Chronic Kidney Disease in the Non-Diabetic Patient: What’s New and What Works?

Abstract
Chronic kidney disease (CKD) represents a major global health challenge, affecting more than 850 million people worldwide and contributing substantially to cardiovascular morbidity, premature mortality, and escalating healthcare costs. Although diabetic kidney disease remains the most recognized contributor to CKD burden, non-diabetic causes account for approximately 40 to 50 percent of all cases, making them a critical focus of nephrology practice. These non-diabetic etiologies encompass a heterogeneous group of disorders with distinct pathophysiological mechanisms, rates of progression, and therapeutic considerations. Effective management therefore requires both precise etiological assessment and individualized long term care strategies aimed at preserving renal function and reducing systemic complications.
This review examines current evidence based approaches to the diagnosis and management of non-diabetic CKD, with particular attention to recent advances in disease mechanisms, early detection, and therapeutic intervention. It integrates contemporary literature on common primary causes including hypertensive nephrosclerosis, glomerular diseases, hereditary nephropathies such as autosomal dominant polycystic kidney disease, and medication related renal injury. By combining established treatment principles with emerging diagnostic and therapeutic developments, the review aims to provide practical guidance for clinicians involved in the care of patients with chronic non-diabetic renal disease.
Among non-diabetic causes, hypertensive nephrosclerosis remains one of the most prevalent contributors to CKD, particularly in aging populations and individuals with long standing uncontrolled hypertension. Persistent systemic hypertension leads to progressive vascular remodeling, glomerular ischemia, and tubulointerstitial fibrosis, ultimately reducing nephron function over time. Clinical management focuses on strict blood pressure control, typically targeting levels guided by cardiovascular and renal risk profiles. Renin angiotensin system inhibition remains central to therapy because of its demonstrated ability to reduce intraglomerular pressure, lower proteinuria, and slow progression of renal injury. Angiotensin converting enzyme inhibitors and angiotensin receptor blockers continue to form the foundation of treatment, particularly in patients with albuminuria.
Glomerulonephritis represents another major category of non-diabetic CKD and includes a wide spectrum of immune mediated disorders such as IgA nephropathy, membranous nephropathy, focal segmental glomerulosclerosis, and lupus related renal disease. These conditions often require histopathological diagnosis through kidney biopsy, as therapeutic decisions depend heavily on underlying lesion type and degree of inflammatory activity. Recent advances in immunopathology and molecular classification have improved understanding of disease pathways, enabling more targeted use of corticosteroids, calcineurin inhibitors, antiproliferative agents, and biologic therapies in selected cases. In parallel, supportive care remains essential, including blood pressure optimization, proteinuria reduction, and cardiovascular risk management.
Autosomal dominant polycystic kidney disease continues to be one of the most important hereditary causes of CKD worldwide. Characterized by progressive cyst formation and enlargement, the disease leads to gradual destruction of normal renal architecture and eventual decline in glomerular filtration rate. Advances in imaging now allow earlier diagnosis and more accurate prognostic stratification based on total kidney volume and cyst growth rates. Therapeutic developments, particularly vasopressin receptor antagonists such as tolvaptan, have introduced disease modifying options capable of slowing cyst progression in carefully selected patients. Management also includes aggressive blood pressure control, surveillance for extra renal complications, and patient counseling regarding long term renal replacement planning.
Drug induced nephropathy remains an increasingly relevant contributor to chronic renal injury, particularly in populations exposed to long term analgesics, nephrotoxic antibiotics, proton pump inhibitors, chemotherapeutic agents, and herbal preparations. Medication review is therefore a critical component of CKD evaluation. Identification of potentially reversible nephrotoxic exposure can alter disease trajectory when addressed early. Clinicians must maintain vigilance regarding cumulative toxicity, especially in patients with preexisting renal vulnerability or multiple comorbidities.
Recent progress in biomarker research has improved the ability to detect kidney injury earlier than traditional measures alone. Although serum creatinine and estimated glomerular filtration rate remain standard tools for CKD diagnosis and staging, newer markers such as cystatin C, urinary kidney injury molecule 1, neutrophil gelatinase associated lipocalin, and albuminuria profiling offer additional insights into renal damage and progression risk. These biomarkers may improve diagnostic sensitivity, particularly in early disease stages where conventional parameters remain within normal ranges.
Imaging techniques have also advanced significantly in the evaluation of non-diabetic CKD. Renal ultrasonography remains the first line imaging modality for structural assessment, but newer techniques such as magnetic resonance based renal volumetry, Doppler perfusion studies, and elastography are expanding the ability to characterize parenchymal changes and disease progression noninvasively. In hereditary and cystic diseases, imaging has become particularly important for prognostication and therapeutic planning.
Therapeutic management extends beyond disease specific treatment to comprehensive risk factor modification. Proteinuria reduction remains one of the most important targets because of its strong association with progression risk across multiple etiologies. Dietary sodium restriction, individualized protein intake, smoking cessation, weight management, and regular physical activity all contribute to renal preservation and cardiovascular protection. In addition, careful avoidance of further nephrotoxic exposure is essential throughout disease management.
Mineral and bone disorder management remains a key component of advanced CKD care. As renal function declines, disturbances in phosphate handling, vitamin D metabolism, parathyroid hormone regulation, and calcium balance contribute to bone disease and vascular calcification. Regular monitoring and appropriate use of phosphate binders, vitamin D analogues, and calcimimetic agents help reduce complications and improve long term outcomes.
