Genitourinary Syndrome of Menopause Why Are So Many Patients Still Suffering

Abstract
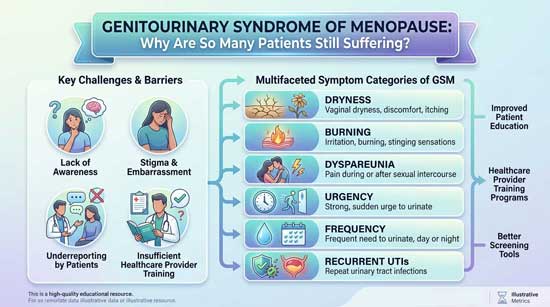
Genitourinary syndrome of menopause (GSM) is a highly prevalent yet frequently underrecognized condition that affects up to 84 percent of postmenopausal women. Previously referred to as vulvovaginal atrophy, GSM encompasses a spectrum of chronic symptoms involving the vulva, vagina, and lower urinary tract that arise primarily from estrogen deficiency. Despite its significant impact on quality of life, sexual function, and urinary health, GSM remains underdiagnosed and undertreated in routine clinical practice.
The pathophysiology of GSM is driven by the decline in circulating estrogen levels that occurs during menopause. Estrogen plays a central role in maintaining the structural integrity, vascularization, elasticity, and lubrication of urogenital tissues. In its absence, the vaginal epithelium becomes thinner, less elastic, and more fragile, with reduced glycogen content and diminished lactobacillus colonization. These changes result in an elevated vaginal pH, increased susceptibility to irritation and infection, and impaired mucosal defense. In addition to vaginal changes, estrogen deficiency affects the urethra and bladder, contributing to urinary urgency, frequency, dysuria, and recurrent urinary tract infections.
Clinically, GSM presents with a broad range of symptoms that may vary in severity and progression. Common vaginal symptoms include dryness, burning, itching, and dyspareunia, while urinary manifestations often include urgency, frequency, nocturia, and recurrent infections. Importantly, GSM is a chronic and progressive condition that does not resolve spontaneously and may worsen over time without intervention. Unlike vasomotor symptoms of menopause, which often diminish with time, genitourinary symptoms tend to persist and markedly impair physical comfort, sexual health, and psychosocial well being.
Accurate diagnosis relies primarily on clinical assessment, including a thorough history and focused physical examination. Patients may not voluntarily report symptoms due to embarrassment, misconceptions about normal aging, or lack of awareness that effective treatments exist. As a result, proactive inquiry by clinicians is essential. Physical examination findings may include pale, dry vaginal mucosa, decreased elasticity, loss of rugal folds, and signs of tissue fragility such as petechiae or fissures. While laboratory testing is not routinely required, assessment of vaginal pH and exclusion of infectious etiologies may support diagnosis in selected cases.
Despite the availability of effective therapies, several barriers contribute to suboptimal management. Physician related factors include limited time during consultations, discomfort discussing sexual health, and insufficient training in menopause related conditions. Patient related barriers include reluctance to initiate discussions, cultural stigmas, and concerns about the safety of hormonal therapies. In addition, variability in clinical guidelines and lack of standardized screening protocols further contribute to inconsistent care.
A range of treatment options is available, allowing for individualized management based on symptom severity, patient preference, and contraindications. First line therapy for mild symptoms typically includes non hormonal approaches such as vaginal moisturizers and lubricants, which provide symptomatic relief but do not reverse underlying atrophic changes. For moderate to severe symptoms, local vaginal estrogen therapy remains the most effective treatment, with strong evidence supporting its ability to restore vaginal epithelium, normalize pH, and improve both vaginal and urinary symptoms. Available formulations include creams, tablets, and rings, all of which deliver low systemic absorption and have favorable safety profiles for most patients.
Additional therapeutic options include selective estrogen receptor modulators such as ospemifene, which provide systemic benefits for dyspareunia, and intravaginal dehydroepiandrosterone, which acts locally to improve tissue health. Energy based therapies, including laser and radiofrequency devices, have emerged as potential alternatives, although evidence regarding their long term efficacy and safety remains limited and requires further investigation.
Improving outcomes in GSM requires a multifaceted approach that addresses both clinical and systemic barriers. Routine screening for genitourinary symptoms should be integrated into menopause care, with clinicians adopting a proactive and patient centered communication style. Education is essential to increase awareness among both healthcare providers and patients regarding the chronic nature of GSM and the availability of safe and effective treatments. Standardized clinical pathways and guideline based management can further enhance consistency of care.
In conclusion, genitourinary syndrome of menopause represents a common and impactful condition that remains insufficiently addressed in clinical practice. With appropriate recognition, timely diagnosis, and individualized treatment, it is possible to significantly improve quality of life, sexual function, and urinary health for millions of affected women. Continued efforts to reduce stigma, enhance education, and expand access to evidence based therapies are critical to closing the gap between disease burden and effective care.
Introduction
Menopause marks a pivotal transition in women’s lives, bringing numerous physiological changes that extend far beyond the cessation of menstruation. Among these changes, genitourinary syndrome of menopause represents one of the most distressing yet frequently overlooked consequences of estrogen deficiency. Unlike vasomotor symptoms such as hot flashes that typically resolve over time, GSM is a chronic, progressive condition that worsens without treatment.
The term “genitourinary syndrome of menopause” was introduced by the International Society for the Study of Women’s Sexual Health and the North American Menopause Society in 2014 to replace the previous terminology of “vulvovaginal atrophy” and “atrophic vaginitis.” This updated nomenclature better reflects the broader spectrum of symptoms affecting the vulva, vagina, and lower urinary tract. The condition encompasses genital symptoms including dryness, burning, and irritation; sexual symptoms such as dyspareunia and decreased lubrication; and urinary symptoms including urgency, frequency, and recurrent infections.
Despite affecting the majority of postmenopausal women, GSM remains shrouded in silence and inadequate care. Research indicates that fewer than 25% of women experiencing symptoms seek medical attention, and among those who do, many receive suboptimal treatment. This disconnect between disease prevalence and appropriate care represents a substantial healthcare gap with profound implications for women’s quality of life, sexual health, and overall well-being.
