Fibromyalgia and Lupus Together Why Composite Disease Scores May Mislead Clinicians

Key Takeaways
Accurate differentiation between fibromyalgia and systemic lupus erythematosus remains one of the most important challenges in rheumatologic practice, particularly because both conditions frequently coexist and share several subjective symptoms that can complicate clinical interpretation. In patients with systemic lupus erythematosus, fibromyalgia occurs far more commonly than in the general population, with pooled prevalence estimates ranging from approximately 15.8 percent to 22.1 percent. This means that nearly one in six patients with lupus may also meet criteria for fibromyalgia, creating a clinically significant overlap that directly affects disease assessment, treatment decisions, and long term outcomes.
The difficulty arises because many of the most prominent symptoms reported by patients, including fatigue, diffuse pain, morning stiffness, poor sleep, and cognitive dysfunction, are common to both disorders. Although these manifestations may appear similar at the bedside, their underlying mechanisms differ substantially. In lupus, such symptoms may reflect active immune mediated inflammation, vascular involvement, or organ specific disease activity. In fibromyalgia, however, they are generally linked to altered central pain processing, sleep dysregulation, and neurobiological amplification of pain rather than peripheral inflammation. Failure to recognize this distinction can lead to major therapeutic consequences.
Traditional lupus disease activity scoring systems were developed to quantify inflammatory burden and guide treatment intensity. Instruments such as the Systemic Lupus Activity Questionnaire and the Systemic Lupus Erythematosus Disease Activity Index remain widely used in both research and clinical care. However, these composite measures often rely heavily on patient reported symptoms that lack specificity for inflammatory disease. As a result, fibromyalgia related complaints may inflate lupus activity scores even in the absence of immunological or clinical evidence of active lupus. This creates a risk of misclassifying non inflammatory symptom burden as disease flare, prompting unnecessary escalation of immunosuppressive therapy.
This distinction is particularly important because fibromyalgia does not respond to corticosteroids, immunosuppressive agents, or biologic therapies used to treat lupus inflammation. Escalating lupus treatment in a patient whose symptoms are primarily driven by fibromyalgia not only fails to improve outcomes but also exposes the patient to avoidable adverse effects, including infection risk, metabolic complications, osteoporosis, and long term organ toxicity.
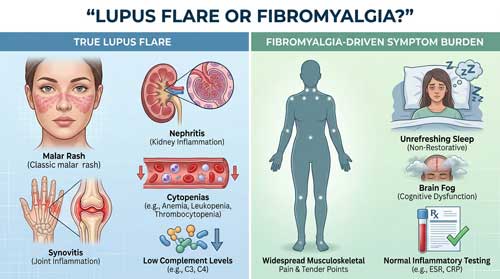
True lupus activity is more reliably suggested by specific objective clinical and laboratory features. Findings such as Raynaud’s phenomenon, malar rash, unexplained fever, easy bruising, alopecia, oral ulcers, serositis, synovitis, nephritis, and hematologic abnormalities point more strongly toward active inflammatory disease. Laboratory evidence including elevated anti double stranded DNA antibodies, reduced complement levels, proteinuria, cytopenias, and inflammatory markers may further support active lupus, although these too must be interpreted carefully in clinical context.
By contrast, fibromyalgia is characterized by widespread musculoskeletal pain that is non inflammatory in nature, often accompanied by unrefreshing sleep, persistent fatigue, cognitive complaints, and morning stiffness without objective evidence of synovitis or organ inflammation. Blood tests in fibromyalgia are typically normal, and inflammatory markers usually remain within expected limits unless another condition is present. The absence of objective inflammatory findings should prompt clinicians to consider fibromyalgia, especially when pain severity appears disproportionate to measurable lupus activity.
Recent research has emphasized the limitations of conventional composite scoring systems in this overlap population and has highlighted the value of more refined assessment tools. Patient Reported Outcomes Measurement Information System instruments provide a broader evaluation of fatigue, pain interference, sleep quality, and physical function, allowing clinicians to characterize symptom burden more precisely. The Symptom Intensity Scale has also demonstrated value in identifying symptom patterns more consistent with fibromyalgia than inflammatory disease.