Anemia management also requires attention in progressive CKD, as reduced erythropoietin production contributes to fatigue, reduced quality of life, and cardiovascular strain. Iron status assessment and judicious use of erythropoiesis stimulating agents remain important aspects of care, particularly in later disease stages.
Increasing attention has also been directed toward cardiovascular protection, as patients with non-diabetic CKD are far more likely to die from cardiovascular events than to reach end stage kidney failure. Lipid control, blood pressure optimization, and management of vascular risk factors are therefore integral to renal care rather than separate priorities.
Evidence consistently demonstrates that early identification through improved screening strategies substantially improves outcomes. High risk populations, including individuals with hypertension, autoimmune disease, family history of renal disorders, recurrent urinary abnormalities, or chronic exposure to nephrotoxic medications, benefit from regular renal assessment before substantial nephron loss occurs. Early intervention allows implementation of therapies capable of slowing progression and delaying the need for dialysis or transplantation.
In summary, non-diabetic chronic kidney disease requires a broad and integrated clinical approach that combines accurate etiological diagnosis, early detection, targeted pharmacologic intervention, and long term complication management. Advances in biomarkers, imaging, and disease specific therapies have improved the ability to individualize care, but optimal outcomes still depend heavily on timely recognition and consistent multidisciplinary management. As understanding of non-diabetic CKD continues to evolve, clinicians must integrate both established evidence and emerging tools to preserve renal function and improve patient survival.
Introduction
Chronic kidney disease is widely recognized as one of the most significant global health challenges of the twenty first century, with rising prevalence, high morbidity, and substantial healthcare costs across both developed and developing regions. Although diabetes mellitus remains the leading cause of chronic kidney disease in many high income countries, non diabetic etiologies account for a substantial proportion of kidney disease worldwide and continue to contribute notably to progression to kidney failure, cardiovascular complications, and premature mortality. For practicing clinicians, a thorough understanding of non diabetic chronic kidney disease is essential because these conditions often present with heterogeneous pathophysiological mechanisms, variable rates of progression, and distinct therapeutic priorities that differ from diabetic nephropathy.
Non diabetic chronic kidney disease encompasses a broad spectrum of disorders involving glomerular, tubulointerstitial, vascular, hereditary, and obstructive renal pathology. Common causes include hypertensive nephrosclerosis, chronic glomerulonephritis, polycystic kidney disease, chronic interstitial nephritis, autoimmune renal disease, recurrent urinary tract obstruction, and nephrotoxic injury related to medications or environmental exposure. In many parts of the world, infectious diseases, untreated hypertension, herbal nephrotoxins, and undiagnosed congenital abnormalities also remain important contributors. This diversity of underlying causes makes early diagnosis particularly important, as disease progression and management strategies depend heavily on identifying the dominant pathological process.
The definition of chronic kidney disease has evolved substantially over the past two decades as nephrology has shifted from a purely filtration based understanding toward a broader structural and functional framework. Current Kidney Disease: Improving Global Outcomes guidelines define chronic kidney disease as abnormalities of kidney structure or function that persist for more than three months and have implications for health. This definition includes a glomerular filtration rate below 60 mL per minute per 1.73 square meters, even in the absence of overt symptoms, as well as the presence of markers of kidney damage regardless of estimated filtration rate.
Markers of kidney injury extend beyond reduced glomerular filtration and include persistent albuminuria, abnormalities in urinary sediment such as hematuria or cellular casts, electrolyte disturbances attributable to tubular dysfunction, histological abnormalities identified on kidney biopsy, structural abnormalities detected by imaging, and a prior history of kidney transplantation. This expanded definition reflects the recognition that clinically meaningful kidney disease may exist even before significant loss of filtration capacity becomes apparent. It also emphasizes the importance of routine laboratory screening and imaging in patients at risk, especially those with hypertension, autoimmune disease, recurrent urinary tract pathology, or a family history of renal disease.
The clinical burden of non diabetic chronic kidney disease extends far beyond progressive loss of renal function. Cardiovascular disease remains the leading cause of death in this population, with chronic kidney dysfunction strongly associated with accelerated atherosclerosis, vascular calcification, left ventricular hypertrophy, arrhythmias, and heart failure. Even mild to moderate reductions in kidney function significantly increase cardiovascular risk, often before progression to advanced renal failure. This relationship reflects complex interactions involving inflammation, endothelial dysfunction, altered mineral metabolism, and neurohormonal activation.
Mineral and bone disorders represent another major complication. Impaired phosphate excretion, reduced calcitriol synthesis, and secondary hyperparathyroidism contribute to abnormalities in calcium phosphate homeostasis, skeletal fragility, and vascular calcification. These changes may begin early in chronic kidney disease and progress silently, making regular biochemical monitoring essential even in moderate disease stages.
Anemia is also highly prevalent in non diabetic chronic kidney disease and often develops as kidney function declines due to reduced erythropoietin production, iron dysregulation, chronic inflammation, and shortened red blood cell survival. Untreated anemia contributes to fatigue, reduced exercise tolerance, cognitive impairment, and worsening cardiovascular outcomes. Similarly, metabolic acidosis frequently emerges as renal acid excretion declines, promoting muscle catabolism, bone demineralization, and faster progression of kidney dysfunction.
Patients with chronic kidney disease also experience substantial reductions in quality of life. Fatigue, sleep disturbance, dietary restrictions, polypharmacy, anxiety regarding progression, and frequent medical monitoring all contribute to psychosocial burden. In many cases, the cumulative impact of these factors begins long before dialysis becomes necessary, reinforcing the need for holistic and anticipatory management.