The purpose of this review is to examine why so many patients with GSM continue to suffer unnecessarily. We will explore the multifaceted barriers to optimal care, ranging from societal taboos surrounding female sexuality to gaps in medical education and clinical practice patterns. Additionally, we will review current evidence-based treatment approaches and identify opportunities to improve patient outcomes through enhanced recognition, diagnosis, and management of this condition.
Pathophysiology and Clinical Presentation
The development of GSM stems primarily from the dramatic decline in estrogen levels that occurs during menopause. Estrogen receptors are abundant throughout the vulvovaginal and urethral tissues, making these areas particularly susceptible to hormonal changes. As estrogen levels decrease, multiple physiological alterations occur that collectively contribute to the syndrome’s clinical manifestations.
Vaginal epithelial changes represent the cornerstone of GSM pathophysiology. Under normal premenopausal conditions, estrogen stimulates epithelial cell proliferation and glycogen storage within vaginal tissues. The glycogen is metabolized by lactobacilli, producing lactic acid that maintains an acidic vaginal pH between 3.5 and 4.5. This acidic environment provides natural protection against pathogenic bacteria and yeast overgrowth.
Following estrogen withdrawal, the vaginal epithelium undergoes progressive thinning and loses its protective multilayered structure. Epithelial cells decrease in size and number, while glycogen stores diminish substantially. The resulting reduction in lactobacilli colonization leads to pH elevation, typically rising above 5.0 in postmenopausal women. This alkaline shift predisposes women to increased risk of urinary tract infections and vaginal infections.
Concurrent with epithelial changes, the vaginal tissue experiences decreased blood flow and reduced collagen synthesis. These alterations result in diminished vaginal elasticity, shortened vaginal length, and narrowed vaginal introitus. The submucosal layer becomes thinner and less vascularized, contributing to decreased vaginal lubrication and increased tissue fragility. Many women report that sexual activity becomes painful or impossible due to these structural changes.
Urological manifestations of GSM arise from similar estrogen-dependent changes affecting the lower urinary tract. The urethral epithelium undergoes atrophic changes comparable to those observed in vaginal tissues. Decreased urethral closure pressure and altered detrusor muscle function contribute to symptoms of urinary urgency, frequency, nocturia, and stress incontinence. The bladder trigone, which contains estrogen receptors, may also be affected, potentially contributing to recurrent urinary tract infections.
The clinical presentation of GSM varies considerably among affected women. Some experience mild, intermittent symptoms that minimally impact daily life, while others develop severe manifestations that substantially impair quality of life and intimate relationships. Vulvar symptoms commonly include dryness, burning, itching, and irritation. The vulvar tissue may appear pale, smooth, and thin upon examination, with potential loss of labial architecture in severe cases.
Vaginal symptoms encompass dryness, burning, discharge, and odor. Sexual symptoms include dyspareunia, decreased arousal, reduced lubrication, and post-coital bleeding. Many women describe a gradual onset of these symptoms, with initial changes often attributed to normal aging or other factors. The progressive nature of GSM means that symptoms typically worsen over time without appropriate intervention.
Urinary symptoms associated with GSM include dysuria, urgency, frequency, nocturia, and recurrent urinary tract infections. Some women experience urinary incontinence, particularly stress incontinence during physical activities. The relationship between urogenital atrophy and lower urinary tract symptoms has been well-established in research studies, though the precise mechanisms continue to be investigated.
Epidemiology and Risk Factors
Understanding the epidemiology of GSM is challenging due to underreporting, varying diagnostic criteria, and cultural factors affecting symptom disclosure. However, available data suggests that GSM affects between 27% and 84% of postmenopausal women, depending on the population studied and diagnostic methods employed. The wide range in prevalence estimates reflects differences in study populations, symptom assessment tools, and cultural attitudes toward discussing intimate health concerns.
Age represents the most important risk factor for GSM development, with symptom prevalence increasing progressively with years since menopause. Unlike vasomotor symptoms that often improve spontaneously, GSM symptoms typically persist and worsen without treatment. Studies indicate that symptom severity correlates with time elapsed since menopause onset, highlighting the progressive nature of this condition.
Surgical menopause resulting from bilateral oophorectomy produces more rapid and severe GSM symptoms compared to natural menopause. The abrupt cessation of ovarian hormone production leaves tissues vulnerable to rapid atrophic changes. Women undergoing surgical menopause at younger ages may experience particularly severe symptoms and require more aggressive treatment approaches.
Breast cancer survivors face unique challenges regarding GSM management. Chemotherapy-induced premature menopause often results in severe, rapid-onset symptoms in relatively young women. Additionally, many breast cancer treatments, including aromatase inhibitors and selective estrogen receptor modulators, can exacerbate GSM symptoms by further suppressing estrogen activity. The contraindication or reluctance to use estrogen-based therapies in this population creates additional treatment challenges.
Smoking has been identified as an independent risk factor for GSM development, likely due to its anti-estrogenic effects and negative impact on tissue vascularization. Women who smoke tend to experience earlier menopause and more severe atrophic changes compared to non-smokers. Counseling regarding smoking cessation should be incorporated into GSM management strategies.
Cultural and ethnic factors influence GSM prevalence and treatment-seeking behavior. Studies suggest varying symptom reporting rates among different ethnic groups, though it remains unclear whether these differences reflect true biological variations or cultural attitudes toward discussing sexual health concerns. Healthcare providers must be sensitive to cultural considerations when assessing and treating women from diverse backgrounds.
Sexual activity patterns also influence GSM symptom development and severity. Regular sexual activity, including penetrative intercourse, masturbation, and use of vaginal moisturizers, appears to provide some protection against severe atrophic changes. The mechanism likely involves increased blood flow and mechanical stimulation that helps maintain tissue elasticity and thickness. However, sexual activity alone is insufficient to prevent GSM progression in most women.
