10.1053/j.seminhematol.2022.03.002
Greinacher, A., Langer, F., Makris, M., Pai, M., Pavord, S., Tran, H., & Warkentin, T. E. (2022). Vaccine-induced immune thrombotic thrombocytopenia (VITT): Update on diagnosis and management considering different resources. Journal of Thrombosis and Haemostasis, 20(1), 149–156. https://doi.org/10.1111/jth.15572
Han, J., Vargo, J., & Semelka, R. C. (2015). Imaging diagnosis of splanchnic venous thrombosis. Gastroenterology Research and Practice, 2015, Article 101029. https://doi.org/10.1155/2015/101029
Huang, C.-T., Chao, T.-H., Hsu, H.-L., Chiou, C.-Y., Lin, J.-C., Chang, H.-W., et al. (2021). Double high-dose immunoglobulin for ChAdOx1 nCov-19 vaccine-induced immune thrombotic thrombocytopenia. Thrombosis Research, 206, 14–17. https://doi.org/10.1016/j.thromres.2021.07.017
Lee, C. S. M., Clarke, L. J., Kershaw, G. W., Tohidi-Esfahani, I., Brighton, T. A., Chunilal, S., Favaloro, E. J., Tran, H., Chen, V. M., et al. (2023). Platelet-activating functional assay resolution in vaccine-induced immune thrombotic thrombocytopenia: Differential alignment to PF4 ELISA platforms. Research and Practice in Thrombosis and Haemostasis, 7(3), 100128. https://doi.org/10.1016/j.rpth.2023.100128
Lloyd, P. C., Hu, M., Wong, H.-L., Shoaibi, A., Ke, C., Anderson, S. A., et al. (2022). Near real-time surveillance of safety outcomes in US COVID-19 vaccine recipients aged 12 to 64 years. Vaccine, 40(45), 6481–6488. https://doi.org/10.1016/j.vaccine.2022.09.060
McCrae, K. R. (2021). Thrombotic thrombocytopenia due to SARS-CoV-2 vaccination. Cleveland Clinic Journal of Medicine. Advance online publication. https://doi.org/10.3949/ccjm.88a.ccc078
Ontario COVID-19 Science Advisory Table. (2021, May 10). Vaccine-induced immune thrombotic thrombocytopenia (VITT) following adenovirus vector COVID-19 vaccination: Interim guidance for healthcare professionals in emergency department and inpatient settings. https://doi.org/10.47326/ocsat.2021.02.21.2.0
Pavord, S., Scully, M., Hunt, B. J., Lester, W., Bagot, C., Craven, B., Rampotas, A., Ambler, G., & Makris, M. (2021). Clinical features of vaccine-induced immune thrombocytopenia and thrombosis. New England Journal of Medicine, 385(18), 1680–1689. https://doi.org/10.1056/NEJMoa2109908
Platelet factor 4 antibody persistence and long-term outcomes after vaccine-induced immune thrombotic thrombocytopenia. (2025, November 27). medRxiv [Preprint]. https://doi.org/10.1101/2025.11.24.25340592
Reguzova, A., Haug, V., Metz, C., Müller, M., Fandrich, M., Dulovic, A., & Amann, R. (2025). Heterologous prime-boost vaccination with VLA2001 and an ORFV-based vector enhances spike- and nucleocapsid-specific immunity in mice. Frontiers in Immunology, 16, 1675859. https://doi.org/10.3389/fimmu.2025.1675859
Schönborn, L., Thiele, T., Kaderali, L., Günther, A., Neumann, C., Köhler, D., Römisch, J., Möckel, M., Hohenstein, S., Tiede, A., Hütelmaier, S., Greinacher, A., et al. (2022). Most anti-PF4 antibodies in vaccine-induced immune thrombotic thrombocytopenia are transient. Blood, 139(12), 1903–1907. https://doi.org/10.1182/blood.2021014214
Siniscalchi, C., Basaglia, M., Tufano, A., Imbalzano, E., & Di Micco, P. (2026). Vaccine-induced immune thrombotic thrombocytopenia (VITT): An immunopathogenic model of dysregulated vaccine-triggered immunity. Vaccines, 14(3), 225. https://doi.org/10.3390/vaccines14030225
Tang, J., Amin, M. A., & Campian, J. L. (2025). Past, present, and future of viral vector vaccine platforms: A comprehensive review. Vaccines, 13(5), 524. https://doi.org/10.3390/vaccines13050524
Uaprasert, N., Watanaboonyongcharoen, P., Vichitratchaneekorn, R., Trithiphen, S., Akkawat, B., Sukperm, A., Tongbai, T., Jantarabenjakul, W., Paitoonpong, L., & Rojnuckarin, P. (2021). Prevalence of thrombocytopenia, anti-platelet factor 4 antibodies and D-dimer elevation in Thai people after ChAdOx1 nCoV-19 vaccination. Research and Practice in Thrombosis and Haemostasis, 5(6), e12580. https://doi.org/10.1002/rth2.12580
U.S. Food and Drug Administration. (2025, January 10). COVID-19 vaccine safety surveillance. https://www.fda.gov/vaccines-blood-biologics/safety-availability-biologics/covid-19-vaccine-safety-surveillance
Van Beusekom, M. (2026, February 12). Researchers pinpoint cause of rare but life-threatening blood clots after adenovirus-based COVID vaccination. CIDRAP. https://www.cidrap.umn.edu/covid-19/researchers-pinpoint-cause-rare-life-threatening-blood-clots-after-adenovirus-based-covid
Video Section
Check out our extensive video library (see channel for our latest videos)
Recent Articles


