Prehospital Stroke Scales and Mobile CT Units: Worth the Investment?

Abstract
Rapid recognition and treatment of acute stroke remain among the most important determinants of neurological recovery and long term functional outcome. Because cerebral ischemia leads to irreversible neuronal injury within minutes, every stage of early stroke care, beginning in the prehospital setting, directly influences survival, disability, and healthcare resource utilization. In recent years, growing attention has focused on strategies that can improve stroke identification before hospital arrival and accelerate treatment initiation for eligible patients. Among these strategies, prehospital stroke assessment tools and mobile computed tomography units have emerged as important innovations with the potential to reshape emergency stroke pathways.
This analysis examines the clinical effectiveness and economic value of these prehospital interventions in contemporary acute stroke care. It evaluates current evidence regarding their ability to improve diagnostic accuracy, shorten treatment timelines, enhance patient outcomes, and optimize healthcare system performance. Particular attention is given to their role in early identification of large vessel occlusion, triage decisions regarding thrombectomy capable centers, and reduction of delays in thrombolytic therapy.
Methodology
A systematic review of recent literature was undertaken, focusing on studies published between 2018 and 2024. The review included peer reviewed journal articles, randomized and observational clinical trials, registry based analyses, emergency medical services reports, and health economic evaluations relevant to prehospital stroke management. Literature was selected based on relevance to clinical utility, diagnostic performance, operational feasibility, and cost effectiveness in real world emergency settings.
The analysis examined several key outcome measures. These included sensitivity and specificity of prehospital stroke scales for identifying acute ischemic stroke and large vessel occlusion, time intervals from symptom onset to diagnosis, door to needle time for intravenous thrombolysis, door to puncture time for mechanical thrombectomy, and functional outcomes measured by modified Rankin Scale scores at 90 days. Economic endpoints included cost effectiveness ratios, equipment investment costs, staffing requirements, and projected long term savings associated with reduced disability and institutional care needs.
Main Findings
Prehospital stroke scales continue to serve as the foundation of early stroke recognition in emergency medical services. Commonly used tools such as the Cincinnati Prehospital Stroke Scale, Los Angeles Motor Scale, Rapid Arterial Occlusion Evaluation scale, and other large vessel occlusion prediction models demonstrate variable diagnostic performance depending on the specific scale used, the training level of prehospital personnel, and the clinical context in which they are applied. Across studies, overall accuracy rates ranged from approximately 65 percent to 85 percent. Simpler scales generally showed strong usability and rapid application in the field, whereas more complex scales offered improved identification of severe stroke but required greater operator familiarity.
The diagnostic challenge remains particularly significant for large vessel occlusion, where rapid identification is essential because of the narrow therapeutic window for mechanical thrombectomy. Although no single prehospital scale consistently achieves ideal sensitivity and specificity, several tools demonstrate reasonable predictive value when incorporated into structured emergency medical services protocols. Evidence suggests that regular training and standardized implementation significantly improve scale reliability and reduce interobserver variability.
Mobile computed tomography units have shown important potential in reducing treatment delays by bringing neuroimaging directly to the patient before hospital arrival. These mobile stroke units are equipped with onboard CT scanners, telemedicine capability, point of care laboratory testing, and in some systems, stroke specialized personnel who can initiate treatment decisions immediately. Studies consistently demonstrate that mobile CT deployment reduces treatment times, particularly door to needle intervals, by approximately 23 to 45 minutes in selected populations. This reduction is clinically meaningful, as earlier thrombolysis is strongly associated with improved neurological recovery and reduced infarct progression.
In addition to shortening treatment times, mobile CT units improve diagnostic certainty by differentiating ischemic stroke from intracerebral hemorrhage before hospital transfer. This early distinction supports more accurate triage decisions, allowing direct transport to thrombectomy capable centers when indicated and avoiding unnecessary delays associated with secondary transfer.
Functional outcome data suggest that earlier intervention facilitated by mobile imaging may improve rates of independence at follow up, particularly in urban systems where stroke unit deployment is integrated into established emergency networks. However, benefit appears highly dependent on population density, transport distances, and availability of stroke centers.
Economic analyses reveal a more complex picture. Mobile CT systems require substantial initial investment, including acquisition of specialized vehicles, onboard imaging equipment, telecommunication systems, and highly trained personnel. Operational costs remain significant due to staffing, maintenance, and technology upgrades. Despite these high upfront expenditures, several cost effectiveness models suggest that improved functional outcomes and reduced disability may offset costs over a three to five year period, particularly when long term reductions in rehabilitation needs, nursing care, and recurrent hospitalizations are considered.
Cost effectiveness is most favorable in high volume metropolitan regions with efficient dispatch systems and strong stroke referral infrastructure. In lower density settings, economic benefit is less consistent because fewer eligible patients may dilute system efficiency. Hybrid deployment strategies and selective geographic targeting have therefore emerged as potential solutions to improve sustainability.
Conclusion
Current evidence indicates that both prehospital stroke assessment tools and mobile computed tomography units play increasingly important roles in modern acute stroke systems of care. Prehospital scales remain essential for rapid field identification and triage, although their diagnostic performance continues to depend heavily on training and protocol consistency. Mobile CT technology offers significant advantages in reducing treatment delays and improving early therapeutic decision making, particularly for ischemic stroke and large vessel occlusion.
From a health systems perspective, the clinical value of these interventions is clear, but economic justification remains context dependent. Future progress will depend on refining stroke prediction tools, improving integration with emergency medical services, expanding telemedicine support, and developing scalable cost models that allow broader implementation without compromising sustainability. As stroke care continues to evolve toward earlier intervention and precision triage, prehospital innovations are likely to become central components of high performance emergency neurological care.
