New Treatments for Atopic Dermatitis in 2026 Why More Options Might Be Making Your Choice Harder

Key Takeaways
The treatment landscape for atopic dermatitis has changed profoundly over the past several years, moving from a limited set of broadly acting immunosuppressive therapies to an increasingly sophisticated range of targeted systemic and topical interventions. This expansion has created new opportunities for individualized care while simultaneously introducing greater complexity into therapeutic decision making. For clinicians managing moderate to severe atopic dermatitis, the challenge is no longer the absence of effective options but rather selecting the most appropriate treatment from a growing number of agents with distinct mechanisms of action, efficacy profiles, safety considerations, and practical implications for long term disease control.
Atopic dermatitis is a chronic inflammatory skin disorder characterized by epidermal barrier dysfunction, immune dysregulation, intense pruritus, and recurrent eczematous lesions. It affects approximately 13 percent of children and 7 percent of adults worldwide, with an estimated 10 to 30 percent of patients experiencing moderate to severe disease that may require systemic therapy. Beyond visible skin involvement, the disease imposes a substantial burden on quality of life, affecting sleep, mental health, work productivity, and social functioning. Persistent itch, recurrent flares, and the psychosocial consequences of chronic disease often make therapeutic goals extend well beyond lesion clearance alone.
Historically, systemic treatment options for moderate to severe atopic dermatitis were limited to conventional immunosuppressive agents such as cyclosporine, methotrexate, azathioprine, and mycophenolate mofetil. While these medications can be effective in selected patients, their use is constrained by non selective immune suppression, cumulative toxicity, laboratory monitoring requirements, and limited suitability for long term disease control. Cyclosporine, for example, often provides rapid symptom improvement but is associated with nephrotoxicity and hypertension, restricting prolonged use. Methotrexate and azathioprine may offer maintenance benefit but require close hematologic and hepatic surveillance and may demonstrate delayed onset of action.
The introduction of biologic therapies has significantly altered this treatment paradigm by targeting specific inflammatory pathways central to atopic dermatitis pathogenesis. Dupilumab, the first widely adopted biologic in this setting, targets the interleukin 4 receptor alpha subunit, thereby inhibiting signaling through both interleukin 4 and interleukin 13 pathways that are central to type 2 inflammation. Its approval established a new standard for targeted systemic treatment and demonstrated that selective immune modulation could achieve substantial disease control with a favorable long term safety profile.
Subsequent biologic agents have further refined this targeted approach. Tralokinumab and lebrikizumab selectively inhibit interleukin 13, reflecting the increasingly precise understanding of cytokine mediated disease mechanisms. Nemolizumab, which targets interleukin 31 receptor signaling, addresses pruritus more directly by interrupting pathways specifically involved in itch transmission. These biologic therapies provide important alternatives for patients who do not achieve adequate response with initial biologic treatment or who require a different adverse effect profile.
At the same time, oral Janus kinase inhibitors have emerged as a powerful new therapeutic class. Agents such as upadacitinib, abrocitinib, and baricitinib inhibit intracellular signaling pathways involved in multiple inflammatory cytokine networks, producing rapid reductions in inflammation and itch. In comparative studies, JAK inhibitors often demonstrate faster symptom relief than biologics, with some patients experiencing meaningful improvement within days of initiation. This rapid onset is particularly valuable in patients with severe pruritus, sleep disruption, or extensive inflammatory burden where immediate disease control is clinically desirable.
However, the clinical advantages of JAK inhibitors are accompanied by important safety considerations. Regulatory agencies have issued boxed warnings related to risks of serious infection, thromboembolic events, cardiovascular complications, and malignancy, particularly in populations with preexisting risk factors. As a result, patient selection for JAK inhibitor therapy requires careful evaluation of cardiovascular history, malignancy risk, infection susceptibility, and laboratory parameters. Ongoing monitoring of complete blood counts, liver function, and lipid profiles remains essential during treatment.
Topical innovation has also contributed significantly to the modern therapeutic era. Newer non steroid topical agents such as tapinarof, roflumilast, and topical ruxolitinib provide targeted anti inflammatory options that may reduce dependence on topical corticosteroids. These agents are particularly relevant for patients with localized disease, steroid sensitive anatomical areas, or long term maintenance needs. Their emergence broadens treatment flexibility and supports earlier intervention before escalation to systemic therapy.
Despite these advances, no single therapy consistently emerges as the best option for every patient. Clinical experience and real world persistence data indicate that treatment switching remains common, even among highly effective agents. Variability in treatment response reflects differences in disease phenotype, immunologic drivers, comorbid conditions, adherence patterns, and patient expectations. Some individuals prioritize rapid itch relief, while others focus on minimizing injection frequency, reducing laboratory monitoring, or preserving reproductive safety.
This divergence between clinician priorities and patient priorities is increasingly recognized as central to treatment success. Clinicians often focus on complete adverse event profiles, long term safety, and objective disease severity measures. Patients, however, frequently emphasize treatment convenience, route of administration, speed of symptom relief, flexibility in stopping therapy when desired, and impact on daily functioning. For some patients, oral therapy may be preferable to injections despite added monitoring requirements. Others may favor biologic therapy because of long term safety reassurance and reduced need for frequent laboratory testing.