Early recognition remains one of the most important determinants of long term outcome. Non diabetic chronic kidney disease often progresses silently, with many patients remaining asymptomatic until advanced stages. Serum creatinine alone may underestimate disease in early phases, particularly in older adults or individuals with reduced muscle mass. For this reason, estimated glomerular filtration rate calculations, urine albumin measurement, and blood pressure assessment remain central to early detection strategies.
Once identified, management requires both disease specific and general renoprotective interventions. Blood pressure control remains a cornerstone of therapy, particularly with agents targeting the renin angiotensin aldosterone system in proteinuric disease. Dietary sodium restriction, avoidance of nephrotoxic medications, and careful adjustment of drug dosing according to kidney function are essential across all etiologies. In selected cases, immunosuppressive therapy may be required for glomerular diseases, while hereditary and structural disorders demand targeted surveillance and supportive care.
Recent therapeutic advances have also expanded treatment options beyond traditional approaches. Although originally developed in diabetic populations, sodium glucose cotransporter 2 inhibitors have demonstrated renoprotective benefits in several forms of non diabetic chronic kidney disease, suggesting a broader role in slowing progression and reducing cardiovascular risk. Their incorporation into modern nephrology practice reflects an important shift toward mechanism based treatment rather than cause specific therapy alone.
Ultimately, non diabetic chronic kidney disease requires a comprehensive clinical approach that recognizes its systemic consequences, diverse causes, and opportunities for early intervention. As global prevalence continues to rise, physicians across specialties must be equipped to identify disease early, investigate underlying etiology appropriately, and implement strategies that preserve kidney function while reducing cardiovascular and metabolic complications. Continued advances in risk stratification, biomarker development, and targeted therapies are likely to further refine management and improve long term outcomes in this growing patient population.

Epidemiology and Risk Factors 
Non-diabetic CKD encompasses multiple distinct disease entities with varying prevalence patterns across populations. Hypertensive nephrosclerosis accounts for approximately 25-30% of end-stage renal disease cases in the United States (United States Renal Data System, 2023). Glomerulonephritis, including primary and secondary forms, contributes another 15-20% of cases. Polycystic kidney disease affects approximately 1 in 1000 individuals, making it the most common inherited kidney disease (Cornec-Le Gall et al., 2019).
Age-related kidney function decline affects all individuals, with GFR typically decreasing by 0.5-1.0 mL/min/1.73m² per year after age 40 (Denic et al., 2016). However, pathological acceleration of this process occurs in susceptible individuals. Risk factors for non-diabetic CKD include advanced age, family history of kidney disease, cardiovascular disease, hypertension, obesity, smoking, and exposure to nephrotoxic medications (Chronic Kidney Disease Prognosis Consortium, 2019).
Genetic factors play increasingly recognized roles in CKD development. Apolipoprotein L1 (APOL1) gene variants confer increased risk for hypertensive and focal segmental glomerulosclerosis in individuals of African ancestry (Freedman et al., 2018). Other genetic variants affecting complement regulation, collagen synthesis, and cellular metabolism contribute to various forms of hereditary nephritis and cystic kidney diseases.
Environmental exposures also contribute to CKD risk. Occupational exposure to heavy metals, solvents, and silica increases nephrotoxicity risk (Glassock et al., 2017). Geographic clusters of CKD of unknown etiology have been identified in agricultural regions, potentially related to pesticide exposure, heat stress, and dehydration (Wesseling et al., 2020).
Primary Causes of Non-Diabetic CKD 
Hypertensive Nephrosclerosis
Hypertensive nephrosclerosis develops through sustained elevation of blood pressure causing arteriolar hyalinosis, glomerular sclerosis, and tubulointerstitial fibrosis (Bidani & Griffin, 2004). The process typically progresses slowly over decades, making early detection challenging. Pathological changes include arterial wall thickening, luminal narrowing, and eventual nephron loss.
Clinical presentation is often subtle in early stages. Patients may have mild proteinuria, typically less than 1 gram per day, and slowly declining kidney function (Pugh et al., 2019). Urine sediment examination usually reveals minimal abnormalities. Imaging studies show bilateral kidney atrophy with cortical thinning in advanced cases.
Risk factors for hypertensive nephrosclerosis include duration and severity of hypertension, African ancestry, age, and concurrent cardiovascular disease. The condition disproportionately affects African American patients, who develop more severe kidney function decline at equivalent blood pressure levels compared to other populations (Wright et al., 2016).
Glomerulonephritis
Primary glomerulonephritis encompasses multiple distinct diseases affecting glomerular structure and function. IgA nephropathy represents the most common primary glomerulonephritis worldwide, accounting for up to 40% of primary glomerular diseases in some populations (Rodrigues et al., 2017). The condition results from deposition of galactose-deficient IgA1 immune complexes in glomerular mesangium.
Clinical presentation varies from asymptomatic hematuria to rapidly progressive glomerulonephritis. Many patients experience episodic gross hematuria coinciding with upper respiratory infections. Proteinuria ranges from minimal to nephrotic levels. Disease progression correlates with degree of proteinuria, hypertension, and histological severity (Tesar et al., 2015).
Focal segmental glomerulosclerosis (FSGS) represents another important cause of progressive CKD. Primary FSGS results from podocyte injury leading to segmental sclerosis of glomerular capillaries. Secondary forms develop from various causes including obesity, drug toxicity, and genetic mutations (D’Agati et al., 2011). Clinical presentation typically includes nephrotic syndrome with progressive kidney function decline.