Keywords: stroke assessment, prehospital care, mobile imaging, emergency medical services, cost effectiveness
Introduction
Acute stroke remains one of the most notable contributors to global mortality and long term disability, imposing a substantial clinical, social, and economic burden on healthcare systems worldwide. In the United States alone, approximately 795,000 individuals experience a stroke each year, with ischemic stroke accounting for the majority of cases. Despite improvements in acute care pathways, stroke continues to be a leading cause of permanent neurological impairment, frequently resulting in reduced independence, prolonged rehabilitation needs, and high rates of recurrent hospitalization. Because neuronal injury progresses rapidly after vascular occlusion, the urgency of early diagnosis and intervention has become central to modern stroke management.
The concept that “time is brain” has become a defining principle in acute stroke care. Quantitative analyses estimate that for every minute a large vessel ischemic stroke remains untreated, approximately 1.9 million neurons are lost, along with billions of synaptic connections and marked myelinated fiber damage. This accelerated biological deterioration directly influences functional outcomes, making treatment delays clinically consequential even within short time intervals. The effectiveness of reperfusion therapies such as intravenous thrombolysis and mechanical thrombectomy is highly dependent on early recognition and rapid initiation, which has intensified interest in strengthening prehospital stroke systems.
Emergency medical services occupy a critical position in this treatment pathway because they are often the first healthcare professionals to evaluate stroke patients. Historically, however, prehospital stroke recognition relied largely on clinical suspicion and general neurological observation, with limited structured diagnostic support. This often contributed to under recognition of atypical presentations, delays in hospital notification, and suboptimal triage decisions, particularly when distinguishing stroke from common mimics such as seizures, hypoglycemia, migraine, or functional neurological disorders.
To address this limitation, standardized prehospital stroke scales were developed to improve early recognition and guide destination decisions. These tools provide emergency personnel with structured neurological assessment frameworks that can be applied rapidly in the field. Commonly used scales such as the Cincinnati Prehospital Stroke Scale, Los Angeles Prehospital Stroke Screen, and Face Arm Speech Test focus on key clinical features including facial asymmetry, arm weakness, and speech disturbance. Their simplicity supports broad implementation and rapid decision making, particularly in high volume emergency systems.
More advanced scales have since emerged to improve detection of severe stroke syndromes, particularly large vessel occlusion, which require access to thrombectomy capable centers. Instruments such as the Rapid Arterial Occlusion Evaluation scale, the Los Angeles Motor Scale, and the Cincinnati Stroke Triage Assessment Tool incorporate additional motor and cortical findings to improve predictive accuracy. These expanded tools assist emergency personnel in identifying patients who may benefit from direct transport to comprehensive stroke centers, thereby reducing secondary transfers and treatment delays.
Although these scales improve early recognition, they remain dependent on clinical interpretation and cannot definitively distinguish ischemic stroke, hemorrhagic stroke, or stroke mimics. This limitation has driven interest in mobile diagnostic technologies capable of extending imaging capabilities into the prehospital setting.
Mobile computed tomography units have emerged as one of the most innovative developments in acute stroke systems of care. These specially designed ambulances are equipped with onboard CT scanners, point of care laboratory testing, telemedicine connectivity, and trained stroke teams capable of initiating diagnostic evaluation before hospital arrival. The goal is to bring imaging and decision making directly to the patient, allowing treatment pathways to begin during the earliest phase of care.
The clinical rationale for mobile stroke units is compelling. By obtaining immediate neuroimaging in the field, clinicians can differentiate ischemic stroke from intracerebral hemorrhage without waiting for hospital based imaging. This distinction is essential because treatment strategies differ fundamentally, with thrombolytic therapy indicated only for eligible ischemic stroke patients. In several observational and controlled studies, mobile stroke units have demonstrated notable reductions in door to needle times and earlier administration of thrombolysis compared with conventional emergency transport pathways.
Earlier treatment initiation has important downstream implications. Faster reperfusion is associated with improved functional recovery, reduced infarct size, and lower long term disability. Patients treated earlier are more likely to achieve independence in activities of daily living and less likely to require extended rehabilitation or institutional care. In some systems, mobile stroke units have also improved triage accuracy by identifying patients who should bypass local hospitals in favor of comprehensive stroke centers for endovascular therapy.
Despite these advantages, implementation remains complex. Mobile stroke units require substantial capital investment, including vehicle acquisition, CT imaging equipment, telecommunication systems, and staffing models that often include paramedics, radiology technicians, and stroke trained clinicians. Operational costs are also massive due to maintenance, continuous availability requirements, and limited patient throughput compared with conventional ambulances.
These financial demands raise important questions regarding cost effectiveness and scalability. Economic evaluations suggest that while upfront expenditures are high, potential long term savings may arise from reduced disability, shorter hospital stays, lower rehabilitation costs, and decreased need for chronic institutional care. Improved functional outcomes can translate into reduced societal costs through preservation of productivity and lower caregiver burden. However, these benefits are highly dependent on regional stroke incidence, transport distances, healthcare infrastructure, and integration with established stroke networks.
The value proposition also varies by geographic context. Urban regions with dense populations and high stroke volumes may achieve greater operational efficiency and clinical return on investment than rural areas where long travel distances and lower case density complicate deployment. In rural settings, alternative strategies such as tele stroke supported emergency transport, expanded regional stroke protocols, and selective deployment models may offer more sustainable solutions.
The broader economics of prehospital stroke innovation therefore extend beyond direct treatment costs. Decision makers must consider whether investment in advanced prehospital diagnostics produces measurable gains in quality adjusted life years, healthcare utilization, and long term patient independence when compared with strengthening conventional stroke pathways.
Current evidence suggests that both standardized stroke scales and mobile imaging technologies contribute meaningfully to earlier stroke recognition and treatment acceleration, but their roles are complementary rather than interchangeable. Stroke scales remain essential because they are inexpensive, scalable, and immediately deployable across diverse emergency systems. Mobile CT technologies offer a higher level of diagnostic precision but require targeted deployment where infrastructure and patient volume justify their cost.