Age and reproductive status further complicate therapeutic selection. Dupilumab remains the only biologic currently approved for children younger than 12 years in many clinical settings, making it a key option in pediatric moderate to severe disease. Adolescents and younger adults may have broader access to additional biologics and JAK inhibitors, but age specific safety profiles must still be considered carefully. Pregnancy and breastfeeding introduce further restrictions, particularly for JAK inhibitors, which are generally avoided because of insufficient safety data and concerns regarding fetal risk. In women of reproductive age, these considerations often influence first line systemic treatment selection.
The expanding number of available therapies has therefore created a paradox in atopic dermatitis management. Increased choice has improved the ability to individualize treatment, yet it has also made therapeutic decisions more complex. Mechanism of action, onset speed, safety monitoring burden, comorbidity profile, patient lifestyle, age, reproductive considerations, and long term treatment goals must all be weighed simultaneously.
This evolving environment makes shared decision making more essential than ever. Effective treatment planning now depends on detailed discussion between dermatologists and patients regarding expected benefits, potential risks, route preferences, monitoring obligations, and practical life considerations. The most successful treatment strategy often emerges not from selecting the most potent medication in abstract terms, but from aligning therapeutic characteristics with the specific priorities and clinical realities of the individual patient.
As the therapeutic scope continues to expand, comparative effectiveness research and long term safety data will become increasingly important in guiding evidence based sequencing of available therapies. The future of atopic dermatitis management lies not only in therapeutic innovation but also in refining how clinicians choose among these options to achieve durable disease control, improved quality of life, and sustainable long term outcomes.
The expanding treatment landscape for atopic dermatitis in 2026
The therapeutic armamentarium for moderate to severe atopic dermatitis has undergone substantial reorganization. Medications now group into four distinct classes: phototherapy, immunosuppressants, biologics, and Janus kinase inhibitors [1]. This classification scheme reflects both mechanistic understanding and regulatory evolution. The pace of drug development has accelerated markedly since 2021, when dupilumab stood as the sole biologic option for atopic dermatitis.
FDA-approved biologics now available
Biologics targeting specific inflammatory pathways have emerged as cornerstone therapies. Dupilumab remains the established first-line biologic, blocking both IL-4 and IL-13 signaling [2]. More recently, selective IL-13 inhibitors entered clinical practice. Lebrikizumab received FDA approval for moderate to severe atopic dermatitis, with phase 3 trial data demonstrating that almost two-thirds of patients with baseline hyperpigmentation showed improvement at week 24 [2]. Tralokinumab provides another IL-13-targeted option [3].
Nemolizumab represents a mechanistic departure as the first FDA-approved monoclonal antibody exclusively targeting the IL-31 receptor alpha [2]. This approval followed positive results from the phase 3 ARCADIA clinical trial program. The IL-31 pathway plays a central role in pruritus transmission, distinguishing nemolizumab’s therapeutic focus from other biologics that primarily target skin inflammation.
New oral JAK inhibitors entering the market
Three oral JAK inhibitors received FDA approval for atopic dermatitis treatment: abrocitinib, upadacitinib, and baricitinib [2] [3]. These small molecules inhibit Janus kinase pathways, with JAK1-centric agents most commonly prescribed in atopic dermatitis to quiet cytokine signaling quickly [2]. As opposed to biologics requiring subcutaneous injection, oral JAK inhibitors offer convenient once-daily dosing.
In 2021, the FDA mandated boxed warnings across the JAK class for increased risks of serious cardiovascular events, certain malignancies, thrombosis, and mortality observed in some inflammatory disease populations [2]. Eczema dosing and risk profiles differ from other indications, yet clinicians maintain careful screening and monitoring protocols. Regional availability varies, with some countries restricting JAK inhibitor use based on age or comorbidity profiles.
Topical treatments gaining traction
The expansion of new topical and systemic treatments for atopic dermatitis includes multiple nonsteroidal options approved between 2024 and 2026. Tapinarof cream received FDA approval for patients aged 2 years and older, with phase 3 ADORING trials showing that 45.4% and 46.4% of patients achieved clear or almost clear skin by week 8, compared with 18% on vehicle [2]. The aryl hydrocarbon receptor agonist supports both anti-inflammatory effects and barrier restoration [4].
Roflumilast cream 0.05%, a topical PDE4 inhibitor, expanded treatment options for very young children with once-daily dosing and good tolerability in diverse cohorts [4] [3]. In February 2026, the FDA approved difamilast for mild to moderate atopic dermatitis [3], further broadening the topical landscape.
Topical JAK inhibitors include ruxolitinib cream, which gained approval for patients as young as 2 years based on phase 3 TRuE-AD3 data [2]. The JAK1/JAK2 inhibitor demonstrates rapid antipruritic efficacy, with itch reduction occurring within minutes to hours and higher rates of achieving a Numeric Rating Scale itch score of 0 or 1 versus vehicle [4]. Delgocitinib received FDA approval for moderate to severe chronic hand eczema, with DELTA 1 and DELTA 2 studies showing overall patient response rates of 20% versus 10% vehicle and 29% versus 7% vehicle, respectively [2] [3]. Tofacitinib ointment 2% demonstrated favorable safety and efficacy in mild-to-moderate atopic dermatitis patients following 4 weeks of treatment in a phase 3 trial [5].