In addition, emerging investigations into cytokine profiles and other biomarker based approaches suggest that immunological signatures may eventually help distinguish central pain syndromes from active autoimmune inflammation. Although these methods are not yet routinely incorporated into standard rheumatology practice, they represent a promising direction for improving diagnostic precision.
A careful clinical interview remains central to differentiation. Patients with fibromyalgia often describe persistent generalized pain that fluctuates with stress, sleep disruption, physical exertion, or emotional burden, whereas lupus flares are more likely to involve new objective findings or measurable laboratory changes. Sleep history is especially important, since disturbed sleep and non restorative rest strongly support fibromyalgia as a major contributor to symptom burden.
When both conditions coexist, clinicians must resist relying solely on elevated composite lupus scores. Instead, a multidimensional assessment that integrates objective examination findings, laboratory trends, validated symptom measures, and longitudinal disease history is required. This approach allows true inflammatory activity to be identified while also recognizing fibromyalgia as a separate but clinically important contributor to reduced quality of life.
Management strategies should reflect this distinction. Active lupus requires immunologically targeted treatment tailored to organ involvement and disease severity. Fibromyalgia management, by contrast, is centered on patient education, graded physical activity, sleep optimization, cognitive behavioral strategies, and selected pharmacologic therapies such as neuromodulators when indicated. Treating fibromyalgia appropriately often improves symptom burden more effectively than increasing lupus medications.
Ultimately, distinguishing fibromyalgia from lupus activity is essential for preventing overtreatment, reducing medication related harm, and improving patient centered outcomes. As understanding of symptom overlap evolves, clinicians must adopt more nuanced diagnostic frameworks that move beyond conventional scoring systems and prioritize precise interpretation of both inflammatory and non inflammatory symptom patterns.
The Overlap Between Fibromyalgia and Lupus
How Common is Fibromyalgia in Lupus Patients
The coexistence of fibromyalgia and lupus occurs at rates far exceeding what chance alone would predict. A systematic review analyzing 56 studies with 58,052 SLE patients detected fibromyalgia in 5,063 individuals, yielding an overall pooled prevalence of 15.8% (95% CI, 13.4-18.5) [1]. Patients with SLE demonstrated a 3.7-fold increased risk of developing fibromyalgia compared to healthy controls [1]. Individual cohort studies reveal even higher rates, with Johns Hopkins reporting 30% of their lupus cohort meeting fibromyalgia criteria [2], while a Brazilian tertiary care center documented 19.1% prevalence among 107 SLE patients [3].
Gender patterns emerge distinctly in this overlap. Among Brazilian patients diagnosed with both conditions, 100% were female, contrasting with the non-fibromyalgia lupus group where 94.1% were female and 5.9% male [3]. This female predominance affects risk assessment, as women represent 90% of lupus diagnoses and face twice the fibromyalgia risk compared to men [3].
The variability across studies reflects genuine population differences rather than measurement error alone. Indian tertiary centers reported 8.2% prevalence among 158 patients, while Mexican clinics documented 9.5% in 106 patients [3]. At the opposite extreme, some investigators report up to 65% of SLE patients exhibit fibromyalgia features [3][2]. Kenyan research screening 60 females with lupus found 65% met fibromyalgia criteria [2]. Geographic, genetic, and healthcare access factors likely contribute to these disparities.
Shared Symptoms That Confuse Diagnosis
Chronic fatigue manifests in both fibromyalgia and lupus, yet stems from distinct physiological processes. In lupus, exhaustion results from inflammation and immune overactivity, whereas fibromyalgia fatigue originates from sleep disturbances and heightened pain sensitivity [4]. Among lupus patients, fatigue correlates more strongly with fibromyalgia presence than with measured lupus disease activity [2].
Muscle and joint pain create diagnostic ambiguity because lupus patients experience joint inflammation and swelling while fibromyalgia produces widespread pain involving tender points across the body [4]. Both conditions trigger cognitive dysfunction, manifesting as memory difficulties, impaired focus, and concentration problems [4]. Sleep disturbances affect patients with either diagnosis, perpetuating cycles of fatigue and pain [4].
Depression emerges as a particularly confounding factor. SLE patients with comorbid fibromyalgia demonstrated depression rates exceeding 65% using PHQ-9 criteria, compared to approximately 18% in those without fibromyalgia [3]. This represents more than three times the depression prevalence [3]. Using SLAQ criteria, 33% of fibromyalgia patients reported mild depression while 31% experienced moderate to severe depression, versus 20% and 13% respectively among those without fibromyalgia [3].