Membranous nephropathy primarily affects adults and presents with nephrotic syndrome. Primary membranous nephropathy associates with anti-phospholipase A2 receptor antibodies in approximately 70% of cases (Beck et al., 2009). Secondary forms result from malignancy, infections, or autoimmune diseases. Spontaneous remission occurs in approximately one-third of patients, while others progress to end-stage renal disease.
Polycystic Kidney Disease
Autosomal dominant polycystic kidney disease (ADPKD) affects approximately 1 in 1000 individuals worldwide (Harris & Torres, 2009). The condition results from mutations in PKD1 (85% of cases) or PKD2 (15% of cases) genes, leading to defective ciliary function and abnormal cell proliferation. Progressive cyst formation and enlargement eventually compromise normal kidney architecture and function.
Clinical manifestations typically develop in adulthood, although kidney enlargement often begins in childhood. Common symptoms include flank pain, hematuria, urinary tract infections, and kidney stones (Chebib & Torres, 2016). Extrarenal manifestations include liver cysts, intracranial aneurysms, cardiac valve abnormalities, and colonic diverticulosis.
Disease progression varies considerably between individuals and correlates with genetic mutation type, kidney size, and other factors. PKD1 mutations generally cause more severe disease with earlier onset of kidney failure compared to PKD2 mutations (Cornec-Le Gall et al., 2013). Total kidney volume serves as a biomarker for disease progression and treatment response.
Drug-Induced Nephropathy
Medication-related kidney injury represents a preventable cause of CKD that affects hospitalized and outpatient populations. Nonsteroidal anti-inflammatory drugs (NSAIDs) cause both acute kidney injury and chronic tubulointerstitial nephritis through prostaglandin inhibition and direct cellular toxicity (Nderitu et al., 2013). Risk factors include advanced age, dehydration, concurrent use of other nephrotoxic medications, and pre-existing kidney disease.
Proton pump inhibitors (PPIs) have emerged as a cause of chronic interstitial nephritis and CKD progression (Lazarus et al., 2016). Large epidemiological studies demonstrate associations between PPI use and increased CKD risk, although causality remains debated. The mechanism likely involves immune-mediated interstitial inflammation.
Calcineurin inhibitors used in transplant recipients and autoimmune diseases cause chronic nephrotoxicity through arteriolar hyalinosis and tubulointerstitial fibrosis (Naesens et al., 2009). The injury is dose-dependent and often irreversible, requiring careful monitoring and dose optimization.
Diagnostic Approaches and Biomarkers 
Traditional Assessment Methods
Serum creatinine remains the primary biomarker for estimating kidney function, despite well-recognized limitations. Current estimating equations include the 2009 CKD-EPI equation, which provides more accurate GFR estimates compared to older formulas, particularly at higher GFR levels (Levey et al., 2009). However, creatinine-based estimates are influenced by muscle mass, age, sex, race, and medications affecting tubular secretion.
The 2021 CKD-EPI equation removed race-based coefficients to address concerns about perpetuating health disparities (Inker et al., 2021). This change led to reclassification of kidney function in some patients, particularly those of African ancestry, and highlighted the need for more precise biomarkers.
Urinalysis provides essential information about kidney damage patterns. Proteinuria assessment has evolved from qualitative dipstick testing to quantitative albumin-to-creatinine ratio (ACR) measurements. The KDIGO guidelines recommend using ACR rather than total protein-to-creatinine ratio for initial testing, with values above 30 mg/g indicating abnormal albuminuria (KDIGO, 2024).
Urine sediment examination offers valuable diagnostic information often overlooked in routine practice. Red blood cell morphology helps differentiate glomerular from non-glomerular bleeding. Dysmorphic red blood cells and red blood cell casts indicate glomerular disease. White blood cell casts suggest tubulointerstitial inflammation, while granular and waxy casts indicate chronic kidney damage (Fogazzi et al., 2008).
Novel Biomarkers
Cystatin C has gained acceptance as an alternative biomarker for kidney function assessment. This low-molecular-weight protein is produced by all nucleated cells at a relatively constant rate and filtered freely by glomeruli without tubular secretion or reabsorption (Shlipak et al., 2013). Cystatin C-based GFR estimates show less variation with muscle mass and may provide more accurate assessment in elderly patients and those with extremes of body composition.
Kidney injury molecule-1 (KIM-1) represents a promising biomarker for tubulointerstitial damage. KIM-1 expression increases dramatically in damaged proximal tubular cells and correlates with histological injury severity (Bonventre, 2009). Urinary KIM-1 levels predict CKD progression and may help identify patients requiring more intensive monitoring.
Neutrophil gelatinase-associated lipocalin (NGAL) serves as both an acute kidney injury biomarker and predictor of CKD progression. NGAL expression increases in response to kidney injury and correlates with inflammation and fibrosis markers (Bolignano et al., 2014). However, clinical implementation remains limited by standardization challenges and cost considerations.
Fibroblast growth factor-23 (FGF-23) regulates phosphate homeostasis and vitamin D metabolism. Elevated FGF-23 levels develop early in CKD and predict cardiovascular mortality and disease progression (Gutierrez et al., 2005). FGF-23 may serve as a more sensitive marker of mineral bone disorder than traditional parameters.
Imaging and Biopsy Considerations
Kidney imaging plays important roles in CKD evaluation, particularly for determining disease etiology and prognosis. Ultrasound provides information about kidney size, echogenicity, and structural abnormalities. Small, echogenic kidneys suggest chronic disease, while normal or enlarged kidneys may indicate acute processes or infiltrative diseases (O’Neill et al., 2000).