Why having more options creates confusion
Patient decision aid research reveals the complexity introduced by treatment expansion. Patients rank efficacy highest among decision factors, followed by adverse events and cost, whereas clinicians prioritize complete adverse event inventories, laboratory monitoring requirements, and granular efficacy data [1]. This misalignment complicates shared decision-making.
Beta testing of patient decision aids with 8 patients and 6 clinicians revealed confusion with graphic design and organization [1]. Some patients expressed that visual layout might bias them toward certain treatments, while clinicians voiced concern that poor visual organization could lead to suboptimal decision-making. A staged approach to presenting treatment information addresses this challenge by allowing patients to process options more effectively and eliminate therapy classes they are not interested in pursuing [1]. The final patient decision aid version incorporates a color-coded arrow system to help visual learners interpret efficacy levels, with medications organized by class to reduce repetition [1].
New topical and systemic treatments for atopic dermatitis 
Nonsteroidal topical agents have reshaped treatment algorithms for mild to moderate atopic dermatitis, offering alternatives to corticosteroids with distinct mechanisms of action. Simultaneously, the decision to escalate from topical to systemic therapy has become more nuanced as treatment options multiply.
Roflumilast and tapinarof for mild to moderate cases
Roflumilast cream 0.15% functions as a phosphodiesterase-4 inhibitor applied once daily. In the INTEGUMENT-1 trial, 32.0% of patients achieved validated Investigator Global Assessment success at week 4 compared with 15.2% on vehicle [1]. The INTEGUMENT-2 trial produced similar outcomes, with 28.9% versus 12.0% reaching the primary endpoint [1]. At week 4, 43.2% and 42.0% of roflumilast-treated patients in the respective trials achieved at least 75% reduction in Eczema Area and Severity Index, versus 22.0% and 19.7% on vehicle [1][1]. Itch reduction occurred within 24 hours after initial application [1]. The formulation uses a water-based vehicle without sensitizers or penetration enhancers, designed to protect rather than disrupt the skin barrier [1]. Application-site pain affected only 0.9% and 2.1% of patients in the two trials [1].
Tapinarof cream 1% represents an aryl hydrocarbon receptor agonist approved in December 2024 for patients aged 2 years and older [1]. The compound activates AhR, a ligand-dependent transcription factor residing in keratinocyte cytoplasm [1]. Upon binding, AhR translocates to the nucleus and forms a heterodimer with AhR nuclear translocator, initiating transcriptional changes that downregulate IL-4, IL-13, and IL-17 while promoting barrier protein expression including filaggrin, loricrin, and involucrin [1]. In the ADORING 1 and ADORING 2 trials, 45.4% and 46.4% of patients achieved clear or almost clear skin at week 8, compared with 13.9% and 18.0% on vehicle [1]. The label carries no warnings, precautions, contraindications, or restrictions on treatment duration or body surface area [1]. For patients achieving complete clearance who then discontinued treatment, the mean duration of the first treatment-free interval reached approximately 80 consecutive days [1].
Topical JAK inhibitors: ruxolitinib and delgocitinib
Ruxolitinib functions as a JAK1/2 inhibitor approved for mild to moderate atopic dermatitis in patients aged 2 years and older since September 2025 [1]. Pharmacokinetic studies demonstrate effective dermal concentrations with minimal systemic exposure [1]. In the TRuE-AD1/2 phase 3 programs, treatment-emergent adverse events occurred in 34% to 67% of patients, with severity predominantly mild to moderate and rates comparable to vehicle [1]. Upper respiratory infections, nasopharyngitis, and headaches represented the most common events, occurring at population-baseline frequencies [1]. Serious infections remained rare and were not causally linked to ruxolitinib treatment [1].
Delgocitinib operates as a pan-JAK inhibitor approved in Japan in 2020 and by the European Medicines Agency in 2024 for chronic hand eczema [1]. The compound exists exclusively as a topical formulation [1]. Adverse event profiles across DELTA 1 and DELTA 2 trials showed treatment-emergent events in 45% to 68.3% of patients, with incidence rates comparable to placebo [1]. Negligible systemic absorption characterizes both topical JAK inhibitors, distinguishing their safety profiles from oral formulations that carry class warnings for thrombosis and cardiovascular events [1].
When to consider topical vs systemic options
The International Eczema Council recommends systemic therapy when aggressive topical therapy fails to achieve disease control [1]. Conversely, this guideline lacks specificity, as one clinician’s definition of aggressive topical therapy may differ substantially from another’s [1]. The Atopic Dermatitis Control Tool addresses this variability through a validated six-item questionnaire assessing sleep, intense itching, emotions, and daily activities [1].
Patients with body surface area exceeding 10% or sensitive area involvement affecting hands, groin, or scalp who use topical steroids consistently yet fail to clear warrant immediate systemic consideration [1]. Likewise, overuse of high-potency topical steroids constitutes treatment failure and risks complications including perioral dermatitis, atrophy, and steroid withdrawal [1]. According to current practice patterns, even highly efficacious systemic agents like upadacitinib rarely achieve complete clearance at every time point, meaning most patients require topical therapy at some stage [1].