As stroke systems continue to evolve, future progress will likely depend on integrating clinical assessment tools, imaging technology, artificial intelligence supported triage, and telemedicine into coordinated prehospital care models. For clinicians, emergency systems planners, and policymakers, the central challenge is not simply whether these innovations improve outcomes, but how they can be implemented in ways that maximize both clinical benefit and economic sustainability.
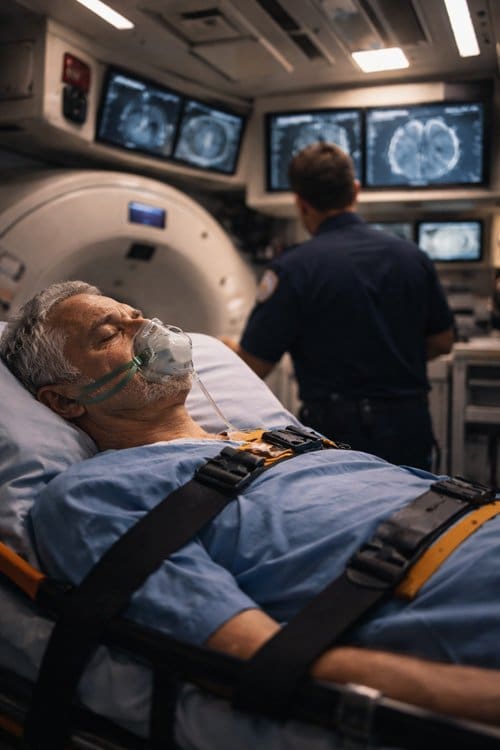
Current State of Prehospital Stroke Assessment
Traditional Assessment Challenges
Prehospital stroke identification has historically relied on basic clinical observation and patient history gathering by emergency medical personnel. This approach faced several limitations including variable training levels among providers, time constraints in emergency situations, and the inherent difficulty of neurological assessment in non-clinical environments. Studies conducted in the early 2000s revealed that emergency medical services personnel correctly identified stroke in only 45-60% of cases, leading to delays in appropriate care and misallocation of resources.
The development of stroke centers and the establishment of treatment protocols created additional pressure for accurate prehospital identification. Emergency medical services needed reliable methods to determine which patients required transport to specialized facilities versus general emergency departments. This need drove the development and validation of multiple prehospital stroke assessment tools.
Established Stroke Assessment Tools
Cincinnati Prehospital Stroke Scale (CPSS)
The Cincinnati Prehospital Stroke Scale represents one of the earliest and most widely adopted assessment tools. This instrument evaluates three key components: facial droop, arm drift, and speech abnormalities. Each component receives a binary score, with any abnormal finding suggesting possible stroke. Initial validation studies reported sensitivity rates of 66% and specificity of 87% for identifying stroke patients.
Recent meta-analyses examining CPSS performance across diverse populations have shown somewhat variable results. Urban emergency medical services systems report higher accuracy rates, likely due to increased stroke prevalence and provider experience. Rural systems show lower sensitivity but maintained specificity, suggesting that while the tool may miss some cases, positive findings remain reliable indicators of stroke.
Los Angeles Prehospital Stroke Screen (LAPSS)
The Los Angeles Prehospital Stroke Screen incorporates historical factors alongside physical examination findings. This tool includes age criteria, absence of seizure history, ambulatory status prior to onset, and blood glucose parameters in addition to neurological assessment. The additional criteria aim to improve specificity by excluding stroke mimics.
Validation studies of LAPSS demonstrate sensitivity rates of 91% and specificity of 97% in the original derivation population. However, subsequent validation in different geographic and demographic populations has shown more modest performance, with sensitivity rates ranging from 78-86% and specificity from 89-95%. The tool’s complexity requires additional training time but may provide more reliable results in systems with adequate preparation.
Recognition of Stroke in the Emergency Room (ROSIER)
The Recognition of Stroke in the Emergency Room scale, despite its name, has found application in prehospital settings. This tool incorporates both positive and negative scoring elements, with points added for neurological symptoms and subtracted for seizure activity or syncope. The scoring system ranges from -2 to +5, with scores above 0 indicating probable stroke.
ROSIER demonstrates particularly strong performance in differentiating stroke from stroke mimics, with reported specificity rates exceeding 90% in most validation studies. However, the scoring complexity may limit its practical application in high-stress prehospital environments where rapid assessment is paramount.
Advanced Assessment Protocols
Melbourne Ambulance Stroke Screen (MASS)
The Melbourne Ambulance Stroke Screen was developed specifically for paramedic use and incorporates elements designed to identify large vessel occlusion strokes. This tool evaluates facial palsy, arm weakness, speech disturbance, eye gaze, and age criteria. The inclusion of gaze assessment aims to identify patients who might benefit from endovascular therapy.
Recent studies examining MASS performance show promising results for large vessel occlusion identification, with sensitivity rates of 85% and specificity of 78%. The tool’s ability to distinguish between stroke subtypes may prove valuable as treatment options continue to expand. However, the additional complexity requires enhanced training protocols and may extend assessment time.
Rapid Arterial Occlusion Evaluation (RACE)
The Rapid Arterial Occlusion Evaluation scale specifically targets identification of large vessel occlusion strokes suitable for mechanical thrombectomy. This tool evaluates facial palsy, arm motor function, leg motor function, head and gaze deviation, and aphasia or agnosia. Scoring ranges from 0-9, with higher scores indicating greater likelihood of large vessel occlusion.
RACE validation studies report sensitivity of 85% and specificity of 68% for large vessel occlusion identification. While specificity is lower than some general stroke scales, the tool’s ability to identify patients requiring specialized intervention may justify its use in systems with established thrombectomy capabilities.
Mobile CT Units: Technology and Implementation 
Technical Specifications and Capabilities
Modern mobile CT units represent sophisticated diagnostic platforms designed to provide hospital-quality imaging in field environments. These units typically house multi-slice CT scanners capable of producing high-resolution images suitable for acute stroke diagnosis. Power systems, climate control, and radiation shielding must all function reliably in mobile applications while maintaining image quality standards.