Table 1: Risk Factors for Genitourinary Syndrome of Menopause
| Risk Factor | Impact Level | Notes |
| Age/Time since menopause | High | Progressive worsening over time |
| Surgical menopause | Very High | Abrupt hormone cessation |
| Cancer treatments | High | Chemotherapy, hormone therapy |
| Smoking | Moderate | Anti-estrogenic effects |
| Lack of sexual activity | Moderate | “Use it or lose it” phenomenon |
| Genetic factors | Low-Moderate | Family history influence |
| Autoimmune conditions | Variable | May affect hormone levels |
| Certain medications | Variable | Antihistamines, antidepressants |
Barriers to Optimal Care
Despite the high prevalence and available effective treatments for GSM, numerous barriers prevent women from receiving appropriate care. These obstacles exist at multiple levels, including patient, provider, and healthcare system factors that collectively contribute to the substantial gap between disease burden and optimal management.
Patient-related barriers represent perhaps the most formidable obstacles to GSM care. Many women experience embarrassment or shame discussing sexual health concerns with healthcare providers. Societal attitudes that normalize sexual dysfunction as an inevitable consequence of aging contribute to women’s reluctance to seek help. Research indicates that women often wait years before discussing GSM symptoms with healthcare providers, during which time their condition progressively worsens.
Lack of awareness about GSM and available treatments constitutes another major patient barrier. Many women attribute their symptoms to normal aging and remain unaware that effective treatments exist. The medicalization of menopause has historically focused on vasomotor symptoms, leaving GSM underrepresented in public health discussions and patient education materials. Consequently, women may suffer in silence, believing their condition is untreatable or not worthy of medical attention.
Provider-related barriers significantly impact GSM care quality. Many healthcare providers receive limited training in women’s sexual health during medical school and residency programs. This educational gap results in provider discomfort discussing intimate symptoms and inadequate knowledge of available treatment options. Time constraints during routine clinical encounters further limit opportunities for thorough GSM assessment and counseling.
The tendency to focus on acute medical problems during clinical visits often relegates GSM concerns to lower priority status. Healthcare providers may postpone discussions about sexual health in favor of addressing more pressing medical issues. Additionally, some providers harbor misconceptions about GSM treatment safety or efficacy, particularly regarding vaginal estrogen therapy.
Healthcare system barriers include inadequate reimbursement for sexual health counseling and limited access to specialists with expertise in menopausal medicine. The fragmentation of women’s healthcare across multiple specialties can result in no single provider taking responsibility for GSM management. Gynecologists may assume primary care providers are addressing the issue, while primary care providers may feel uncomfortable managing intimate concerns typically associated with gynecologic care.
Regulatory and pharmaceutical industry factors have also contributed to treatment access challenges. The removal of certain vaginal estrogen products from the market has limited treatment options for some women. Additionally, concerns about hormone therapy safety following the Women’s Health Initiative results have created lasting apprehension among both providers and patients regarding estrogen-based treatments, even topical formulations with minimal systemic absorption.
One particularly memorable patient encounter illustrates these barriers perfectly. A 67-year-old retired schoolteacher sheepishly mentioned at the end of her annual physical that she had developed “some sensitivity down there” that made intimacy with her husband of 45 years impossible. When asked how long this had been occurring, she admitted it had been over five years but she was “too embarrassed to bring it up” and thought it was “just part of getting older.” After education and treatment with vaginal estrogen, she returned three months later with a huge smile and announced that she and her husband had “rediscovered their honeymoon phase” – much to the amusement of the entire clinic staff who overheard her enthusiastic report from the hallway.
Cultural and linguistic barriers further compound access challenges for many women. Healthcare providers may lack cultural competency to address sexual health concerns in diverse populations. Language barriers can impede effective communication about intimate symptoms, and some cultures have strong taboos against discussing sexual matters with healthcare providers, particularly male physicians.
Current Diagnostic Approaches
Accurate diagnosis of GSM requires careful clinical assessment combining patient history, physical examination, and appropriate laboratory studies when indicated. However, diagnostic approaches vary considerably among healthcare providers, and many cases remain undiagnosed due to inadequate screening and assessment protocols.
The patient history represents the cornerstone of GSM diagnosis. Healthcare providers should systematically inquire about vulvar, vaginal, and urinary symptoms using open-ended questions that encourage honest disclosure. Important historical elements include symptom onset, duration, severity, and impact on quality of life and sexual function. Providers should assess the relationship between symptoms and menopause onset, as well as any previous treatments attempted.
Sexual history taking requires sensitivity and clinical skill to create a comfortable environment for patient disclosure. Relevant topics include current sexual activity levels, dyspareunia severity, lubrication adequacy, and impact on intimate relationships. Some women may require multiple visits to feel comfortable discussing these intimate concerns, emphasizing the importance of building trust and rapport.
The physical examination should include careful inspection of the vulva, vestibule, and vaginal tissues. Characteristic findings in GSM include pale, thin vulvar skin with potential loss of rugae and labial architecture. The vaginal mucosa may appear smooth, pale, and friable, with decreased moisture and elasticity. Palpation should assess for tenderness, tissue thickness, and any masses or lesions.
Vaginal pH measurement provides valuable diagnostic information and should be routinely performed when GSM is suspected. A pH above 5.0 in postmenopausal women not using hormone therapy supports the diagnosis of GSM. However, pH elevation can also result from infections or other conditions, so this finding must be interpreted in clinical context.
The vaginal maturation index, obtained through cytologic assessment of exfoliated vaginal cells, can provide objective evidence of estrogen deficiency. This test evaluates the ratio of parabasal, intermediate, and superficial epithelial cells, with GSM characterized by increased parabasal cells and decreased superficial cells. However, this test is not routinely performed in clinical practice due to cost and limited availability.
Laboratory studies are generally not required for GSM diagnosis but may be helpful in specific clinical situations. Follicle-stimulating hormone and estradiol levels can confirm menopausal status in women with uncertain menopausal timing. Urinalysis and urine culture should be obtained when urinary symptoms are present to exclude urinary tract infections.