Biologics: understanding IL-4, IL-13, and IL-31 inhibitors 
Monoclonal antibodies represent targeted therapies that inhibit specific cytokines within the type 2 inflammatory cascade underlying atopic dermatitis pathophysiology. These biologics demonstrate distinct mechanisms, translating to varied clinical outcomes and safety profiles.
Dupilumab: the established first-line biologic
Dupilumab functions as a fully human IgG4 monoclonal antibody that selectively binds to IL-4 receptor alpha, thereby inhibiting signaling of both IL-4 and IL-13 pathways [1]. The FDA approved dupilumab in March 2017 for adults with moderate to severe atopic dermatitis inadequately controlled by topical treatments, with subsequent approval extending to patients aged 6 months and older in 2022 [1]. The standard adult dosing involves an initial 600 mg subcutaneous administration followed by 300 mg every two weeks [1].
Real-world evidence demonstrates robust treatment persistence. Global studies report dupilumab drug survival rates of 80% to 90% through 3 years in adult patients [1]. Pediatric outcomes mirror adult results, with drug survival reaching 90.1% over 3 years, although insufficient effectiveness constitutes the primary discontinuation reason [1]. Dupilumab holds approval for severe asthma treatment, with BioDay registry data confirming beneficial effects on comorbid asthma in both adult and pediatric atopic dermatitis populations [1]. The medication does not function as an immunosuppressant, distinguishing it from conventional systemic therapies [6].
Tralokinumab and lebrikizumab: selective IL-13 targeting
Tralokinumab operates as a fully human IgG4 monoclonal antibody that potently neutralizes IL-13 by preventing its binding to IL-13 receptor α1 and IL-13 receptor α2 [1]. The European Commission approved tralokinumab for patients aged 12 years and older with moderate to severe disease. Dosing begins with 600 mg administered as four subcutaneous injections, followed by 300 mg every two weeks, with the option to extend intervals to every four weeks for responders after 16 weeks [1]. Real-world data show 1-year and 2-year drug survival rates of 78.1% to 96.5% and 66.8%, respectively [1]. In contrast to dupilumab, tralokinumab demonstrated no clinically meaningful benefit in phase 3 asthma trials [1].
Lebrikizumab represents a high-affinity IgG4 monoclonal antibody that selectively binds soluble IL-13, preventing formation of the IL-4 receptor α–IL-13 receptor α1 heterodimer signaling complex while maintaining IL-4 signaling [6]. The European Commission approved lebrikizumab for patients aged 12 years and older weighing more than 40 kg, though FDA approval remains pending as of early 2026 [1]. Phase 3 trials demonstrated IGA response rates of 43.1% and 33.2% at week 16, compared with 12.7% and 10.8% for placebo [6]. EASI-75 responses occurred in 58.8% and 52.1% of lebrikizumab-treated patients versus 16.2% and 18.1% on placebo [6]. Both tralokinumab and lebrikizumab permit every four week dosing following loading regimens, whereas dupilumab carries only every two week labeling for adults [1].
Nemolizumab: targeting itch through IL-31
Nemolizumab represents a humanized IgG2 monoclonal antibody that antagonizes IL-31 receptor alpha, inhibiting IL-31 signaling on neurons and immune cells [7]. IL-31 drives pruritus transmission, making this pathway particularly relevant for patients reporting itch as their primary burden. The FDA approved nemolizumab in December 2025 for adolescents and adults with moderate to severe atopic dermatitis, with Japan granting earlier approval for patients aged 13 years and older at 60 mg every four weeks [1].
Phase 3 ARCADIA trials enrolled 1,728 patients receiving nemolizumab plus topical corticosteroids. At week 16, IGA success rates reached 36% and 38% in the two trials, compared with 25% and 26% for placebo [8]. EASI-75 responses occurred in 44% and 42% of nemolizumab arms versus 29% and 30% of placebo groups [8]. Notably, 68% of patients achieved itch relief by day 2, sustained through week 16 [7].
How biologics differ in efficacy and side effects
Ocular surface disease emerges as a class-related concern with varying incidence. Clinical practice reports identify ocular complications in up to 40% of dupilumab-treated patients, substantially exceeding the 3.0% to 11.1% rates observed in clinical trials [1]. Tralokinumab trials documented ocular events in 3.0% to 11.1% of patients [1]. Lebrikizumab demonstrated conjunctivitis in approximately 7.5% during short-term trials, increasing to 14% after 52 weeks [1].
Additional dupilumab-associated events include psoriasiform manifestations in 1.8% to 3.3% of patients, facial redness in 11% to 35% of adults, and musculoskeletal adverse events at an incidence of 4.3 per 100 patient-years [1].
JAK inhibitors: comparing upadacitinib, abrocitinib, and baricitinib 
Three oral JAK inhibitors received FDA approval for moderate to severe atopic dermatitis: upadacitinib, abrocitinib, and baricitinib. These agents differ in selectivity profiles, with upadacitinib and abrocitinib demonstrating preferential JAK1 inhibition while baricitinib inhibits both JAK1 and JAK2. No head-to-head trials directly compare these medications, necessitating reliance on network meta-analyzes for comparative effectiveness assessment.