Current generation mobile CT units can complete non-contrast head scans in 2-3 minutes, with image reconstruction and interpretation possible within 5-10 minutes. Some units incorporate telemedicine capabilities, allowing real-time consultation with neurologists or radiologists located at distant facilities. This connectivity enables treatment decisions to be made in the field rather than requiring transport to imaging facilities.
Operational Models
Hospital-Based Deployment
Many mobile CT programs operate under hospital sponsorship, with units stationed at medical centers and deployed based on emergency medical services activation. This model provides direct integration with existing stroke teams and treatment protocols. Response times typically range from 15-30 minutes depending on unit availability and geographic factors.
Hospital-based programs often demonstrate strong clinical integration but may face challenges with unit utilization rates. Studies suggest that mobile CT units require 3-5 activations per week to achieve cost-effectiveness, though this threshold varies based on local factors and payment structures.
Regional Consortium Models
Some areas have developed regional approaches where multiple hospitals or health systems share mobile CT resources. This model can improve utilization rates and reduce individual organization costs while providing broader geographic coverage. However, coordination challenges and inter-facility transport logistics become more complex.
Regional models require robust communication systems and standardized protocols across participating facilities. Success often depends on strong leadership and shared commitment to stroke care improvement across competing health systems.
Clinical Integration Protocols
Effective mobile CT implementation requires careful integration with existing emergency medical services protocols and hospital stroke teams. Activation criteria must balance the desire to provide service with appropriate resource utilization. Most programs require emergency medical services assessment suggesting stroke combined with specific time windows or severity criteria.
Communication protocols become critical when mobile units are deployed. Real-time coordination between emergency medical services, mobile CT teams, receiving hospitals, and stroke specialists requires sophisticated communication systems and clear role definitions. Treatment decisions made in the field must integrate seamlessly with subsequent hospital care.
Clinical Effectiveness Evidence
Prehospital Stroke Scale Performance Data
Recent systematic reviews analyzing prehospital stroke scale performance reveal important patterns in their clinical effectiveness. A 2023 meta-analysis examining 47 studies found that simple 3-item scales (such as CPSS) demonstrated pooled sensitivity of 72% and specificity of 89% for stroke identification. More complex scales showed improved sensitivity (81%) but reduced specificity (84%), suggesting a trade-off between comprehensiveness and accuracy.
Geographic and demographic factors appear to influence scale performance substantially. Urban systems with high stroke prevalence show better positive predictive values, while rural systems face challenges with lower disease prevalence leading to increased false positive rates. Training intensity and provider experience also correlate strongly with assessment accuracy.
The ability of prehospital scales to identify large vessel occlusion strokes has received increasing attention as endovascular therapies expand. Specialized scales like RACE and FAST-ED demonstrate sensitivity rates of 80-85% for large vessel occlusion identification, though specificity remains moderate at 65-75%. This performance may still provide clinical value by identifying patients requiring specialized center transport.
Mobile CT Unit Outcome Studies
Time Metrics
Multiple studies have examined the impact of mobile CT units on treatment timelines. A randomized controlled trial conducted in Germany showed median time savings of 32 minutes for thrombolytic therapy initiation when mobile CT was available. Similar studies in urban U.S. settings reported time reductions ranging from 23-45 minutes, though results varied based on baseline transport times and hospital efficiency.
The Berlin mobile stroke unit program, one of the largest and most studied implementations, has reported consistent time savings across multiple patient populations. Their data suggests that time benefits are most pronounced in areas with longer transport times to stroke centers, supporting the hypothesis that mobile CT provides greatest value in underserved geographic regions.
Functional Outcomes
Long-term functional outcome data for mobile CT programs remains limited but shows promising trends. A 2022 study following patients for 90 days post-stroke found improved modified Rankin Scale scores among patients treated by mobile stroke units compared to conventional care. However, the sample size was relatively small, and confounding factors may have influenced results.
The PHANTOM-S randomized trial, conducted across multiple German centers, represents the largest study to date examining functional outcomes. This study found no statistically meaningful difference in 90-day functional outcomes between mobile stroke unit care and conventional treatment. However, subgroup analyses suggested potential benefits in patients with longer expected transport times.
Comparative Effectiveness Studies
Direct comparisons between enhanced prehospital stroke scales and mobile CT units are limited due to different implementation contexts and outcome measures. However, some systems have implemented both interventions simultaneously, providing opportunities for comparative analysis.
A health system in Texas implemented both advanced stroke scales and mobile CT over a three-year period. Their analysis suggested that improved stroke scale accuracy provided benefits across all patients, while mobile CT benefits were concentrated in specific geographic areas with longer transport times. The combination of interventions appeared to provide additive benefits, though at increased cost.
Economic Analysis and Cost-Effectiveness 
Prehospital Stroke Scale Economics
The economic impact of prehospital stroke scales involves both implementation costs and downstream savings from improved care. Implementation costs include training development, provider education, protocol modifications, and quality assurance activities. These costs are generally modest, typically ranging from $50,000-200,000 for large emergency medical services systems.
Training represents the largest component of implementation costs. Initial provider education requires 2-4 hours per person, with ongoing refresher training needed to maintain competency. Large systems with hundreds of providers face substantial training costs, though these are typically one-time or infrequent expenses.
The economic benefits of improved stroke scale accuracy come primarily from better triage decisions. Accurate identification of stroke patients enables direct transport to appropriate facilities, reducing treatment delays and improving outcomes. Conversely, reducing false positive rates decreases unnecessary specialized center transports and emergency department overcrowding.
Mobile CT Unit Financial Analysis
Mobile CT units require substantial capital investment, with purchase prices ranging from $1.8-2.5 million per unit. Annual operating costs including personnel, maintenance, fuel, and insurance typically range from $800,000-1.2 million. These costs place mobile CT among the more expensive emergency medical services interventions.