Differential diagnosis considerations include vulvar dermatologic conditions, infections, and malignancy. Lichen sclerosus can produce vulvar symptoms similar to GSM but typically involves characteristic white, scarred-appearing lesions with possible architectural distortion. Lichen planus may present with erosive vulvar lesions and dyspareunia. Vulvar intraepithelial neoplasia and vulvar cancer should be considered when concerning lesions are present.
Vaginal infections, including bacterial vaginosis, yeast infections, and trichomoniasis, can produce symptoms overlapping with GSM. However, these conditions typically present with characteristic discharge and odor patterns that differ from GSM presentation. Concurrent infections and GSM can coexist, particularly given the altered vaginal environment in postmenopausal women.
Standardized assessment tools have been developed to aid in GSM diagnosis and symptom monitoring. The Vulvovaginal Symptom Questionnaire and the Day-to-Day Impact of Vaginal Aging Questionnaire provide validated instruments for quantifying symptom severity and treatment response. These tools can enhance clinical assessment and facilitate consistent outcome measurement.
Despite available diagnostic approaches, many cases of GSM remain undiagnosed due to inadequate screening practices. Routine inquiry about genitourinary symptoms should be incorporated into all postmenopausal women’s healthcare visits. Healthcare providers must create comfortable environments for symptom disclosure and maintain high clinical suspicion for GSM in appropriate patient populations.
Evidence-Based Treatment Options
Effective treatment options for GSM have evolved substantially over recent decades, offering hope for symptom relief and improved quality of life. Treatment approaches can be broadly categorized into hormonal and non-hormonal therapies, with selection based on symptom severity, patient preferences, medical contraindications, and individual response patterns.
Vaginal estrogen therapy represents the gold standard treatment for moderate to severe GSM symptoms. Multiple formulations are available, including creams, tablets, and rings, each offering distinct advantages and disadvantages. Vaginal estrogen works by restoring local tissue estrogen levels, promoting epithelial proliferation, increasing vaginal blood flow, and normalizing vaginal pH.
Vaginal estrogen cream is available in multiple formulations and concentrations. Conjugated equine estrogens cream and estradiol cream are commonly prescribed options. These preparations allow dose flexibility and can be applied directly to symptomatic areas. However, some women find creams messy and difficult to use consistently.
Vaginal estradiol tablets offer convenient, standardized dosing with minimal systemic absorption. The 10-microgram tablets are inserted using a disposable applicator and provide sustained local estrogen delivery. Clinical studies demonstrate excellent efficacy for vulvovaginal symptoms with this formulation. The pre-measured dose reduces concerns about over- or under-dosing common with creams.
Estradiol vaginal rings provide continuous hormone delivery over three-month periods. The ring releases estradiol directly to vaginal tissues while maintaining minimal systemic absorption. Many women prefer the convenience of quarterly insertion, though some experience ring-related discomfort or expulsion.
Safety considerations regarding vaginal estrogen therapy have been extensively studied. Systemic estrogen absorption from vaginal preparations is minimal, particularly with low-dose formulations. Serum estradiol levels typically remain within postmenopausal ranges during vaginal estrogen treatment. Long-term safety data support the use of vaginal estrogen even in women with contraindications to systemic hormone therapy, though individual risk-benefit assessment is always warranted.
Non-hormonal treatment options play important roles in GSM management, either as standalone therapy or in combination with hormonal treatments. Vaginal moisturizers provide sustained hydration to vaginal tissues and can be used regularly regardless of sexual activity timing. These products work by binding water to vaginal tissues and maintaining moisture between applications.
Personal lubricants offer temporary symptom relief during sexual activity. Water-based lubricants are compatible with condoms and sex toys but may require reapplication during prolonged intimate contact. Silicone-based lubricants provide longer-lasting lubrication but are not compatible with silicone-based devices. Oil-based products should be avoided with latex condoms due to material degradation risk.
Ospemifene represents a novel treatment approach as the first oral selective estrogen receptor modulator approved specifically for dyspareunia associated with GSM. This medication acts as an estrogen agonist in vaginal tissues while functioning as an estrogen antagonist in breast and endometrial tissues. Clinical trials demonstrate efficacy for vaginal dryness and dyspareunia, though some women experience vasomotor symptoms as side effects.
Energy-based therapies have emerged as newer treatment options for GSM, though long-term safety and efficacy data remain limited. Vaginal laser therapy using fractional CO2 or erbium lasers aims to stimulate collagen production and improve tissue vascularity. Radiofrequency treatments work through similar mechanisms to promote tissue remodeling and increased moisture production.
Preliminary studies of energy-based therapies show promising short-term results for GSM symptoms. However, the FDA has issued safety communications regarding vaginal laser and radiofrequency treatments due to reports of serious adverse events including burns, scarring, and pain. Additional research is needed to establish optimal treatment protocols and long-term safety profiles.
Combination therapy approaches may provide enhanced benefit for some women with severe or refractory symptoms. Vaginal estrogen can be combined with moisturizers, lubricants, or other treatments to optimize symptom control. Some women benefit from initial intensive treatment followed by maintenance therapy at reduced frequencies.
Lifestyle modifications complement medical treatments for GSM management. Regular sexual activity helps maintain vaginal tissue health through increased blood flow and mechanical stimulation. Pelvic floor physical therapy can address muscle dysfunction contributing to dyspareunia. Smoking cessation supports overall tissue health and may slow atrophic progression.
Treatment monitoring involves regular follow-up to assess symptom response and adjust therapy as needed. Most women experience symptom improvement within 2-4 weeks of starting vaginal estrogen therapy, with continued improvement over several months. Treatment response should be evaluated using standardized symptom scales when possible to provide objective outcome measures.
Applications and Clinical Use Cases
The clinical management of GSM requires individualized treatment approaches tailored to each patient’s specific symptoms, medical history, and personal preferences. Understanding various clinical scenarios and appropriate management strategies helps healthcare providers optimize patient outcomes across diverse populations and presentations.