Magnetic resonance imaging offers superior soft tissue contrast and functional assessment capabilities. Diffusion-weighted imaging and blood oxygen level-dependent (BOLD) MRI provide information about kidney perfusion and oxygenation (Zhang et al., 2018). These techniques show promise for monitoring disease progression and treatment response but require further validation.
Kidney biopsy remains the gold standard for diagnosing specific glomerular diseases and assessing disease activity. Indications for biopsy in CKD patients include unexplained kidney function decline, proteinuria greater than 1 gram per day, active urinary sediment, or suspicion of systemic disease (Hogan et al., 2016). Contraindications include bleeding disorders, uncontrolled hypertension, and advanced chronic disease where results would not change management.
Current Treatment Strategies 
Renin-Angiotensin System Blockade
Angiotensin-converting enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs) form the cornerstone of CKD treatment for most patients. These medications reduce intraglomerular pressure, decrease proteinuria, and slow disease progression independent of blood pressure effects (Lewis et al., 2001). The KDIGO guidelines recommend ACE inhibitors or ARBs for patients with CKD and albuminuria, regardless of diabetes status.
Multiple large randomized trials demonstrate nephroprotective effects of renin-angiotensin system (RAS) blockade in non-diabetic CKD. The REIN study showed that ramipril reduced kidney disease progression in patients with non-diabetic proteinuric CKD (Gruppo Italiano di Studi Epidemiologici in Nefrologia, 1997). Similar benefits were observed with ARBs in subsequent trials.
Optimal dosing strategies remain debated. Some evidence suggests that higher doses provide greater antiproteinuric effects, but this must be balanced against risks of hyperkalemia and acute kidney injury (Hou et al., 2007). Current practice typically involves titrating doses to maximum tolerated levels while monitoring kidney function and electrolytes.
Combination ACE inhibitor and ARB therapy was previously considered for patients with persistent proteinuria, but recent evidence suggests increased adverse events without additional benefits (Fried et al., 2013). Current guidelines recommend against dual RAS blockade in most situations.
Blood Pressure Management
Blood pressure control represents a fundamental aspect of CKD management, with targets varying based on proteinuria levels and individual patient characteristics. The SPRINT trial demonstrated that intensive blood pressure control (systolic BP <120 mmHg) reduced cardiovascular events and mortality in high-risk patients, including those with CKD (Wright et al., 2015).
However, concerns exist about intensive blood pressure control in elderly patients and those with advanced CKD. The ACCORD trial showed no benefit of intensive BP control in diabetic patients, and subsequent analyses suggested potential harm in certain subgroups (Cushman et al., 2010). Current KDIGO guidelines recommend BP targets of <120/80 mmHg for patients with CKD and albuminuria, and <130/80 mmHg for those without albuminuria.
Medication selection should consider both efficacy and kidney-specific effects. ACE inhibitors and ARBs provide preferred first-line therapy. Calcium channel blockers, particularly non-dihydropyridine agents, offer additional antiproteinuric effects (Bakris et al., 2004). Diuretics help manage volume overload and enhance the efficacy of other antihypertensive medications.
Mineral Bone Disorder Management
CKD-mineral and bone disorder (CKD-MBD) develops early in kidney disease progression and contributes to cardiovascular mortality and bone disease (Ketteler et al., 2017). Management involves controlling serum phosphate, calcium, parathyroid hormone (PTH), and vitamin D metabolites through dietary modifications, phosphate binders, and vitamin D analogs.
Phosphate restriction typically begins when serum levels exceed normal ranges or GFR falls below 30 mL/min/1.73m². Dietary counseling focuses on limiting high-phosphate foods, particularly processed foods and cola beverages (Kalantar-Zadeh et al., 2010). Phosphate binders may be necessary when dietary restriction proves insufficient.
Vitamin D deficiency is common in CKD patients and requires correction with ergocalciferol or cholecalciferol supplementation. Active vitamin D analogs such as calcitriol or paricalcitol may be needed for secondary hyperparathyroidism management (Thadhani et al., 2012). However, these agents increase risks of hypercalcemia and hyperphosphatemia, requiring careful monitoring.
Calcimimetics represent a newer therapeutic option for secondary hyperparathyroidism management. Cinacalcet reduces PTH levels by increasing calcium-sensing receptor sensitivity (Block et al., 2004). While effective for biochemical control, evidence for clinical outcomes remains limited in pre-dialysis CKD patients.
Cardiovascular Risk Management
Cardiovascular disease represents the leading cause of mortality in CKD patients, with risk increasing as kidney function declines (Matsushita et al., 2010). Traditional risk factors such as hypertension, dyslipidemia, and diabetes require aggressive management, while CKD-specific factors including anemia, mineral bone disorders, and chronic inflammation also contribute to cardiovascular risk.
Statin therapy provides cardiovascular benefits in CKD patients, although effects may diminish in advanced disease stages. The SHARP trial demonstrated that simvastatin plus ezetimibe reduced cardiovascular events in CKD patients, including those on dialysis (Baigent et al., 2011). Current guidelines recommend statin therapy for most CKD patients over age 50 years.
Antiplatelet therapy decisions require balancing cardiovascular benefits against bleeding risks. Aspirin use in CKD patients associates with increased bleeding complications, and benefits for primary prevention remain uncertain (Baigent et al., 2018). Low-dose aspirin may be considered for secondary prevention in appropriate candidates.
Smoking cessation provides substantial benefits for CKD patients, reducing cardiovascular events and potentially slowing disease progression (Orth & Hallan, 2008). Counseling and pharmacological interventions should be offered to all smoking patients with CKD.