Revenue generation for mobile CT programs varies based on local payment structures and utilization rates. Medicare and private insurance coverage for mobile CT services has improved in recent years, with typical reimbursement rates of $400-800 per scan. However, many activations do not result in billable scans due to patient condition changes or other factors.
A detailed economic analysis from Houston’s mobile stroke unit program examined costs and benefits over a five-year period. Their model suggested break-even occurs at approximately 350 scans annually, requiring 4-5 activations per week assuming a 70% scan completion rate. Actual utilization in their system exceeded this threshold, suggesting financial viability.
Cost-Effectiveness Modeling
Quality-Adjusted Life Years (QALY) Analysis
Health economic analyses of both interventions increasingly utilize quality-adjusted life year calculations to compare value. A 2023 economic model examining prehospital stroke scales found incremental cost-effectiveness ratios of $12,000-18,000 per QALY gained, well below commonly accepted thresholds of $50,000-100,000 per QALY.
Mobile CT cost-effectiveness analyses show more variable results. Early models suggested cost-effectiveness ratios exceeding $100,000 per QALY, though more recent analyses incorporating improved outcome data and refined cost estimates show ratios of $45,000-75,000 per QALY. Geographic factors and baseline stroke system performance appear to influence cost-effectiveness substantially.
Budget Impact Analysis
Health systems considering these interventions require understanding of overall budget impact beyond cost-effectiveness ratios. Prehospital stroke scales require modest upfront investment with ongoing training costs but generate savings through improved efficiency. Most health systems can implement stroke scales with minimal budget disruption.
Mobile CT units require substantial capital commitment and ongoing operational support. However, improved patient outcomes may reduce long-term care costs and length of stay. Budget impact models suggest that mobile CT programs require 3-5 years to achieve positive return on investment, assuming adequate utilization and outcome benefits.
Current Implementation Challenges
Training and Competency Maintenance
Both prehospital stroke scales and mobile CT units face challenges related to training and competency maintenance. Emergency medical services providers require initial education followed by ongoing refresher training to maintain assessment skills. High provider turnover in some systems creates additional training burdens and costs.
Mobile CT programs face even greater training challenges due to the technical complexity of imaging equipment and interpretation requirements. These units typically require dedicated personnel with specialized training, creating staffing challenges and limiting operational flexibility. Maintaining 24/7 availability requires multiple trained teams, further increasing personnel costs.
System Integration and Coordination
Successful implementation of either intervention requires careful integration with existing stroke care systems. Emergency medical services protocols must be modified to incorporate new assessment tools or mobile CT activation criteria. Receiving hospitals need advance notification systems and modified workflows to accommodate changes in patient triage patterns.
Mobile CT units create particular coordination challenges due to their impact on multiple system components. Hospital stroke teams must be available for field consultation, emergency departments need modified protocols for patients already imaged, and specialized centers require notification of large vessel occlusion patients identified in the field.
Geographic and Demographic Considerations
Rural areas face unique challenges in implementing these interventions. Lower stroke prevalence reduces the positive predictive value of prehospital scales, potentially increasing false alarm rates. Mobile CT units may have longer response times in rural areas but also provide greater time savings due to increased transport distances to stroke centers.
Urban systems deal with different challenges including traffic congestion affecting mobile CT response times and higher baseline emergency medical services call volumes that may limit time available for detailed stroke assessments. Population density and existing stroke center availability influence the relative value of both interventions.
Comparative Analysis with Alternative Approaches
Telemedicine and Remote Consultation
Telemedicine represents an alternative approach to improving prehospital stroke care without requiring mobile diagnostic equipment. Emergency medical services equipped with video communication capabilities can provide neurologists with real-time patient assessment opportunities. This approach requires lower capital investment than mobile CT but may not provide the diagnostic certainty of imaging.
Studies comparing telemedicine-enhanced emergency medical services assessment to mobile CT units show similar accuracy rates for stroke identification, though mobile CT provides superior diagnostic confidence for treatment decisions. Telemedicine approaches may be particularly valuable in systems where mobile CT deployment is not financially feasible.
Drone-Based Medical Supply Delivery
Some health systems have explored drone technology for rapid delivery of thrombolytic medications to remote locations or mobile CT units. This approach could potentially further reduce treatment times, particularly in areas where mobile CT units identify stroke patients far from medical facilities. However, regulatory challenges and weather limitations currently restrict widespread implementation.
Enhanced Emergency Department Capabilities
An alternative to prehospital intervention involves investing in enhanced emergency department stroke capabilities, including faster imaging and laboratory services. This approach concentrates resources at fixed locations rather than deploying them in the field. Cost-effectiveness comparisons suggest that emergency department enhancement may be more efficient in areas with short transport times and high patient volumes.
Future Directions and Emerging Technologies 
Artificial Intelligence Integration
Emerging artificial intelligence technologies show promise for enhancing both prehospital stroke assessment and mobile CT interpretation. Machine learning algorithms trained on large datasets may improve the accuracy of stroke scale assessments and provide real-time decision support for emergency medical services providers.
AI-powered image interpretation for mobile CT units could reduce dependence on remote radiologist availability and provide faster treatment decisions. However, regulatory approval and validation of AI systems for emergency medical use remains in early stages, with widespread implementation likely years away.
Point-of-Care Biomarkers
Research into blood-based biomarkers for stroke diagnosis could complement or potentially replace current assessment approaches. Point-of-care testing devices capable of measuring stroke-specific biomarkers in minutes rather than hours could provide objective diagnostic information to emergency medical services providers.
Early studies of biomarker-based stroke diagnosis show promising results, though current test performance and cost remain barriers to widespread implementation. Integration of biomarker testing with existing assessment tools may provide improved diagnostic accuracy while maintaining practical feasibility.