Why Both Conditions Can Occur Together
Fibromyalgia may develop as a secondary condition in lupus patients through specific pathophysiological mechanisms. Chronic pain and immune dysregulation in lupus can alter how the brain processes pain signals, potentially triggering fibromyalgia [4]. Shared risk factors extend beyond gender to include genetic predisposition, with lupus carrying a 5% to 13% familial risk and fibromyalgia showing eight-fold increased risk among first-degree relatives [3].
Environmental triggers overlap between conditions. Epstein-Barr virus infections and psychological stress activate both lupus and fibromyalgia [3]. Stress and poor sleep quality precipitate flares in either condition, while lupus patients face additional triggers including illness, injury, and sun exposure [3].
The presence of fibromyalgia acts as a confounding factor when assessing lupus disease activity and treatment response [1]. Patients experiencing both conditions report lower quality of life, cognitive dysfunction, and increased opioid usage compared to those with lupus alone [1]. An intriguing inverse relationship emerges with lupus nephritis: only 10.5% of nephritis patients had fibromyalgia versus 23% without nephritis [3]. Patients with both active and inactive nephritis demonstrated lower rates of fibromyalgia, fatigue, sleep dysfunction, and cognitive impairment [3].
How Composite Disease Scores Work 
Clinicians treating patients with fibromyalgia and lupus rely on validated composite indices to quantify disease activity and guide treatment decisions. These scoring systems aggregate multiple clinical features and laboratory values into single numerical scores. Three primary instruments dominate clinical practice and research: the Systemic Lupus Activity Questionnaire (SLAQ), the Systemic Lupus Activity Measure (SLAM), and the Systemic Lupus Erythematosus Disease Activity Index (SLEDAI). Each tool approaches assessment differently, yet all share a fundamental limitation when fibromyalgia and lupus occur together.
SLAQ and SLAM Scoring Systems
The SLAQ represents the only validated patient self-report instrument for SLE disease activity [3]. Developed from items in the SLAM that patients could report independently, the questionnaire includes 24 questions covering disease activity across multiple domains: weight loss, fatigue, fevers, oral ulcers, malar rash, photosensitivity, vasculitis, other rashes, alopecia, lymphadenopathy, dyspnea, chest pain, Raynaud’s phenomenon, abdominal pain, paresthesias, seizures, stroke, memory loss, depression, headaches, myalgias, muscle weakness, arthralgias, and joint swelling [3][3].
The SLAQ employs four distinct scoring methods. The primary SLAQ score weights and aggregates the 24 symptom questions analogously to SLAM scoring, producing a range of 0 to 44 points [3][3]. The Symptom Score counts positive responses within the 24 questions, ranging from 0 to 24 [3]. A patient global assessment rates lupus flare presence and severity from 0 to 3, while a numerical rating scale asks patients to rate disease activity from 0 (no activity) to 10 (most activity) [3]. A cutoff of 9 or higher demonstrates 83% sensitivity and 70% specificity for predicting clinically meaningful disease activity [3]. Validation studies showed SLAQ scores correlated with the SLAM at r = 0.62 [3][5], with positive predictive values ranging from 56% to 89% for detecting clinically meaningful disease activity [3][3].
In contrast, the SLAM requires physician completion based on history, physical examination, and laboratory evaluation [3]. The instrument evaluates manifestations across 9 organ systems plus 7 laboratory features [4][6]. Organ items receive scores of 0 to 3 points based on severity, while the laboratory category can score up to 21 points [4]. Total scores range from 0 to 81 or 83 depending on the version [4][2]. A SLAM score exceeding 6 or 7 is considered clinically important and influences treatment initiation decisions in more than 50% of cases [4][2]. The correlation between SLAM-no lab and patient-reported Q-SLAQ scores reached 0.71 [5][2].
SLEDAI Activity Index Components
The SLEDAI and its successor SLEDAI-2K measure disease activity through 24 items covering 9 organ systems: neurological, musculoskeletal, renal, mucocutaneous, general, cardiovascular, respiratory, vascular, and hematological [4][6]. The assessment includes 16 clinical descriptors and 8 laboratory items [2]. Scores range from 0 to 105 points, with different organ systems weighted according to severity [4][7]. Two systems score a maximum of 8 points each, two systems score 4 points maximum, three systems reach 2 points maximum, and two systems score 1 point maximum [4].