Faster symptom relief but safety concerns
Network meta-analysis reveals upadacitinib 30 mg demonstrates superior efficacy across all treatment regimens for both IGA and EASI response measures [9]. Upadacitinib 15 mg outperforms remaining regimens except abrocitinib 200 mg [9]. Moreover, abrocitinib 200 mg surpasses abrocitinib 100 mg and all baricitinib doses in clinical efficacy [9]. Patients achieve higher odds of reaching 50% improvement in EASI with upadacitinib 30 mg daily compared with dupilumab, with an odds ratio of 2.5 [6]. Conversely, baricitinib at both 2 mg and 4 mg doses consistently ranks in the lowest treatment positions across efficacy endpoints [10].
Upadacitinib 30 mg attains probabilities ranging from 97% to 98% of being the most effective option across different endpoints [10]. In assessments of EASI 75 scores, upadacitinib 30 mg yields an odds ratio of 12.3, while IGA-AD 0/1 response produces an odds ratio of 18.9 [10]. Pruritus severity reduction of at least 4 points on the Itch Numeric Rating Scale shows an odds ratio of 11.1 with upadacitinib 30 mg treatment [10].
However, treatment-emergent adverse events occur more frequently with abrocitinib and upadacitinib compared with placebo regardless of dose [9][6]. Upadacitinib 30 mg causes more TEAEs, with subgroup network meta-analysis revealing an odds ratio of 6.71 for increased incidence [6]. Real-world data from 202 patients demonstrates treatment suspension rates of 41.18% in the JAK inhibitor group compared with 22.39% in the biologics group [11]. Common manageable adverse effects include upper respiratory infections, nasopharyngitis, nausea, headache, and acne [7].
Black box warnings you need to know
The FDA requires boxed warnings for upadacitinib, baricitinib, and abrocitinib regarding increased risks of serious heart-related events, cancer, blood clots, and death [8]. These warnings stem from a completed safety trial of tofacitinib in rheumatoid arthritis patients aged 50 years or older with cardiovascular risk factors [8]. Since upadacitinib and baricitinib share mechanisms of action with tofacitinib but lack similar large safety trials, FDA considers these medicines may have similar risks [8].
Nevertheless, cardiovascular disease concerns in rheumatoid arthritis may not translate directly to atopic dermatitis populations. Rheumatoid arthritis patients tend to be older, inactive, overweight, and have smoking histories, whereas atopic dermatitis patients are younger and more active with fewer cardiovascular risk factors [12]. In dermatology-specific phase 3 clinical trials, venous thromboembolism rates ranged from no events to 0.1% to 0.5% compared with no events in placebo groups [7]. Cardiovascular event rates spanned from no events to 0.4% to 1.2%, similar to placebo rates of no events to 0.5% to 1.2% [7]. Most patients developing adverse events possessed baseline risk factors for the specific event [7].
Regional availability differences
JAK inhibitors gained approval in Europe, with abrocitinib marketed as Cibinqo in the United Kingdom [12]. In the United States, these inhibitors faced regulatory delays with PDUFA dates postponed and revised timelines not yet announced as of the data collection period [12].
Why choosing between treatments has become harder 
Multiple therapeutic options present clinicians with unprecedented complexity in selecting optimal treatments for atopic dermatitis. The absence of predictive biomarkers compounds this challenge, leaving treatment selection dependent on trial-and-error approaches.
No clear ‘best’ option for everyone
Treatment responses vary substantially across individuals, with no single agent demonstrating universal superiority. Atopic dermatitis represents a heterogeneous condition with multiple endotypes and phenotypes, yet current diagnostic tools cannot identify which patients will respond to specific therapies [13]. Persistence rates underscore this variability, as real-world data demonstrate treatment switching occurs frequently regardless of initial medication choice. Patients who switch treatments incur higher healthcare resource utilization, with 96.4% requiring at least one atopic dermatitis-related outpatient service compared with 82.8% of nonswitchers [1]. The annualized visit rate reaches 4.9 among switchers versus 3.1 for nonswitchers [1].
Conflicting guidance from different studies
Professional organizations issue divergent recommendations based on differing evidence evaluation methodologies. The 2023 American Academy of Allergy, Asthma and Immunology/American College of Allergy, Asthma and Immunology Joint Task Force strongly recommends dupilumab and tralokinumab but only conditionally recommends JAK inhibitors abrocitinib and baricitinib after trial of systemic treatments inclusive of a biologic [1]. This conditional recommendation stems from safety concerns regarding cardiovascular events and malignancies observed in other disease populations. In contrast, the American Academy of Dermatology moderately recommends abrocitinib and upadacitinib based on high efficacy with rapid onset demonstrated in phase 3 trials [14]. The Joint Task Force guidelines consider patient preferences for convenient and nonvisible treatments, accounting for willingness to undergo recurrent injections or self-inject [14]. Meanwhile, the American Academy of Dermatology workgroup acknowledges that costs may prove prohibitive without adequate insurance coverage [15].