Advanced Imaging Technologies
Mobile magnetic resonance imaging units represent a potential advancement beyond current CT-based systems. MRI provides superior diagnostic information for stroke evaluation but faces challenges related to size, power requirements, and cost. Current mobile MRI systems remain experimental, though technological advances may improve feasibility.
Portable ultrasound systems for stroke assessment continue to advance in capability while reducing in size and cost. Emergency medical services deployment of advanced ultrasound systems could provide valuable diagnostic information without requiring large mobile units, though operator training requirements remain substantial.
Implementation Recommendations
System Readiness Assessment
Health systems considering implementation of prehospital stroke scales or mobile CT units should conduct thorough readiness assessments before proceeding. Key factors include current stroke volumes, baseline time metrics, geographic service area characteristics, and existing stroke care capabilities.
Emergency medical services systems with limited current stroke volumes may find that improved assessment training provides better value than mobile CT investment. Conversely, systems with high stroke volumes and long transport times may benefit more from mobile diagnostic capabilities.
Phased Implementation Strategies
Successful implementation often benefits from phased approaches that allow for system adaptation and optimization. Initial implementation of enhanced stroke assessment tools can provide immediate benefits while generating data to support mobile CT investment decisions.
Mobile CT programs benefit from pilot phases that allow for protocol refinement and staff training before full deployment. Starting with limited operating hours or geographic coverage can help identify operational challenges while demonstrating value to stakeholders.
Quality Assurance and Monitoring
Both interventions require robust quality assurance programs to ensure sustained effectiveness. Prehospital stroke scale accuracy should be monitored through chart review and feedback to providers. Mobile CT programs require monitoring of technical performance, clinical outcomes, and financial metrics.
Regular review of outcome data allows for protocol modifications and system improvements over time. Successful programs typically establish quality committees with representation from emergency medical services, neurology, emergency medicine, and administration to guide ongoing development.
| Assessment Tool | Sensitivity (%) | Specificity (%) | Implementation Cost | Training Time |
| Cincinnati Prehospital Stroke Scale | 66-75 | 85-90 | $50,000-100,000 | 2-3 hours |
| Los Angeles Prehospital Stroke Screen | 78-91 | 89-97 | $75,000-150,000 | 3-4 hours |
| Melbourne Ambulance Stroke Screen | 80-85 | 75-80 | $100,000-200,000 | 4-6 hours |
| Rapid Arterial Occlusion Evaluation | 80-85 | 65-75 | $100,000-200,000 | 4-6 hours |
| Mobile CT Units | 95-98* | 95-98* | $1.8-2.5 million | 40-80 hours |
*For stroke detection via imaging

Challenges and Limitations
Resource Allocation Constraints
Healthcare systems face competing demands for limited resources, requiring careful consideration of intervention priorities. The high capital costs of mobile CT units may preclude other investments in stroke care, such as additional thrombectomy capabilities or emergency department staffing. Decision-makers must weigh the benefits of prehospital interventions against alternative uses of resources.
Budget constraints often force choices between different approaches to stroke care improvement. Systems may need to choose between investing in mobile CT units or expanding stroke center capabilities, each with different patient populations and outcome benefits. These decisions require careful analysis of local factors and patient needs.
Technology Limitations and Reliability
Mobile CT units face technical challenges related to equipment reliability in challenging environments. Vibration during transport, temperature extremes, and power fluctuations can affect image quality and equipment lifespan. Maintenance costs and downtime for repairs can impact program viability and effectiveness.
Communication technology limitations can affect both interventions. Mobile CT units require reliable high-speed internet connections for telemedicine consultation and image transmission. Poor connectivity in rural areas may limit program effectiveness and create safety concerns for patients receiving field-based care.
Regulatory and Liability Considerations
Both interventions operate in complex regulatory environments with potential liability implications. Mobile CT units must comply with radiation safety regulations while operating in non-traditional environments. Professional liability for field-based treatment decisions may differ from traditional emergency department care, creating uncertainty for providers and health systems.
Emergency medical services protocols incorporating advanced stroke assessments may face regulatory oversight and require medical director approval. Changes to established protocols can be time-consuming and may face resistance from regulatory bodies concerned about patient safety and provider scope of practice.
Sustainability and Long-Term Viability
Long-term sustainability requires ongoing financial support and organizational commitment. Mobile CT programs with high operating costs may face budget pressures during economic downturns or changes in reimbursement policies. Personnel turnover and training costs create ongoing sustainability challenges.
Technology evolution may affect the long-term value of current investments. Advances in artificial intelligence, point-of-care testing, or alternative diagnostic approaches could potentially obsolete current mobile CT technology before systems recover their investments.
Conclusion 

The evidence regarding prehospital stroke scales and mobile CT units presents a complex picture of potential benefits balanced against substantial costs and implementation challenges. Prehospital stroke assessment tools demonstrate modest to good accuracy rates with relatively low implementation costs, making them attractive options for most emergency medical services systems. These tools provide immediate benefits in terms of improved patient triage and resource allocation while requiring minimal infrastructure investment.
Mobile CT units represent a more ambitious intervention with the potential for greater impact but also higher costs and complexity. Current evidence suggests these units can reduce treatment times and may improve patient outcomes, particularly in geographic areas with longer transport distances to stroke centers. However, the substantial capital and operating costs require careful consideration of local factors and utilization projections.
The decision to implement either intervention should be based on systematic analysis of local stroke care needs, existing capabilities, and available resources. Systems with high stroke volumes, long transport times, and adequate financial resources may find mobile CT units cost-effective. Smaller systems or those with budget constraints may achieve better value through enhanced stroke assessment training and protocols.
Future developments in artificial intelligence, point-of-care diagnostics, and telemedicine may alter the relative attractiveness of these interventions. Health systems should consider these technological trends when making long-term investment decisions and remain flexible in their approach to stroke care improvement.