The original SLEDAI counted variables like proteinuria, rash, alopecia, and mucous membrane lesions as active only at first occurrence or upon recurrence [8]. SLEDAI-2K modified these definitions to include persistent activity in these descriptors, not merely new or recurrent manifestations [8]. This modification allows proteinuria to count as active when new, recurrent, or persistently exceeding 0.5 g per 24 hours [8]. SLEDAI-2K correlated highly with the original SLEDAI at r = 0.97 [8]. Both versions predicted mortality equally [8]. The recall period spans the 10 days preceding assessment [4], though recent validation showed similarity with a 30-day timeframe [4].
What These Scores Are Supposed to Measure
These composite indices aim to quantify true inflammatory disease activity from lupus itself. A SLEDAI score of 3 to 4 defines active disease requiring treatment escalation [7]. For instance, ultrasonography studies of joint inflammation in SLE patients demonstrated positive correlations between inflammatory scores and composite indices: DAS28-CRP showed r = 0.3, while the swollen-to-tender joint ratio reached r = 0.42 [3][3].
The instruments demonstrate varying responsiveness depending on disease activity levels. SLAM showed higher correlation with patient assessments in low disease activity groups, whereas SLEDAI-2K performed better in high disease activity groups [5][2]. This differential performance matters when fibromyalgia and lupus symptoms overlap, as fatigue, depression, headaches, myalgias, muscle weakness, and abdominal pain appear in both SLAQ and SLEDAI scoring systems despite potentially originating from fibromyalgia rather than active lupus inflammation.
The Problem: When Fibromyalgia Symptoms Inflate Lupus Scores 
Measurement discordance between patient-reported outcomes and physician assessments reveals the fundamental flaw in composite disease activity indices when fibromyalgia and lupus occur together. Patients with both conditions consistently report higher disease activity scores than objective clinical measures support, creating diagnostic confusion and potential treatment errors.
Fatigue and Pain Overlap in Both Conditions
Patient Global Assessment scores, SLAQ scores, and PHQ-9 depression scores reached at least twice the values in patients with inactive SLE and fibromyalgia compared to those with inactive SLE alone (P < .0001) [4]. This pattern persisted across activity levels. Among patients with active SLE and fibromyalgia, 100% self-reported flares versus 64% of those without fibromyalgia (P = .0001) [4]. The correlation between fibromyalgianess measured by the Symptom Intensity Scale and SLE activity measured by SLAQ reached 0.676 [6]. This strong association demonstrates how fibromyalgia symptoms systematically inflate lupus activity measurements.
In reality, these elevated scores often reflect Type 2 symptoms rather than inflammatory disease activity. Type 2 symptoms include fatigue, brain fog, and mood disturbance, which have marked impact on quality of life but remain discordant with inflammatory symptoms and respond poorly to immunosuppression [9]. Research categorizing SLE patients into fibromyalgia chronic pain, non-fibromyalgia chronic pain, and no chronic pain groups found that Type 2 symptoms existed across all chronic pain groups regardless of fibromyalgia diagnosis [6]. Specifically, 13% of patients with fibromyalgia were in lupus remission despite reporting severe symptoms [6].
Cognitive Problems and Depression Effects
Cognitive dysfunction in lupus presents as deficits in attention, working memory, psychomotor speed, and executive function [10]. Depression, fatigue, and pain have been associated with cognitive dysfunction in lupus patients [10]. Studies found that 40% to 50% of SLE patients demonstrate measurable cognitive impairment on formal testing [11]. Equally problematic, cognitive dysfunction was vastly under-detected in clinical practice, with only 4 of 49 patients with objective cognitive dysfunction recognized by clinicians on SDI scoring [11].
The overlap between cognitive symptoms and psychological disorders complicates assessment further. Anxiety and depression scores consistently related to a cognitive dimension incorporating memory, executive function, and complex processing speed [8]. Many patients with cognitive dysfunction exhibit clinically significant anxiety and depression [8]. Given that fibromyalgia patients report higher rates of cognitive dysfunction, depression, and anxiety [4], composite scores capturing these symptoms misattribute fibromyalgia features to active lupus.