Insurance and cost considerations complicate decisions
Financial barriers affect treatment access across income levels. Among patients surveyed, 21% reported inability to afford medications at some point, with this challenge not confined to the lowest income group [16]. The median out-of-pocket yearly cost reaches USD 600.00, while 42% of patients spend more than USD 1000.00 per year [9]. Approximately 8.5% of patients incur expenses exceeding USD 5000.00 annually [9]. Over-the-counter treatments and supplies constitute the most expensive aspect of atopic dermatitis care at 36%, followed by clinic visit copays at 23% and prescription copays at 17% [16]. Treatment switching elevates costs further, with switchers accumulating average atopic dermatitis-related drug costs of USD 59246.00 compared with USD 51027.00 for nonswitchers [1]. Roughly 17.6% of patients delay treatment due to cost concerns, while 13.1% forgo care altogether and 15.7% cannot fill prescriptions [9].
Patient preference factors: pills vs injections
Patients prioritize treatment attributes differently than clinicians anticipate. While efficacy remains most valued, patients demonstrate willingness to accept decreased possibility of achieving clear skin to obtain treatments that can be paused, with a maximum acceptable difference of 24.1% [6]. Oral administration once or twice daily proves preferable to subcutaneous injection every two weeks, with patients accepting a 16.6% reduction in clear skin probability for this convenience [6]. Patients without systemic therapy experience place higher value on avoiding serious adverse events than those with treatment history [10].
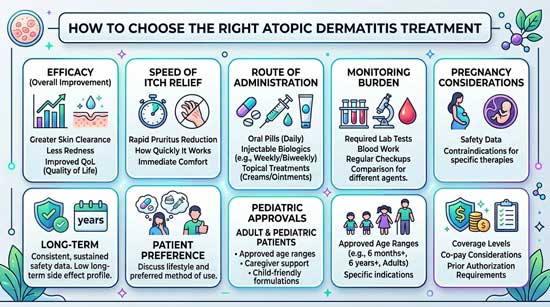
Key factors that should guide your treatment choice 
Treatment selection requires evaluation of multiple clinical parameters that extend beyond efficacy comparisons. Patient age, reproductive status, comorbidities, symptom profiles, and tolerance for risk each influence appropriate medication choice among new treatments for atopic dermatitis.
Age and pediatric considerations
Medication approvals establish distinct age thresholds that constrain pediatric treatment options. Dupilumab received FDA approval for infants and children aged 6 months and older [17][7]. In contrast, tralokinumab, lebrikizumab, and nemolizumab restrict use to patients aged 12 years and older, with lebrikizumab requiring a minimum weight of 40 kg [7]. Upadacitinib permits administration in children aged 12 years and older weighing more than 40 kg, though a black box warning addresses risks of serious cardiovascular events, cancer, blood clots, and death [17].
Topical corticosteroid selection depends on patient age and treatment location. Infants require low-potency preparations such as hydrocortisone 1% or 2.5% [17][7]. Young children excluding facial areas may use low-potency or mid-potency formulations including triamcinolone 0.025% to 0.1% [17][7]. Older children and adolescents tolerate mid-potency preparations for non-facial areas, with high-potency agents like mometasone 0.1% or fluocinonide 0.05% reserved for resistant flares [17][7].
Pregnancy and breastfeeding safety
Cyclosporine A emerged as the first-choice long-term systemic treatment for women during preconception, pregnancy, and breastfeeding periods, with short-course prednisolone managing acute flares [18]. Mild, moderate, and potent topical steroids demonstrate safety in short treatment bursts up to two weeks during pregnancy [19]. Intermittent use of mild-to-moderate potency topical corticosteroids including hydrocortisone, fluocinolone, and triamcinolone during pregnancy has not been associated with fetal adverse events [20]. However, very high cumulative doses exceeding 300 g of potent or ultrapotent corticosteroids may associate with fetal growth restriction [20].
JAK inhibitors, methotrexate, and mycophenolate mofetil require avoidance during preconception, pregnancy, and breastfeeding [18]. Patients of childbearing potential must use reliable contraception during treatment and for at least 4 weeks after stopping abrocitinib, upadacitinib, or tofacitinib, and for at least 1 week after discontinuing baricitinib [20].
Existing health conditions and risk factors
Patients face elevated atopic dermatitis risk due to condition history in biological family members, asthma diagnosis, or hay fever and other allergies [21]. Eczema itself links to poor health outcomes including reduced quality of life and increased risk of fractures, lymphoma, and cardiovascular disease [22].
Severity of itch vs skin lesions
Itch constitutes a primary treatment target, with 75% of patients reporting this symptom as “very much better” after 30 to 36 months of dupilumab treatment [8]. Disease severity assessment incorporates skin involvement extent, comorbidity presence, pruritus intensity, and effects on sleep and quality of life [23].
Speed of relief vs long-term safety
Long-term safety evidence remains limited for newer agents. Topical corticosteroids used intermittently for eczema management demonstrate safety over periods up to 5 years, with only one episode of skin atrophy reported among 1,213 patients in the topical corticosteroid arm of a five-year trial [22].
Comparison of old and new systemic treatments for moderate to severe atopic dermatitis 
Conventional systemic immunosuppressants served as standard therapy before targeted biologics and JAK inhibitors entered clinical practice. Direct comparisons between old and new systemic treatments for moderate to severe atopic dermatitis reveal distinct efficacy and safety profiles that inform contemporary treatment algorithms.