Mild GSM symptoms may respond adequately to non-hormonal interventions. Women experiencing occasional vaginal dryness or mild dyspareunia might benefit from regular vaginal moisturizer use and appropriate lubricants during sexual activity. This approach is particularly suitable for women who prefer to avoid hormonal treatments or have contraindications to estrogen therapy.
Moderate to severe GSM symptoms typically require more aggressive intervention, usually involving vaginal estrogen therapy. Women with substantial vulvovaginal atrophy, severe dyspareunia, or recurrent urinary tract infections often achieve optimal results with prescription hormonal treatments. The choice of specific estrogen formulation depends on patient preferences, manual dexterity, and individual response patterns.
Breast cancer survivors present unique management challenges due to concerns about estrogen exposure and potential cancer recurrence risk. While vaginal estrogen absorption is minimal, many oncologists and patients remain hesitant about any estrogen-containing treatments. Non-hormonal options including moisturizers, lubricants, and potentially ospemifene may be preferred in this population. Some breast cancer survivors may be candidates for low-dose vaginal estrogen after careful risk-benefit analysis and oncology consultation.
Women with recurrent urinary tract infections associated with GSM often benefit from vaginal estrogen therapy, which can help normalize vaginal pH and restore protective lactobacilli populations. The restoration of normal vaginal flora reduces urinary tract infection recurrence rates and may eliminate the need for prophylactic antibiotics. Cranberry products and increased fluid intake provide additional supportive measures.
Postmenopausal women resuming sexual activity after periods of abstinence may require graduated treatment approaches. Initial therapy might focus on tissue restoration using vaginal estrogen, followed by gradual reintroduction of sexual activity with appropriate lubrication. Pelvic floor physical therapy can address muscle tension and coordination issues that may develop during periods of sexual inactivity.
Women undergoing chemotherapy for various cancers may develop rapid-onset, severe GSM symptoms due to acute estrogen suppression. These patients often require prompt, aggressive treatment to prevent severe atrophic changes. Close collaboration between oncology and women’s health specialists helps optimize symptom management while respecting cancer treatment protocols.
Surgical menopause cases require particular attention due to the abrupt nature of hormonal changes. Women undergoing bilateral oophorectomy may benefit from immediate initiation of vaginal estrogen therapy to prevent severe atrophic changes. Early intervention appears more effective than waiting for symptoms to develop before starting treatment.
Perimenopausal women experiencing GSM symptoms present diagnostic and treatment challenges due to fluctuating hormone levels. Some women develop vulvovaginal symptoms before menstruation cessation, particularly those using hormonal contraceptives or experiencing irregular cycles. Treatment approaches may need modification as women transition through different menopausal stages.
Women with multiple medical comorbidities require careful consideration of drug interactions and contraindications. Certain medications, including antihistamines and antidepressants, can worsen GSM symptoms through anticholinergic effects. Diabetes management affects infection risk and tissue healing capacity, potentially influencing treatment selection and monitoring requirements.
Cultural considerations significantly impact GSM management approaches. Some cultures have strong taboos regarding discussion of sexual health, requiring sensitive communication strategies and possibly involvement of female healthcare providers. Religious considerations may influence treatment acceptability, particularly regarding hormone therapy use.
Economic factors affect treatment accessibility and adherence. Insurance coverage varies substantially for GSM treatments, with some moisturizers and lubricants requiring out-of-pocket payment. Cost considerations may influence treatment selection, emphasizing the importance of discussing affordable options with patients.
Long-term care facility residents represent an often-overlooked population with GSM management needs. Many elderly women in nursing homes experience severe atrophic changes but receive inadequate assessment and treatment. Healthcare providers in these settings require education about GSM recognition and management strategies appropriate for this vulnerable population.
Comparison with Related Conditions
Understanding GSM within the broader context of vulvovaginal and urologic conditions helps healthcare providers establish accurate diagnoses and implement appropriate treatment strategies. Several conditions share overlapping symptoms with GSM, requiring careful differential diagnosis and sometimes concurrent management approaches.
Vulvar dermatologic conditions frequently present with symptoms similar to GSM, particularly vulvar burning, itching, and dyspareunia. Lichen sclerosus affects approximately 1-3% of postmenopausal women and can coexist with GSM. This condition produces characteristic white, porcelain-like vulvar lesions with potential scarring and architectural distortion. Unlike GSM, lichen sclerosus typically requires high-potency topical corticosteroids as first-line treatment.
Lichen planus represents another inflammatory vulvar condition that can mimic GSM symptoms. The erosive variant produces painful vulvar and vaginal lesions with potential for severe scarring. Treatment usually involves topical immunosuppressants or corticosteroids rather than estrogen therapy. Biopsy may be required to distinguish between these conditions and GSM.
Contact dermatitis affecting the vulvar area can produce burning and irritation similar to GSM. However, this condition typically has identifiable triggers such as soaps, detergents, or personal care products. Treatment focuses on trigger avoidance and short-term topical anti-inflammatory medications.
Vulvodynia, characterized by chronic vulvar pain without identifiable cause, shares the symptom of dyspareunia with GSM. However, vulvodynia can affect women of any age and typically involves neuropathic-type pain rather than the dryness and atrophy characteristic of GSM. Treatment approaches differ substantially, often involving tricyclic antidepressants, anticonvulsants, or topical anesthetics.
Vaginal infections can produce symptoms overlapping with GSM, including discharge, odor, and discomfort. Bacterial vaginosis presents with malodorous, grayish discharge and elevated vaginal pH, similar to the pH changes seen in GSM. However, bacterial vaginosis typically involves characteristic fishy odor and clue cells on microscopic examination.
Candida vulvovaginitis produces vulvar itching and burning that can be confused with GSM symptoms. The thick, white, cottage cheese-like discharge characteristic of yeast infections usually distinguishes this condition from GSM. However, recurrent yeast infections may occur more frequently in women with GSM due to altered vaginal pH and immune function.