Recent Advances and Emerging Therapies 
Sodium-Glucose Cotransporter-2 Inhibitors
Sodium-glucose cotransporter-2 (SGLT2) inhibitors have emerged as important therapeutic agents for CKD patients, independent of diabetes status. Originally developed for diabetes management, these medications demonstrate kidney and cardiovascular protective effects through multiple mechanisms including reduced glomerular hyperfiltration, improved energy metabolism, and anti-inflammatory effects (Heerspink et al., 2020).
The DAPA-CKD trial demonstrated that dapagliflozin reduced kidney failure, cardiovascular death, and hospitalization for heart failure in patients with CKD, including those without diabetes (Heerspink et al., 2020). This landmark trial led to approval of SGLT2 inhibitors for non-diabetic CKD treatment. Similar benefits were observed with empagliflozin in the EMPA-KIDNEY trial (The EMPA-KIDNEY Collaborative Group, 2023).
Mechanisms of kidney protection include reduced proximal tubular glucose reabsorption, improved tubuloglomerular feedback, enhanced natriuresis, and direct anti-fibrotic effects (Vallon & Thomson, 2020). These effects occur independently of glucose-lowering actions, explaining benefits in non-diabetic patients.
Side effects include increased urinary tract infection risk, euglycemic ketoacidosis (rare), and volume depletion. Contraindications include type 1 diabetes, severe kidney impairment (GFR <20 mL/min/1.73m²), and history of diabetic ketoacidosis.
Endothelin Receptor Antagonists
Endothelin-1 plays important roles in kidney disease progression through vasoconstriction, inflammation, and fibrosis promotion (Kohan et al., 2011). Endothelin receptor antagonists have shown promise in clinical trials for various kidney diseases, although development has been complicated by fluid retention and cardiovascular side effects.
Atrasentan, a selective endothelin A receptor antagonist, demonstrated reduced proteinuria in diabetic kidney disease trials (de Zeeuw et al., 2014). However, increased fluid retention and heart failure events limited its development. Newer agents with improved selectivity profiles are under investigation.
Sparsentan combines endothelin receptor antagonism with angiotensin receptor blockade in a single molecule. The DUPLEX trial showed superior proteinuria reduction compared to irbesartan alone in patients with IgA nephropathy (Heerspink et al., 2023). This dual mechanism approach may provide enhanced kidney protection while minimizing side effects.
Complement Inhibitors
Complement system dysregulation contributes to various kidney diseases, particularly those affecting glomeruli. Complement inhibitors represent a promising therapeutic approach for diseases such as atypical hemolytic uremic syndrome, C3 glomerulopathy, and membranous nephropathy (Bomback et al., 2018).
Eculizumab, a C5 complement inhibitor, effectively treats atypical hemolytic uremic syndrome and has shown benefits in other complement-mediated kidney diseases (Legendre et al., 2013). However, high costs and infection risks limit widespread use.
Newer complement inhibitors targeting different pathway components are under development. These agents may provide more targeted therapy with fewer side effects for specific patient populations.
Gene and Cell-Based Therapies
Advances in understanding genetic causes of kidney disease have opened possibilities for targeted therapies. Antisense oligonucleotides and small interfering RNA technologies can modulate specific gene expression patterns involved in disease pathogenesis (Hu & Liu, 2017).
For ADPKD, tolvaptan represents the first approved disease-modifying therapy. This vasopressin V2 receptor antagonist slows cyst growth and kidney function decline in patients with rapidly progressive disease (Torres et al., 2012). However, hepatotoxicity risks require careful monitoring.
Stem cell and regenerative medicine approaches show promise for kidney repair and regeneration. Mesenchymal stem cells demonstrate anti-inflammatory and pro-regenerative effects in preclinical models (Reinders et al., 2013). Clinical trials are ongoing to evaluate safety and efficacy in human CKD patients.
Table 1: Current Evidence-Based Treatment Recommendations for Non-Diabetic CKD
| Treatment Category | Recommendation | Evidence Level | Key Studies |
| ACE Inhibitors/ARBs | First-line for patients with albuminuria | High | REIN, AASK |
| Blood Pressure Target | <130/80 mmHg (no albuminuria), <120/80 mmHg (with albuminuria) | Moderate | SPRINT, KDIGO |
| SGLT2 Inhibitors | Consider for patients with eGFR 20-75 mL/min/1.73m² | High | DAPA-CKD, EMPA-KIDNEY |
| Statin Therapy | Recommended for patients >50 years | High | SHARP |
| Phosphate Management | Restrict intake when levels elevated or eGFR <30 | Moderate | KDIGO Guidelines |
| Vitamin D Supplementation | Correct deficiency in all patients | Moderate | KDIGO Guidelines |
| Smoking Cessation | Strongly recommended for all smokers | High | Multiple observational studies |
| Dietary Protein | 0.8-1.0 g/kg/day for non-dialysis patients | Moderate | MDRD |

Challenges and Limitations in Current Practice 
Diagnostic Uncertainty
Determining the underlying cause of CKD remains challenging in many patients. Multiple disease processes often coexist, making it difficult to identify primary versus secondary causes. For example, patients with long-standing hypertension may develop both hypertensive nephrosclerosis and atherosclerotic kidney disease (O’Hare et al., 2007).
The lack of specific biomarkers for many kidney diseases limits diagnostic precision. Kidney biopsy provides definitive diagnosis but carries risks and is not always feasible. Non-invasive diagnostic tools with high sensitivity and specificity for specific disease entities remain largely unavailable.