Muscle Weakness vs Inflammatory Symptoms
SLE-related symptoms including muscle weakness were reported more frequently in patients with fibromyalgia [4]. Meanwhile, self-reported fibromyalgia symptoms of muscle pain, joint stiffness, fatigue, and poor sleep remained similar regardless of SLE activity level (P = .05 for all symptoms) [4]. This dissociation between symptom reporting and lupus activity creates scoring inflation. Patients with SLE and fibromyalgia demonstrated higher rates of functional symptoms including headache, numbness, and stomach pain [4].
Why High Scores Don’t Always Mean Active Lupus
The presence or absence of fibromyalgia did not result in any differences in physician assessment of disease activity in SLE [4]. Patients with SLE and fibromyalgia proved overall similar to those without fibromyalgia and did not differ in serologies or overall SLEDAI scores [7]. Symptom severity scores and widespread pain index did not correlate with SLEDAI scores [7]. In patients without fibromyalgia, solid correlation existed between patient and physician measures [4]. This discordance suggests that physicians may not be aware of the severity of symptoms this subgroup of patients experience [4], leading to either inappropriate treatment intensification or inadequate symptom management.
Which Symptoms Are Actually From Lupus 
Distinguishing true lupus activity from fibromyalgia symptoms requires recognition of inflammatory markers and organ-specific manifestations that rarely occur in fibromyalgia alone. When evaluating patients with fibromyalgia and lupus together, clinicians must identify features that point specifically to active SLE rather than widespread pain syndromes.
Raynaud’s Phenomenon and Rash Patterns
Raynaud’s phenomenon affects approximately one-third of lupus patients [12][3], with prevalence ranging from 18% to 49% across different cohorts [13]. This vasospastic condition restricts blood flow to extremities, causing sequential color changes from white to blue to red in response to cold temperatures or emotional stress [14][13]. The digits turn pale from lack of blood flow, then blue from oxygen deprivation, followed by redness as circulation resumes [12]. Unlike primary Raynaud’s phenomenon, lupus-related Raynaud’s typically involves thumbs and results from inflammation of nerves or blood vessels [12][14].
The characteristic butterfly rash, spanning the width of the face from cheek to cheek across the nasal bridge, appears in approximately 50% of lupus patients [5][3]. This malar rash distinguishes itself by sparing the nasolabial folds, the spaces directly beneath each side of the nose [5]. The rash may manifest spontaneously or following sun exposure, with some patients recognizing its appearance as a harbinger of disease flares [5].
Discoid lupus rashes present as raised, red, scaly patches typically appearing on the face and scalp [3][2]. These lesions carry risk for permanent scarring and skin discoloration when healing occurs [3][2]. Subacute cutaneous lupus erythematosus (SCLE) develops rapidly on sun-exposed areas including arms, shoulders, chest, and neck, forming interlocking circular patterns without causing pain or itching [2]. Acute cutaneous lupus erythematosus (ACLE) manifests during active disease flares with bumps, discoloration, scales, and burning sensations [2].
Fever and Easy Bruising as Lupus Markers
Fever represents a prevalent lupus manifestation, occurring in 36% to 86% of SLE patients [15][16]. In certain circumstances, fever serves as the sole indication of lupus, particularly in individuals with fever of undetermined origin [15]. The Modified SLEDAI incorporates fever as a disease activity scoring component [16]. Despite these figures, differentiating lupus flares from infections presents a persistent diagnostic challenge, as both conditions produce similar initial clinical presentations during acute febrile phases [15][16].
Purpura develops in approximately 15% of lupus patients during the disease course [3]. These small red or purple discolorations result from blood vessel leakage beneath the skin [3]. Petechiae describe small purpura spots, while ecchymoses refer to larger discolorations [3]. Easy bruising patterns may indicate insufficient platelet levels, medication effects, or other hematological complications [3][4]. Teenagers diagnosed initially with isolated low platelet counts and easy bruising sometimes receive lupus diagnoses months or years later when additional manifestations emerge, suggesting thrombocytopenia represented early lupus signs [4].