How new treatments stack up against cyclosporine and methotrexate
Cyclosporine demonstrates faster efficacy than methotrexate at standard doses. At week 8, cyclosporine 2.5 mg/kg daily achieved SCORAD 50 in 42% of patients compared with 8% receiving methotrexate 15 mg weekly [12]. Subsequently, dose escalation produced comparable outcomes, with EASI 50 reached by 92% of methotrexate patients and 87% of cyclosporine patients at week 20 [12]. However, cyclosporine caused more treatment-related adverse events [12]. Pediatric data shows methotrexate provides longer sustained efficacy, with superior o-SCORAD-50 rates at week 60 and fewer post-treatment flares [24]. Cost analysis demonstrates methotrexate savings of £489.67 per participant compared with cyclosporine [25].
Dupilumab surpasses methotrexate in patient-reported outcomes, delivering greater improvements in itch and quality of life [26].
Head-to-head trials: biologics vs JAK inhibitors
The JADE DARE trial demonstrated abrocitinib 200 mg daily outperformed dupilumab through week 26 [27]. Among dupilumab nonresponders, 77% achieved EASI-75 within 12 weeks after switching to abrocitinib [27]. Real-world studies confirm JAK inhibitors produce faster responses. At week 16, EASI75 occurred in 73.7% of JAK inhibitor patients versus 56.6% receiving biologics, while EASI90 reached 57.9% versus 36.9% [11]. Complete clearance favored JAK inhibitors at 36.8% versus 19.7% [11]. By week 24, efficacy differences attenuated, though absolute EASI scores remained lower with JAK inhibitors [11].
Real-world effectiveness and persistence rates
Dupilumab persistence reached 77.3% at 12 months with a 30-day grace period and 83.2% with 45-day allowances [28][29]. Pediatric persistence rates were 79.8% at 12 months for children and 81.9% for adolescents [30]. Concomitant systemic therapy requirements occurred in 35.07% of biologic patients versus 10.29% receiving JAK inhibitors [11].
Navigating the decision: a practical framework 
Starting with patient priorities and goals
Establishing treatment priorities requires collaborative conversations addressing itch reduction, sleep improvement, and quality-of-life enhancement [31]. Among 286 respondents, no pruritus emerged as the top treatment goal at 51.3%, followed by preventing new lesions at 16.9% and eliminating existing lesions at 12.6% [32]. Treatment characteristics prioritized high effectiveness at 38.1% and long-term safety at 29.1% overall [32]. Patients should identify one to three features most important to them from six options: itch, skin appearance, sleep disturbance, mental health, skin pain, and impact on daily life [13].
When to switch between treatment classes
Failure to achieve EASI 90, IGA 0/1, and near-complete itch control by approximately 4 months signals inadequate response [33]. Guidelines recommend considering treatment modification if agreed targets remain unmet within 3 to 6 months [13][33]. The LEVEL UP study demonstrated that nearly 70% of dupilumab nonresponders achieved EASI 75 within 4 weeks after switching to upadacitinib [33].
Working with your dermatologist on shared decision-making
Among patients surveyed, 49.6% preferred making final decisions after seriously considering physician opinions, while 69.4% reported confidence engaging in future shared decision-making [34]. Time constraints affect 38% of dermatologists, with 15% considering shared decision-making unrealistic [35].
Emerging tools and biomarkers for personalized selection
Baseline lesional skin CCL22 expression predicts clinical improvement across multiple therapies [36]. Baseline CXCL2 levels demonstrate strong predictive value for dupilumab response [36].
Conclusion 

The therapeutic expansion for atopic dermatitis presents both opportunity and complexity. While biologics and JAK inhibitors demonstrate superior efficacy compared with conventional immunosuppressants, no single agent proves optimal for every patient. As a result, treatment selection requires careful evaluation of age-specific approvals, pregnancy considerations, comorbidity profiles, and individual symptom burdens. Shared decision-making addresses the misalignment between patient priorities and clinical data presentation. Emerging biomarkers such as baseline CCL22 and CXCL2 expression may eventually guide personalized treatment selection. For now, clinicians must weigh efficacy data against safety profiles while integrating patient preferences to navigate this evolving landscape effectively.
FAQs 
Q1. What new treatment options became available for atopic dermatitis in 2026? The treatment landscape in 2026 includes several FDA-approved biologics like dupilumab, tralokinumab, lebrikizumab, and nemolizumab, which target specific inflammatory pathways. Oral JAK inhibitors such as upadacitinib, abrocitinib, and baricitinib offer convenient once-daily dosing. Additionally, new topical treatments including roflumilast cream, tapinarof cream, and topical JAK inhibitors like ruxolitinib provide nonsteroidal options for mild to moderate cases.
Q2. How does nemolizumab differ from other biologics like dupilumab? Nemolizumab works differently by targeting the IL-31 receptor alpha, which specifically addresses the itch pathway in atopic dermatitis. In contrast, dupilumab blocks both IL-4 and IL-13 signaling to reduce skin inflammation. Clinical trials showed that 68% of nemolizumab patients achieved itch relief by day 2, making it particularly beneficial for those whose primary concern is severe itching rather than skin lesions alone.
Q3. Are JAK inhibitors more effective than biologics for treating eczema? JAK inhibitors generally provide faster symptom relief, with studies showing higher clearance rates at week 16 compared to biologics. However, they carry FDA black box warnings for increased risks of cardiovascular events, blood clots, and certain cancers. While network meta-analyzes suggest upadacitinib 30 mg demonstrates superior efficacy, the choice between JAK inhibitors and biologics should consider individual risk factors, age, and tolerance for potential side effects.