Urologic conditions affecting postmenopausal women often coexist with GSM, complicating diagnosis and treatment. Overactive bladder syndrome shares urinary urgency and frequency symptoms with GSM but typically responds to anticholinergic medications or beta-3 agonists rather than estrogen therapy. However, some women experience improvement in overactive bladder symptoms with vaginal estrogen treatment.
Stress urinary incontinence commonly affects postmenopausal women and may be influenced by estrogen deficiency effects on urethral tissues. While vaginal estrogen may provide some benefit for stress incontinence, definitive treatment usually requires surgical intervention or pessary devices.
Recurrent urinary tract infections can result from various factors in postmenopausal women, including GSM-related pH changes, incomplete bladder emptying, and immune system changes. While vaginal estrogen can help prevent UTIs associated with GSM, other causes such as bladder dysfunction or anatomical abnormalities may require different interventions.
Pelvic organ prolapse affects many postmenopausal women and can contribute to urinary symptoms and sexual dysfunction. While estrogen deficiency may contribute to prolapse development, treatment typically involves pessaries or surgical repair rather than hormonal therapy alone.
Sexual dysfunction in postmenopausal women encompasses multiple factors beyond GSM, including decreased libido, arousal difficulties, and orgasmic dysfunction. While treating GSM-related dyspareunia may improve overall sexual function, additional interventions such as counseling, relationship therapy, or other medications may be required for optimal outcomes.
Thyroid disorders can affect vaginal lubrication and sexual function in postmenopausal women. Hypothyroidism may exacerbate GSM symptoms, while hyperthyroidism can cause vaginal dryness through different mechanisms. Appropriate thyroid hormone management should complement GSM treatment in affected women.
Depression and anxiety frequently affect postmenopausal women and can impact sexual function and treatment adherence. Some antidepressants may worsen GSM symptoms through anticholinergic effects, requiring medication adjustments or additional symptomatic treatments.
Challenges and Limitations
Despite advances in understanding and treating GSM, numerous challenges and limitations continue to impede optimal patient care. These obstacles span clinical, research, regulatory, and societal domains, requiring coordinated efforts to address gaps in knowledge and practice.
Research limitations significantly impact GSM understanding and treatment development. Many studies suffer from small sample sizes, short follow-up periods, and heterogeneous patient populations. The subjective nature of many GSM symptoms makes objective outcome measurement challenging, leading to inconsistent research findings and difficulty comparing treatment efficacy across studies.
Long-term safety data for newer treatments remain limited, particularly for energy-based therapies and novel pharmaceutical agents. The lack of standardized treatment protocols and outcome measures makes it difficult to establish evidence-based practice guidelines. Additionally, most research focuses on Caucasian women, limiting generalizability to diverse populations.
Clinical practice variations contribute to inconsistent GSM care across different healthcare settings and geographic regions. Provider knowledge gaps regarding GSM pathophysiology, diagnosis, and treatment options result in suboptimal patient management. The lack of standardized clinical assessment tools leads to inconsistent diagnostic approaches and difficulty monitoring treatment response.
Time constraints during routine clinical encounters limit opportunities for thorough GSM assessment and patient education. Many healthcare providers lack confidence discussing sexual health topics, leading to inadequate screening and counseling. The absence of systematic screening protocols means many cases remain undiagnosed until symptoms become severe.
Regulatory challenges affect treatment access and development of new therapeutic options. The FDA’s safety communications regarding vaginal laser therapy have created uncertainty about these treatment modalities among both providers and patients. Stringent regulatory requirements for new drug approvals may discourage pharmaceutical company investment in GSM treatment development.
Insurance coverage limitations create financial barriers to treatment access for many women. Some effective treatments, including vaginal moisturizers and certain hormone preparations, may not be covered by insurance plans. High copayments for prescription medications can limit treatment adherence, particularly among elderly women on fixed incomes.
Societal attitudes and cultural factors continue to stigmatize discussions about female sexuality and aging. The medicalization of menopause has historically focused on vasomotor symptoms while neglecting sexual health concerns. Media representation of postmenopausal women often perpetuates myths about inevitable sexual decline with aging.
Educational deficiencies affect both healthcare providers and patients. Medical school curricula often provide limited training in women’s sexual health and menopause management. Continuing education opportunities in GSM are not uniformly available across medical specialties. Patient educational materials frequently lack cultural sensitivity and may not address diverse populations’ needs.
Healthcare delivery system fragmentation creates coordination challenges for GSM management. The division of women’s healthcare across primary care, gynecology, urology, and other specialties can result in care gaps and duplicated services. Electronic health record systems may not adequately support screening for and tracking of GSM symptoms.
Geographic disparities in specialist availability affect access to expertise in menopause and sexual medicine. Rural and underserved areas often lack healthcare providers with advanced training in GSM management. Telemedicine options may help address these disparities but face regulatory and reimbursement challenges.
Age-related biases in healthcare delivery affect how GSM symptoms are perceived and prioritized. Some healthcare providers may dismiss sexual health concerns in older women as normal aging rather than treatable medical conditions. Ageist attitudes can lead to inadequate symptom assessment and limited treatment options being offered.
Comorbidity management complexity increases with aging, potentially affecting GSM treatment selection and monitoring. Drug interactions, polypharmacy concerns, and multiple chronic conditions may limit treatment options or require careful coordination among multiple healthcare providers.
Patient adherence challenges affect treatment outcomes even when appropriate therapies are prescribed. Some women discontinue treatment due to side effects, inconvenience, or failure to achieve expected results. Lack of follow-up monitoring can result in missed opportunities to adjust treatments or address adherence barriers.
Future Directions and Research Opportunities
The field of GSM research and clinical care continues to evolve, with numerous opportunities for advancement in understanding, diagnosis, and treatment. Future research directions should address current knowledge gaps while developing innovative approaches to improve patient outcomes and quality of life.
Novel therapeutic targets offer promising avenues for GSM treatment development. Research into selective estrogen receptor modulators with tissue-specific activity may yield new medications with improved efficacy and safety profiles. Investigation of androgen therapy for GSM symptoms represents another potential therapeutic approach, given the role of testosterone in maintaining genital tissue health.