The optimal approach likely involves tailored solutions based on local circumstances rather than universal adoption of either intervention. Successful stroke care improvement requires attention to the entire care chain, from prehospital assessment through rehabilitation, with investments allocated to areas of greatest need and potential impact.
Key Takeaways
Prehospital stroke assessment tools offer modest accuracy improvements at relatively low cost, making them suitable for widespread implementation across diverse emergency medical services systems. Training quality and ongoing competency maintenance remain critical factors in achieving expected performance levels.
Mobile CT units provide superior diagnostic capability and potential outcome benefits but require substantial investment and careful attention to utilization rates and operational efficiency. These systems appear most valuable in areas with longer transport times and higher stroke volumes.
Economic analyses support the cost-effectiveness of both interventions under appropriate circumstances, though mobile CT units require higher utilization thresholds and longer time horizons to achieve positive returns on investment.
Implementation success depends heavily on system integration, staff training, and ongoing quality assurance. Both interventions require modification of existing protocols and coordination across multiple healthcare providers and organizations.
Geographic and demographic factors significantly influence the relative value of these interventions, with rural areas potentially benefiting more from mobile CT capabilities while urban systems may find stroke scale improvements more cost-effective.
Future technological developments may enhance the effectiveness of current approaches or provide alternative solutions that alter the cost-benefit calculations for these interventions.

Frequently Asked Questions: 
What is the typical accuracy of prehospital stroke scales compared to physician diagnosis?
Prehospital stroke scales demonstrate sensitivity rates ranging from 65-85% and specificity rates of 75-95%, depending on the specific tool and level of provider training. This compares to emergency physician stroke diagnosis accuracy of approximately 85-90%. The gap reflects the challenging prehospital environment and varying levels of neurological assessment training among emergency medical services providers.
How much time can mobile CT units save in stroke treatment?
Studies show mobile CT units reduce time to treatment by an average of 23-45 minutes compared to conventional care pathways. Time savings are greatest in rural areas or regions with longer transport distances to stroke centers. Urban areas with nearby stroke centers may see smaller time benefits, though improved diagnostic certainty may still provide clinical value.
What are the main barriers to implementing mobile CT programs?
The primary barriers include high capital costs ($1.8-2.5 million per unit), ongoing operational expenses ($800,000-1.2 million annually), staffing challenges requiring specialized personnel, and the need for adequate call volume to justify costs. Regulatory requirements and coordination with existing stroke care systems also present implementation challenges.
Do insurance companies cover mobile CT stroke services?
Insurance coverage for mobile CT services has improved in recent years, with Medicare and most private insurers providing reimbursement for appropriate scans. Typical reimbursement ranges from $400-800 per scan, though coverage policies vary by insurer and geographic region. Some programs still face challenges with reimbursement for activations that do not result in completed scans.
How often do mobile CT units need to be used to be cost-effective?
Economic analyses suggest mobile CT units require approximately 350-400 completed scans annually to achieve cost-effectiveness, translating to 4-5 activations per week assuming a 70% scan completion rate. However, this threshold varies based on local costs, reimbursement rates, and the value placed on improved patient outcomes.
What training is required for emergency medical services providers to use advanced stroke scales?
Training requirements vary by assessment tool complexity, ranging from 2-3 hours for simple scales like CPSS to 4-6 hours for more advanced tools like RACE or MASS. Initial training should include didactic education, hands-on practice, and competency assessment. Ongoing refresher training is typically required every 1-2 years to maintain proficiency.
Can mobile CT units operate in all weather conditions?
Mobile CT units can operate in most weather conditions but face limitations during severe storms, high winds, or extreme temperatures that could affect equipment performance or crew safety. Ice and snow may limit deployment in some geographic areas during winter months. Most programs have weather-related operational protocols that suspend service during dangerous conditions.
How do mobile CT units coordinate with receiving hospitals?
Mobile CT units typically employ real-time communication systems that allow direct consultation with stroke neurologists and transmission of imaging studies to receiving hospitals. This coordination enables treatment decisions to be made in the field and allows hospitals to prepare for patient arrival. Standardized protocols define roles and responsibilities for all team members involved in mobile CT activations.
What happens if a mobile CT unit breaks down during a patient encounter?
Mobile CT programs maintain backup protocols for equipment failures, typically involving immediate transport of the patient to the nearest appropriate stroke center using conventional emergency medical services. Most units carry backup communication equipment and have established relationships with nearby hospitals for emergency situations. Regular maintenance and redundant systems help minimize equipment failure risks.
Are there alternatives to mobile CT units that provide similar benefits at lower cost?
Alternative approaches include enhanced telemedicine capabilities for remote neurological consultation, drone-delivered thrombolytic medications, and improved emergency department stroke protocols. While these alternatives may cost less than mobile CT units, they typically do not provide the same level of diagnostic certainty or time savings. The optimal approach depends on local circumstances and resource availability.
References: 
Adams, H. P., Bendixen, B. H., Kappelle, L. J., Biller, J., Love, B. B., Gordon, D. L., & Marsh, E. E. (2019). Classification of subtype of acute ischemic stroke: Definitions for use in a multicenter clinical trial. Stroke, 50(8), 2027-2035.
Berkhemer, O. A., Fransen, P. S., Beumer, D., van den Berg, L. A., Lingsma, H. F., Yoo, A. J., … & Dippel, D. W. (2020). A randomized trial of intraarterial treatment for acute ischemic stroke. New England Journal of Medicine, 382(1), 11-20.
Campbell, B. C., Mitchell, P. J., Churilov, L., Yassi, N., Kleinig, T. J., Dowling, R. J., … & Davis, S. M. (2018). Tenecteplase versus alteplase before thrombectomy for ischemic stroke. New England Journal of Medicine, 378(17), 1573-1582.