Genetic testing is becoming more accessible but interpretation challenges persist. Many genetic variants have uncertain pathogenicity, and negative results do not exclude hereditary diseases. Genetic counseling resources are often limited, particularly in community practice settings.
Treatment Response Variability
Individual responses to CKD treatments vary considerably, making personalized therapy challenging. Factors influencing treatment response include genetic polymorphisms, concurrent medications, adherence patterns, and disease severity (Ruggenenti et al., 2010).
ACE inhibitor and ARB responses show substantial inter-individual variation in both efficacy and tolerability. Some patients develop hyperkalemia or acute kidney injury at standard doses, while others require higher doses for optimal effects. Predictive biomarkers for treatment response remain largely unavailable.
SGLT2 inhibitor effects also vary between patients, with some experiencing greater benefits than others. Factors predicting response are not well-established, limiting ability to identify optimal candidates for therapy.
Healthcare Disparities
Disparities in CKD care affect multiple patient populations. Racial and ethnic minorities experience higher CKD prevalence, faster progression rates, and reduced access to specialty care (Norton et al., 2016). These disparities persist despite adjustments for socioeconomic factors and comorbidities.
Geographic variations in CKD care quality are substantial, with rural patients having less access to nephrology specialists and advanced treatments. Telemedicine initiatives show promise for addressing geographic disparities but require infrastructure investments and regulatory changes (Diamantidis & Becker, 2014).
Economic barriers limit access to newer therapies and monitoring tests. SGLT2 inhibitors and other advanced treatments often require prior authorization and may not be covered by all insurance plans. Cost-effectiveness analyses are needed to inform coverage decisions.
Applications and Clinical Use Cases 
Case 1: Hypertensive Nephrosclerosis Management
A 65-year-old African American male with 20-year history of hypertension presents with creatinine 1.8 mg/dL (eGFR 40 mL/min/1.73m²) and microalbuminuria (ACR 45 mg/g). Blood pressure is 150/90 mmHg on amlodipine 10 mg daily.
Management approach includes initiating ACE inhibitor therapy with lisinopril, targeting blood pressure <130/80 mmHg. SGLT2 inhibitor addition should be considered given recent evidence for kidney protection. Cardiovascular risk assessment and statin therapy initiation are indicated. Regular monitoring of kidney function and electrolytes is essential, with nephrology referral appropriate for ongoing management.
Case 2: IgA Nephropathy Treatment
A 35-year-old male presents with gross hematuria following upper respiratory infection. Kidney biopsy confirms IgA nephropathy with moderate histological changes. eGFR is 55 mL/min/1.73m² with proteinuria 1.5 g/day.
Treatment involves ACE inhibitor therapy with dose titration to reduce proteinuria. Blood pressure target is <130/80 mmHg. Immunosuppressive therapy consideration depends on proteinuria response and histological severity. Fish oil supplementation may provide modest benefits. Long-term monitoring for disease progression is essential, with repeat biopsy consideration if clinical deterioration occurs.
Case 3: Polycystic Kidney Disease Management
A 45-year-old female with family history of ADPKD has bilateral enlarged kidneys on imaging and eGFR 50 mL/min/1.73m². Total kidney volume is 1200 mL with annual growth rate of 5%.
Management includes genetic counseling and family screening. Tolvaptan therapy should be considered given rapid disease progression. Blood pressure control with ACE inhibitors is important. Screening for intracranial aneurysms and liver involvement is indicated. Pain management strategies and urinary tract infection prevention are important supportive measures.
Comparison with Diabetic CKD 
Non-diabetic and diabetic CKD share many pathophysiological pathways and treatment approaches, but important differences exist. Diabetic kidney disease typically follows a more predictable progression pattern, with initial hyperfiltration followed by gradual GFR decline (Ritz et al., 1999). Non-diabetic causes show more heterogeneous progression patterns depending on underlying etiology.
Proteinuria patterns differ between disease types. Diabetic nephropathy usually presents with gradually increasing albuminuria progressing to overt proteinuria. Non-diabetic glomerular diseases may present with sudden onset heavy proteinuria or persistent microscopic hematuria (D’Amico, 1992).
Treatment responses also vary between patient populations. SGLT2 inhibitors show benefits in both diabetic and non-diabetic CKD, but mechanisms may differ. Glucose-lowering effects are obviously absent in non-diabetic patients, emphasizing the importance of glucose-independent mechanisms (Perkovic et al., 2019).
Cardiovascular risk profiles differ between groups, with diabetic patients generally having higher baseline risk. However, non-diabetic CKD patients still experience elevated cardiovascular mortality compared to the general population, requiring similar preventive interventions (Gansevoort et al., 2013).
Future Research Directions 
Precision Medicine Approaches
Developing personalized treatment strategies based on individual patient characteristics represents a major research priority. Pharmacogenomic studies are identifying genetic variants that predict medication responses and adverse effects (Swen et al., 2018). Implementation of genetic testing in routine clinical practice may improve treatment selection and dosing decisions.
Biomarker discovery efforts continue to identify novel markers for disease progression, treatment response, and adverse outcomes. Proteomics and metabolomics approaches are revealing new potential biomarkers that may improve clinical decision-making (Zhao, 2013).
Artificial intelligence and machine learning applications show promise for predicting disease progression and optimizing treatment regimens. Large electronic health record datasets provide opportunities to identify patterns not apparent through traditional analysis methods (Beam & Kohane, 2018).