Regenerative medicine approaches, including stem cell therapy and growth factor treatments, may offer future treatment options for severe GSM. These interventions aim to restore normal tissue structure and function rather than simply managing symptoms. However, substantial research is needed to establish safety and efficacy for these experimental approaches.
Personalized medicine strategies could optimize GSM treatment selection based on individual patient characteristics. Genetic markers affecting estrogen receptor sensitivity or drug metabolism might guide treatment choices. Biomarkers predicting treatment response could help identify patients most likely to benefit from specific therapies.
Digital health technologies present opportunities to improve GSM care delivery and monitoring. Mobile applications could facilitate symptom tracking, medication reminders, and patient education. Telemedicine platforms may expand access to specialist care for women in underserved areas. Artificial intelligence tools might assist healthcare providers in diagnosis and treatment selection.
Diagnostic innovations could enhance GSM recognition and monitoring. Point-of-care testing for vaginal biomarkers might provide objective diagnostic information during clinical encounters. Imaging technologies could assess tissue changes and treatment response more accurately than current visual examination methods.
Patient-reported outcome measures require further development and validation to standardize GSM assessment and research. Culturally sensitive instruments are needed to ensure accurate symptom assessment across diverse populations. Electronic data capture systems could facilitate large-scale studies and real-world evidence generation.
Prevention strategies represent important research opportunities given GSM’s progressive nature. Early intervention studies could determine whether treatment initiation before symptom development prevents severe atrophic changes. Lifestyle interventions and dietary supplements might offer preventive benefits worthy of investigation.
Healthcare delivery research should focus on developing effective screening protocols and provider education strategies. Implementation science approaches could identify optimal methods for integrating GSM care into routine clinical practice. Cost-effectiveness studies are needed to support insurance coverage decisions and healthcare policy development.
Epidemiologic research in diverse populations would improve understanding of GSM prevalence, risk factors, and natural history across different ethnic and cultural groups. Longitudinal studies following women from perimenopause through advanced postmenopausal years could provide insights into disease progression and optimal intervention timing.
Safety studies for existing and emerging treatments require continued attention, particularly regarding long-term outcomes and special populations. Post-marketing surveillance of energy-based therapies could clarify appropriate patient selection and treatment protocols. Studies in cancer survivors and other high-risk populations are needed to establish safety profiles.
Mechanistic research into GSM pathophysiology could identify new therapeutic targets and improve understanding of treatment mechanisms. Investigation of the role of inflammation, immune function, and tissue remodeling in GSM development might yield novel intervention strategies.
Educational research should focus on developing effective training programs for healthcare providers and educational materials for patients. Evaluation of different educational approaches could identify optimal methods for improving provider competency and patient knowledge about GSM.
Quality improvement initiatives within healthcare systems could demonstrate effective strategies for improving GSM care delivery. These studies might evaluate screening protocols, provider training programs, and care coordination models to identify best practices for widespread implementation.
Genitourinary syndrome of menopause represents a highly prevalent yet persistently undertreated condition affecting millions of postmenopausal women worldwide. Despite the availability of effective treatments, substantial barriers continue to prevent many women from receiving appropriate care, leaving them to suffer unnecessarily with symptoms that profoundly impact their quality of life and intimate relationships.
The evidence clearly demonstrates that GSM is not an inevitable consequence of aging that women must simply endure. Rather, it is a treatable medical condition with well-established pathophysiology and proven therapeutic interventions. Vaginal estrogen therapy remains the gold standard treatment for moderate to severe symptoms, offering excellent efficacy with minimal safety concerns. Non-hormonal options provide alternatives for women who cannot or prefer not to use hormone therapy.
Addressing the current care gaps requires coordinated efforts across multiple domains. Healthcare providers need improved education about GSM recognition, diagnosis, and management. Medical training programs should incorporate women’s sexual health and menopause medicine into their curricula. Continuing education opportunities must be expanded to reach practicing clinicians across all relevant specialties.
Patient education and awareness campaigns are essential to help women understand that GSM symptoms are treatable and that seeking help is appropriate. These efforts must be culturally sensitive and address diverse populations’ needs. Healthcare systems should implement systematic screening protocols and provide adequate time and resources for GSM assessment and management.
The development of new treatments and improvement of existing therapies will require continued research investment. Novel therapeutic approaches, including regenerative medicine and personalized treatment strategies, hold promise for the future. However, immediate improvements in care can be achieved through better utilization of currently available treatments.
Healthcare policy changes, including improved insurance coverage for GSM treatments and support for specialized care, could remove financial barriers to treatment access. Professional organizations should develop and disseminate evidence-based practice guidelines to standardize care approaches.
The ultimate goal is to ensure that no woman suffers unnecessarily from GSM symptoms due to lack of awareness, inadequate care, or treatment barriers. With coordinated efforts to address current challenges, we can substantially improve outcomes for the millions of women affected by this condition.
The journey toward optimal GSM care requires commitment from healthcare providers, patients, healthcare systems, and society as a whole. By working together to overcome existing barriers and implement evidence-based solutions, we can transform the landscape of menopausal healthcare and restore quality of life for countless women who deserve better than silent suffering.
Key Takeaways
- GSM affects up to 84% of postmenopausal women but remains underdiagnosed and undertreated due to multiple barriers
- The condition is chronic and progressive, worsening without treatment unlike vasomotor symptoms that often resolve spontaneously
- Vaginal estrogen therapy represents the gold standard treatment with excellent efficacy and minimal systemic absorption
- Non-hormonal options including moisturizers, lubricants, and ospemifene provide alternatives for women who cannot use estrogen
- Healthcare provider education and systematic screening protocols are essential to improve recognition and management
- Patient awareness campaigns must address cultural barriers and normalize discussions about sexual health in older women
- Early intervention may prevent severe atrophic changes and improve long-term outcomes
- Individualized treatment approaches should consider patient preferences, medical history, and cultural factors
- Research opportunities exist for novel treatments, diagnostic tools, and healthcare delivery improvements
- Quality of life can be substantially improved with appropriate recognition and treatment of GSM
Frequently Asked Questions
Q: Is GSM a normal part of aging that women should just accept?