Hair Loss Specific to Lupus Activity
Hair loss occurs in 70% to 85% of SLE patients at some point during disease progression [3][7]. Non-scarring alopecia achieved inclusion in the Systemic Lupus International Collaborating Clinics (SLICC) classification criteria based on 95.7% specificity to SLE [7][6]. This form of hair loss manifests abruptly, predominantly in patients with severe, uncontrolled lupus, and correlates with underlying disease activity including proteinuria exceeding 1 g per day [7].
Research demonstrates that certain trichoscopic features correlate with disease severity [7]. Patients exhibiting hair shaft changes including decreased hair number, decreased diameter, and hypopigmentation demonstrated more active systemic disease with higher mean SLEDAI scores than those without these features [7]. Non-scarring alopecia assumes diffuse, patchy, or “lupus hair” patterns [7]. The patchy pattern shows erythema and reduced hair density rather than complete hair loss, distinguishing it from alopecia areata [7].
Lupus hair refers specifically to frontal hairline alopecia with dry, coarse appearance due to increased hair fragility [7][17]. This striking sign results from severe inflammation promoting a catabolic state and negative nitrogen balance that impairs hair growth [7]. Diffuse non-scarring alopecias generally reflect underlying disease activity or severity, improving once acute lupus flares subside [6].

Which Symptoms Point More to Fibromyalgia 
Certain clinical features point more reliably toward fibromyalgia than lupus when evaluating patients with fibromyalgia and lupus together. These distinguishing characteristics assist clinicians in attributing symptoms correctly rather than misinterpreting fibromyalgia manifestations as active lupus requiring immunosuppression.
Widespread Body Pain Without Inflammation
Physical examination of fibromyalgia patients reveals joints that move normally without swelling [18]. In contrast, pressing over muscles causes discomfort at tender areas [18]. Fibromyalgia diagnosis requires widespread pain lasting at least three months [19] [20], affecting at least four of five body regions: left upper, right upper, left lower, right lower, or axial areas [10] [11]. The pain manifests as chronic multisite musculoskeletal discomfort [8], often described as constant dull ache, burning sensation, or sharp stabbing [21].
Laboratory testing provides critical differentiation. In fibromyalgia patients, all blood tests typically return normal results [18]. Inflammatory markers likewise show normal values [8]. Owing to this pattern, scenarios suggesting fibromyalgia rather than lupus include symptoms unresponsive to corticosteroids, widespread pain despite successful lupus control, and absence of previously positive lupus laboratory tests [8].
Headaches and Abdominal Pain Patterns
Headaches affect more than half of fibromyalgia patients, with migraine prevalence ranging from 36% to 80% depending on the study [22] [23]. Migraine represents a common symptom in fibromyalgia [24], often presenting with sharp pulsing pain, photophobia, and nausea [25]. The prodrome phase produces irritability and mood changes, while the postdrome phase triggers extreme fatigue and confusion [23].
Tension-type headaches produce dull, pressure-like sensations [23], typically worsening throughout the day when patients feel tired, stressed, or hungry [23]. Abdominal pain patterns likewise point toward fibromyalgia, with irritable bowel syndrome occurring frequently [24] [20]. Digestive and bladder problems represent associated fibromyalgia symptoms [19] [20].
Sleep Disturbance and Morning Stiffness
Sleep problems constitute a hallmark of fibromyalgia [26], with disordered sleep experienced by upwards of 90% of patients [27]. Non-restorative sleep ranks as one of the main fibromyalgia symptoms [28]. Patients wake feeling tired despite lengthy sleep duration [24] [18], describing their sleep as shallow, disrupted, or unrefreshing [27]. Insomnia and restless legs syndrome link closely with fibromyalgia [28] [24].
Polysomnographic studies demonstrate wakefulness during non-rapid eye movement stages, resulting in less slow-wave sleep [27]. Sleep deprivation in healthy individuals can produce fibromyalgia-like symptoms including myalgia and fatigue [27]. Morning stiffness appears most severe after prolonged immobility, particularly upon first awakening [21] [26].
Better Ways to Separate Fibromyalgia from Lupus Activity 
Advanced diagnostic strategies offer clinicians objective methods for separating fibromyalgia from lupus activity when evaluating patients with both conditions. These evidence-based approaches move beyond traditional composite scores to provide more accurate disease characterization.