Q4. What makes choosing an atopic dermatitis treatment more difficult now than before? The abundance of treatment options creates complexity because there’s no single “best” medication for everyone. Patients and doctors must weigh multiple factors including efficacy rates, side effect profiles, administration methods (pills versus injections), insurance coverage, and individual priorities. Additionally, professional organizations issue different recommendations, and without predictive biomarkers, treatment selection often involves trial-and-error approaches.
Q5. Which systemic treatments are safe during pregnancy and breastfeeding? Cyclosporine A is considered the first-choice long-term systemic treatment during preconception, pregnancy, and breastfeeding, with short-course prednisolone for acute flares. Mild to moderate topical corticosteroids used intermittently for up to two weeks are generally safe. However, JAK inhibitors, methotrexate, and mycophenolate mofetil must be avoided, and patients of childbearing potential need reliable contraception during treatment and for specified periods after stopping these medications.
References: 
[2] – https://www.ajmc.com/view/recent-fda-approvals-expand-dermatology-options-for-patients-with-skin-of-color
[3] – https://www.fda.gov/drugs/novel-drug-approvals-fda/novel-drug-approvals-2026
[4] – https://www.dermatologytimes.com/view/disease-modifying-topicals-redefine-atopic-dermatitis-management
[5] – https://pubmed.ncbi.nlm.nih.gov/41585823/
[6] – https://pmc.ncbi.nlm.nih.gov/articles/PMC9351316/
[7] – https://www.aap.org/en/patient-care/atopic-dermatitis/treatment-of-atopic-dermatitis/?srsltid=AfmBOooLyKIp7xgxkrYDr2M574PEuqKAy9xWBOz5t6cxCagITAmo4_2e
[8] – https://pubmed.ncbi.nlm.nih.gov/37552431/
[9] – https://www.healthline.com/health/atopic-dermatitis/understanding-the-expenses-of-managing-eczema
[10] – https://www.tandfonline.com/doi/full/10.1080/09546634.2024.2345739
[11] – https://www.frontiersin.org/journals/medicine/articles/10.3389/
fmed.2025.1658843/full
[12] – https://www.jaci-inpractice.org/article/S2213-2198(17)30531-7/abstract
[13] – https://www.livderm.org/articles/ahead-recommendations-for-atopic-dermatitis/
[14] – https://pmc.ncbi.nlm.nih.gov/articles/PMC12710874/
[15] – https://www.jaad.org/article/S0190-9622(25)02125-5/fulltext
[16] – https://www.dermatoljournal.com/articles/the-cost-of-relief-financial-and-educational-impacts-on-atopic-dermatitis-care.html
[17] – https://www.aap.org/en/patient-care/atopic-dermatitis/treatment-of-atopic-dermatitis/?srsltid=AfmBOor2NGZALyrJCKDebYMYCYhY4lUPDn6dZZbuKLtqm737l_QpbkR7
[18] – https://pubmed.ncbi.nlm.nih.gov/37818828/
[19] – https://eczema.org/information-and-advice/living-with-eczema/pregnancy-and-eczema/
[20] – https://www.emjreviews.com/dermatology/article/safe-medical-management-of-atopic-dermatitis-in-pregnancy-and-lactation-j030123/
[21] – https://my.clevelandclinic.org/health/diseases/24299-atopic-dermatitis
[22] – https://pmc.ncbi.nlm.nih.gov/articles/PMC10549798/
[23] – https://pmc.ncbi.nlm.nih.gov/articles/PMC11627575/
[24] – https://www.healio.com/news/dermatology/20230929/methotrexate-shows-longer-efficacy-in-pediatric-atopic-dermatitis-than-cyclosporine
[25] – https://pubmed.ncbi.nlm.nih.gov/41337631/
[26] – https://academic.oup.com/bjd/article/191/6/988/7718940
[27] – https://www.hcplive.com/view/jak-inhibitors-after-biologic-therapy-in-atopic-dermatitis-clinical-lessons-from-jade-extend-and-real-world-studies
[28] – https://www.annallergy.org/article/S1081-1206(20)30517-2/fulltext
[29] – https://www.ajmc.com/view/real-world-study-finds-high-persistence-with-dupilumab-in-atopic-dermatitis
[30] – https://pubmed.ncbi.nlm.nih.gov/41037533/
[31] – https://www.hcplive.com/view/establishing-atopic-dermatitis-treatment-goals-and-expectations-with-patients
[32] – https://www.dermatologyadvisor.com/news/atopic-dermatitis-treatment-goals-preferences-differ-age-young-patients/
[33] – https://www.hcplive.com/view/special-report-inadequate-response-and-when-to-switch-systemic-therapy-in-atopic-dermatitis
[34] – https://jamanetwork.com/journals/jamadermatology/fullarticle/2793785
[35] – https://pmc.ncbi.nlm.nih.gov/articles/PMC7818257/
[36] – https://www.jacionline.org/article/S0091-6749(23)00143-4/fulltext
Video Section
Check out our extensive video library (see channel for our latest videos)
Recent Articles