A: No, GSM is a treatable medical condition, not an inevitable consequence of aging. While estrogen deficiency after menopause commonly causes these symptoms, effective treatments are available to restore comfort and quality of life.
Q: Are vaginal estrogen treatments safe for women with a history of breast cancer?
A: Vaginal estrogen has minimal systemic absorption, but decisions about its use in breast cancer survivors should always involve consultation with the patient’s oncology team to weigh individual risks and benefits.
Q: How long does it take to see improvement with GSM treatments?
A: Most women begin experiencing symptom relief within 2-4 weeks of starting vaginal estrogen therapy, with continued improvement over several months. Non-hormonal treatments may provide more immediate but temporary relief.
Q: Can GSM symptoms return after stopping treatment?
A: Yes, GSM is a chronic condition that typically returns when treatment is discontinued. Most women require ongoing therapy to maintain symptom relief, though maintenance regimens may be less frequent than initial treatment.
Q: Are over-the-counter treatments effective for GSM?
A: Over-the-counter vaginal moisturizers and lubricants can be helpful for mild symptoms or as adjunctive treatments. However, moderate to severe GSM typically requires prescription therapies for optimal management.
Q: Should women avoid sexual activity if they have painful GSM symptoms?
A: Women should not avoid sexual activity due to GSM symptoms. Instead, they should seek treatment to address the underlying cause of pain. Regular sexual activity with appropriate lubrication may actually help maintain tissue health.
Q: How do I know if my symptoms are due to GSM or another condition?
A: GSM symptoms typically develop gradually after menopause and include vaginal dryness, burning, and painful intercourse. A healthcare provider can perform appropriate examinations and tests to distinguish GSM from other conditions.
Q: Is it embarrassing to discuss these symptoms with my doctor?
A: While it’s natural to feel some embarrassment, healthcare providers are trained to discuss intimate health concerns professionally. GSM is a common medical condition that affects most postmenopausal women, and discussing symptoms is important for proper treatment.
References
Baber, R. J., Panay, N., Fenton, A., & IMS Writing Group. (2016). 2016 IMS recommendations on women’s midlife health and menopause hormone therapy. Climacteric, 19(2), 109-150.
Constantine, G. D., Goldstein, S. R., Archer, D. F., Kagan, R., Kaunitz, A. M., Kingsberg, S. A., … & Stuenkel, C. A. (2019). Endometrial safety of ultra-low-dose vaginal estrogens in postmenopausal women: A systematic review. Menopause, 26(9), 1019-1026.
Gandhi, J., Chen, A., Dagur, G., Suh, Y., Smith, N., Cali, B., & Khan, S. A. (2016). Genitourinary syndrome of menopause: An overview of clinical manifestations, pathophysiology, etiology, evaluation, and management. American Journal of Obstetrics and Gynecology, 215(6), 704-711.
Kingsberg, S. A., Wysocki, S., Magnus, L., & Krychman, M. L. (2013). Vulvar and vaginal atrophy in postmenopausal women: Findings from the REVIVE survey. Journal of Sexual Medicine, 10(7), 1790-1799.
Mac Bride, M. B., Rhodes, D. J., & Shuster, L. T. (2010). Vulvovaginal atrophy. Mayo Clinic Proceedings, 85(1), 87-94.
Management of symptomatic vulvovaginal atrophy: 2013 position statement of The North American Menopause Society. (2013). Menopause, 20(9), 888-902.
Moral, E., Delgado, J. L., Carmona, F., Caballero, B., Guillán, C., González, P. M., … & Coronado, P. (2018). The impact of genitourinary syndrome of menopause on well-being, functioning, and quality of life in postmenopausal women. Menopause, 25(12), 1418-1423.
Nappi, R. E., & Kokot-Kierepa, M. (2012). Vaginal health: Insights, views & attitudes (VIVA)—results from an international survey. Climacteric, 15(1), 36-44.
Parish, S. J., Nappi, R. E., Krychman, M. L., Kellogg-Spadt, S., Simon, J. A., Goldstein, J. A., & Kingsberg, S. A. (2013). Impact of vulvovaginal health on postmenopausal women: A review of surveys on symptoms of vulvovaginal atrophy. International Journal of Women’s Health, 5, 437-447.
Portman, D. J., & Gass, M. L. (2014). Genitourinary syndrome of menopause: New terminology for vulvovaginal atrophy from the International Society for the Study of Women’s Sexual Health and the North American Menopause Society. Menopause, 21(10), 1063-1068.
Santen, R. J. (2014). Vaginal administration of estradiol: Effects of dose, preparation and timing on plasma estradiol levels. Climacteric, 18(2), 121-134.
Simon, J. A., Kokot-Kierepa, M., Goldstein, J., & Nappi, R. E. (2013). Vaginal health in the United States: Results from the Vaginal Health: Insights, Views & Attitudes survey. Menopause, 20(10), 1043-1048.
The NAMS 2020 GSM Position Statement Editorial Panel. (2020). The 2020 genitourinary syndrome of menopause position statement of The North American Menopause Society. Menopause, 27(9), 976-992.
Thurston, R. C., Chang, Y., Mancuso, P., & Matthews, K. A. (2013). Adipokines, adiposity, and vasomotor symptoms during the menopause transition: Findings from the Study of Women’s Health Across the Nation. Fertility and Sterility, 100(3), 793-800.
Wierman, M. E., Nappi, R. E., Avis, N., Davis, S. R., Labrie, F., Rosner, W., & Shifren, J. L. (2010). Endocrine aspects of women’s sexual function. Journal of Sexual Medicine, 7(1), 561-585.
Recent Articles


Integrative Perspectives on Cognition, Emotion, and Digital Behavior

Sleep-related:
Longevity/Nutrition & Diet:
Philosophical / Happiness:
Other:
Video Section 