Using PROMIS Patient-Reported Measures
Patient-Reported Outcomes Measurement Information System (PROMIS) measures demonstrate superior discriminatory capability compared to standard fibromyalgia criteria. Mean PROMIS scores for fatigue, sleep disturbance, cognitive function, and depression differed across pain groups (p< 0.001), with pain groups distinct from no-pain groups (p< 0.01) [29]. Four PROMIS measures including cognitive function, fatigue, pain intensity, and pain interference showed differences between fibromyalgia pain and non-fibromyalgia pain groups (p< 0.02) [29]. This disease-independent measurement tool allows exploration and definition of patient subtypes across a wider array of measures than fibromyalgia criteria surveys [30].
Cytokine Testing to Detect Real Inflammation
Serum cytokine profiling detects genuine lupus inflammation absent in fibromyalgia. Among 12 cytokines tested, IL8, MIP1α, MIP1β, and anti-C1q levels elevated in active SLE compared with inactive disease [31]. In receiver operating characteristic analysis, MIP1α alone showed diagnostic utility (AUC = 0.717), while adding IL8 increased the AUC to 0.803 [31]. Combining IL8, MIP1β, and anti-C1q antibodies achieved AUC of 0.976 [31].
Cell-bound complement activation products testing provides complete specificity for lupus versus fibromyalgia. The CB-CAPs in multi-analyte assay with algorithm yielded 60% sensitivity for SLE while producing negative results for every fibromyalgia patient tested, rendering 100% specificity [32].
The Symptom Intensity Scale Approach
The Symptom Intensity Scale measures fibromyalgianess as a continuous latent variable, avoiding the dichotomizing problem created by fibromyalgia diagnosis [9]. The SI score increased minimally in SLE compared with arthritis (0.4 units) but substantially in fibromyalgia (2.2 units) [9].
Combining Multiple Assessment Tools
The Fibromyalgia Assessment Screening Tool (FAST3) composite measure evaluates pain (≥6/10), joint count (≥16/48), and symptom checklist (≥16/60), correctly classifying 89% of patients (AUC=0.90, kappa=0.71) [33]. In active disease presence, FAST3 accuracy reached 91% [33]. Communication among primary care doctors and rheumatologists facilitates agreed-upon diagnosis [8]. Symptoms unresponsive to corticosteroids or widespread pain despite successful lupus control suggest fibromyalgia rather than lupus [8].
Conclusion 

Patients with fibromyalgia and lupus together present a critical diagnostic dilemma that standard composite disease activity scores fail to resolve. Traditional instruments like SLAQ and SLEDAI systematically misattribute fibromyalgia symptoms to active lupus inflammation, leading clinicians toward inappropriate immunosuppressive treatment escalation. Fatigue, cognitive dysfunction, and widespread pain elevate these scores despite absent inflammatory markers. Evidently, physicians require objective assessment tools that distinguish Type 2 symptoms from genuine lupus activity. PROMIS measures, cytokine profiling, and the Symptom Intensity Scale offer superior discriminatory capability. Accurate differentiation between these overlapping conditions ultimately determines whether patients receive targeted fibromyalgia management or unnecessary immunosuppression with attendant risks.
FAQs 
Q1. How can doctors tell the difference between fibromyalgia and lupus symptoms? During a physical examination, fibromyalgia patients typically have tender areas over muscles that cause discomfort when pressed, but their joints move normally without swelling. In contrast, lupus patients may show joint pain, stiffness, swelling, or abnormal movement. Additionally, fibromyalgia patients usually have normal blood test results and inflammatory markers, while lupus often shows abnormal laboratory values.
Q2. How common is it to have both fibromyalgia and lupus at the same time? Fibromyalgia occurs in approximately 15.8% to 22.1% of patients with systemic lupus erythematosus (SLE), with some studies reporting rates as high as 30-65%. Patients with lupus have a 3.7-fold increased risk of developing fibromyalgia compared to healthy individuals, making this overlap significantly more common than would occur by chance alone.
Q3. Why do lupus disease activity scores sometimes give misleading results? Standard lupus scoring systems like SLAQ and SLEDAI include symptoms such as fatigue, pain, cognitive problems, and depression that appear in both lupus and fibromyalgia. When patients have both conditions, these scores often capture fibromyalgia symptoms instead of true lupus inflammation, leading to artificially elevated scores that don’t accurately reflect actual lupus disease activity.