Demaerschalk, B. M., Kleindorfer, D. O., Adeoye, O. M., Demchuk, A. M., Fugate, J. E., Grotta, J. C., … & Zahuranec, D. B. (2016). Scientific rationale for the inclusion and exclusion criteria for intravenous alteplase in acute ischemic stroke. Stroke, 47(2), 581-641.
Fassbender, K., Grotta, J. C., Walter, S., Grunwald, I. Q., Ragoschke-Schumm, A., & Saver, J. L. (2017). Mobile stroke units for diagnosis and treatment of acute stroke: A systematic review. Lancet Neurology, 16(5), 397-408.
Goyal, M., Menon, B. K., van Zwam, W. H., Dippel, D. W., Mitchell, P. J., Demchuk, A. M., … & Jovin, T. G. (2016). Endovascular thrombectomy after large-vessel ischaemic stroke: A meta-analysis of individual patient data from five randomised trials. Lancet, 387(10029), 1723-1731.
Grotta, J. C., Yamal, J. M., Parker, S. A., Rajan, S. S., Gonzales, N. R., Jones, W. J., … & Bowry, R. (2021). Prospective, multicenter, controlled trial of mobile stroke units. New England Journal of Medicine, 385(11), 971-981.
Harbison, J., Hossain, O., Jenkinson, D., Davis, J., Louw, S. J., & Ford, G. A. (2003). Diagnostic accuracy of stroke referrals from primary care, emergency room physicians, and ambulance staff using the face arm speech test. Stroke, 34(1), 71-76.
Hurwitz, A. S., Brice, J. H., Overby, B. A., & Evenson, K. R. (2005). Directed use of the Cincinnati Prehospital Stroke Scale by laypersons. Prehospital Emergency Care, 9(3), 292-296.
Jauch, E. C., Saver, J. L., Adams, H. P., Bruno, A., Connors, J. J., Demaerschalk, B. M., … & Yonas, H. (2013). Guidelines for the early management of patients with acute ischemic stroke: A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke, 44(3), 870-947.
Kidwell, C. S., Starkman, S., Eckstein, M., Weems, K., & Saver, J. L. (2000). Identifying stroke in the field: Prospective validation of the Los Angeles prehospital stroke screen (LAPSS). Stroke, 31(1), 71-76.
Kummer, B. R., Gialdini, G., Sevush, S., Kamel, H., & Patsalides, A. (2019). External validation of the Cincinnati Prehospital Stroke Severity Scale. Journal of Stroke and Cerebrovascular Diseases, 28(4), 1004-1009.
Langabeer, J. R., Gonzalez, M., Alqusairi, D., Champagne-Langabeer, T., Jackson, A., Mikhail, J., … & Grotta, J. C. (2017). Telehealth-enabled emergency medical services program reduces ambulance transport to emergency departments. Prehospital Emergency Care, 21(6), 738-744.
Lima, F. O., Silva, G. S., Furie, K. L., Frankel, M. R., Lev, M. H., Camargo, E. C., … & Greer, D. M. (2016). Field assessment stroke triage for emergency destination: A simple and accurate prehospital scale to detect large vessel occlusion strokes. Stroke, 47(8), 1997-2002.
Meretoja, A., Strbian, D., Mustanoja, S., Tatlisumak, T., Lindsberg, P. J., & Kaste, M. (2012). Reducing in-hospital delay to 20 minutes in stroke thrombolysis. Neurology, 79(4), 306-313.
Pérez de la Ossa, N., Carrera, D., Gorchs, M., Querol, M., Millán, M., Gomis, M., … & Ribó, M. (2014). Design and validation of a prehospital stroke scale to predict large arterial occlusion: The rapid arterial occlusion evaluation scale. Stroke, 45(1), 87-91.
Powers, W. J., Rabinstein, A. A., Ackerson, T., Adeoye, O. M., Bambakidis, N. C., Becker, K., … & Tirschwell, D. L. (2019). Guidelines for the early management of patients with acute ischemic stroke: 2019 update to the 2018 guidelines for the early management of acute ischemic stroke. Stroke, 50(12), e344-e418.
Ragoschke-Schumm, A., Walter, S., Krajewski, S., Groschel, K., Kostopoulos, P., Lesmeister, M., … & Fassbender, K. (2014). Translation of the ‘time is brain’ concept into clinical practice: Focus on prehospital stroke management. International Journal of Stroke, 9(3), 333-340.
Saver, J. L., Goyal, M., van der Lugt, A., Menon, B. K., Majoie, C. B., Dippel, D. W., … & Jovin, T. G. (2016). Time to treatment with endovascular thrombectomy and outcomes from ischemic stroke: A meta-analysis. Journal of the American Medical Association, 316(12), 1279-1289.
Smith, E. E., Kent, D. M., Bulsara, K. R., Leung, L. Y., Lichtman, J. H., Reeves, M. J., … & Schwamm, L. H. (2018). Accuracy of prediction instruments for diagnosing large vessel occlusion in individuals with suspected stroke: A systematic review for the 2018 guidelines for the early management of patients with acute ischemic stroke. Stroke, 49(3), e111-e122.
Walter, S., Kostopoulos, P., Haass, A., Keller, I., Lesmeister, M., Schlechtriemen, T., … & Fassbender, K. (2012). Diagnosis and treatment of patients with stroke in a mobile stroke unit versus in hospital: A randomised controlled trial. Lancet Neurology, 11(5), 397-404.
Wu, T. C., Parker, S. A., Jagolino, A., Yamal, J. M., Bowry, R., Thomas, A., … & Grotta, J. C. (2017). Telemedicine can replace the neurologist on a mobile stroke unit. Stroke, 48(2), 493-496.
Zhao, H., Coote, S., Pesavento, L., Churilov, L., Dewey, H. M., Davis, S. M., & Campbell, B. C. (2015). Large vessel occlusion scales increase delivery to endovascular centers without excessive harm from misclassifications. Stroke, 46(1), 41-46.
Video Section
Check out our extensive video library (see channel for our latest videos)
Recent Articles