Q4. What symptoms are more specific to lupus rather than fibromyalgia? Symptoms that point specifically to lupus include the butterfly-shaped malar rash across the cheeks, Raynaud’s phenomenon (color changes in fingers and toes), fever, easy bruising, and hair loss that correlates with disease activity. These manifestations involve inflammation and organ-specific changes that rarely occur in fibromyalgia alone.
Q5. What are better ways to distinguish fibromyalgia from active lupus? More accurate assessment methods include PROMIS patient-reported measures, cytokine testing to detect actual inflammation, and the Symptom Intensity Scale. Cytokine profiling can achieve up to 97.6% accuracy in detecting true lupus inflammation, while PROMIS measures effectively differentiate between pain types. Combining multiple assessment tools provides the most reliable diagnosis.
References: 
[2] – https://my.clevelandclinic.org/health/symptoms/23163-lupus-rash
[3] – https://www.hopkinslupus.org/lupus-info/lupus-affects-body/skin-lupus/
[4] – https://creakyjoints.org/about-arthritis/lupus/lupus-symptoms/lupus-signs-and-symptoms/
[5] – https://www.lupus.org/resources/is-a-butterfly-rash-definite-indicator-of-lupus
[6] – https://lupus.bmj.com/content/5/1/e000291
[7] – https://pmc.ncbi.nlm.nih.gov/articles/PMC8020452/
[8] – https://www.hss.edu/health-library/conditions-and-treatments/lupus-and-fibromyalgia
[9] – https://pmc.ncbi.nlm.nih.gov/articles/PMC2944223/
[10] – https://www.mayoclinic.org/diseases-conditions/fibromyalgia/diagnosis-treatment/drc-20354785
[11] – https://www.arthritis.org/diseases/more-about/fibromyalgia-tests
[12] – https://www.healthline.com/health/lupus/lupus-raynauds-syndrome
[13] – https://pmc.ncbi.nlm.nih.gov/articles/PMC4689071/
[14] – https://www.lupus.org/lonestar/resources/about-raynauds-disease
[15] – https://pmc.ncbi.nlm.nih.gov/articles/PMC11323654/
[16] – https://europepmc.org/article/pmc/5866120
[17] – https://www.verywellhealth.com/hair-loss-and-lupus-alopecia-symptoms-and-facts-2249993
[18] – https://www.arthritis.org/diseases/more-about/fibromyalgia,-lupus,-or-both
[19] – https://www.moregooddays.com/post/fibromyalgia-vs-lupus
[20] – https://www.mylupusteam.com/resources/lupus-vs-fibromyalgia-differences-similarities-and-when-they-overlap
[21] – https://www.nhs.uk/conditions/fibromyalgia/symptoms/
[22] – https://pmc.ncbi.nlm.nih.gov/articles/PMC6500182/
[23] – https://www.healthrising.org/blog/2019/03/17/migraines-headaches-fibromyalgia-chronic-fatigue/
[24] – https://www.mayoclinic.org/diseases-conditions/fibromyalgia/symptoms-causes/syc-20354780
[25] – https://coloradopaincare.com/blog-fibromyalgia-chronic-headache-causes-symptoms-treatments/
[26] – https://www.bannerhealth.com/services/pain-management/fibromyalgia/symptoms
[27] – https://www.rheumatologyadvisor.com/features/managing-fibromyalgia-and-sleep-disorders/
[28] – https://www.sleepfoundation.org/physical-health/fibromyalgia-and-sleep
[29] – https://acrabstracts.org/abstract/patterns-of-patient-reported-outcome-measures-in-patients-with-systemic-lupus-erythematosus-with-or-without-concurrent-fibromyalgia/
[30] – https://pmc.ncbi.nlm.nih.gov/articles/PMC12872186/
[31] – https://pmc.ncbi.nlm.nih.gov/articles/PMC7271280/
[32] – https://lupusnewstoday.com/news/systemic-lupus-erythematosus-and-primary-fibromyalgia-can-be-distinguished-by-testing-for-cell-bound-complement-activation-products/
[33] – https://pubmed.ncbi.nlm.nih.gov/32741302/
Video Section
Check out our extensive video library (see channel for our latest videos)
Recent Articles