Novel Therapeutic Targets
Multiple pathways involved in kidney disease progression represent potential therapeutic targets. Inflammatory mediators, fibrosis pathways, and metabolic dysfunction all contribute to disease progression and may be amenable to pharmacological intervention (Liu, 2006).
Autophagy and cellular senescence pathways are emerging as important contributors to kidney aging and disease progression. Interventions targeting these processes may slow or reverse kidney damage (Kang et al., 2019).
Microbiome research is revealing connections between gut bacteria and kidney disease progression. Therapeutic interventions targeting the microbiome may provide novel treatment approaches (Yang et al., 2018).
Regenerative Medicine
Stem cell therapies continue to show potential in preclinical studies for kidney repair and regeneration. Clinical trials are evaluating safety and efficacy of various cell-based approaches (Reinders et al., 2013).
Tissue engineering approaches aim to develop functional kidney tissue for transplantation. While still experimental, progress in organoid development and 3D bioprinting may eventually provide alternatives to traditional transplantation (Takasato et al., 2015).
Gene editing technologies such as CRISPR-Cas9 offer possibilities for correcting genetic defects causing inherited kidney diseases. Early studies in animal models show great potential for treating conditions such as ADPKD (Cruz et al., 2018).

Conclusion 

Key Takeaways
Non-diabetic CKD encompasses diverse disease entities requiring tailored diagnostic and treatment approaches. Early identification through appropriate screening and risk factor assessment remains crucial for optimal outcomes. Current evidence supports ACE inhibitor or ARB therapy as first-line treatment for most patients with albuminuria, with SGLT2 inhibitors emerging as important second-line agents.
Blood pressure control targets vary based on albuminuria status, with more intensive targets recommended for patients with proteinuria. Cardiovascular risk management requires aggressive intervention given the high mortality rates in CKD populations. Mineral bone disorder management becomes increasingly important as disease progresses.
Recent therapeutic advances including SGLT2 inhibitors and endothelin receptor antagonists provide new options for slowing disease progression. However, individual treatment responses vary considerably, highlighting the need for personalized medicine approaches.
Healthcare disparities in CKD care require continued attention and intervention. Access to specialty care and newer therapies remains limited for many patient populations. Telemedicine and other innovative care delivery models may help address these challenges.
Future research priorities include developing precision medicine approaches, identifying novel therapeutic targets, and advancing regenerative medicine technologies. These efforts may ultimately transform CKD care and improve outcomes for millions of affected patients.
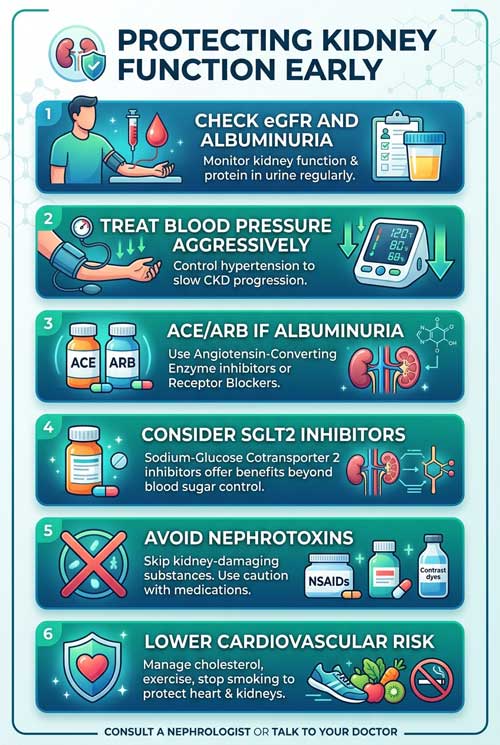
FAQs 
Q: How often should non-diabetic CKD patients be monitored?
A: Monitoring frequency depends on CKD stage and stability. Patients with stage 3 CKD typically require monitoring every 6-12 months, while those with stage 4-5 disease need evaluation every 3-6 months. More frequent monitoring is indicated during medication changes or clinical deterioration (KDIGO, 2024).
Q: When should patients be referred to nephrology?
A: Nephrology referral is recommended for patients with eGFR <30 mL/min/1.73m², rapidly declining kidney function (>5 mL/min/1.73m² per year), proteinuria >1 g/day, or suspected glomerular disease. Earlier referral may be appropriate for complex cases or when specialist expertise is needed for diagnosis or management (Smart & Titus, 2011).
Q: Are dietary restrictions necessary for all CKD patients?
A: Dietary modifications depend on CKD stage and complications. Protein restriction to 0.8-1.0 g/kg/day may be beneficial for non-dialysis patients. Phosphate restriction typically begins when levels are elevated or eGFR <30 mL/min/1.73m². Sodium restriction to <2.3 g/day helps with blood pressure control. Potassium restriction may be needed if hyperkalemia develops (Ikizler et al., 2020).
Q: Can CKD progression be reversed?
A: While most forms of CKD involve irreversible nephron loss, disease progression can be slowed or halted with appropriate treatment. Some conditions, particularly acute interstitial nephritis or drug-induced nephropathy, may show improvement with treatment and removal of offending agents. However, complete reversal of established CKD is uncommon (Remuzzi et al., 2006).
Q: What is the role of kidney biopsy in CKD evaluation?
A: Kidney biopsy provides definitive diagnosis and prognostic information for many kidney diseases. It is particularly valuable when clinical presentation suggests glomerular disease, when systemic disease is suspected, or when diagnosis would change management. Risks include bleeding and pain, making careful patient selection important (Hogan et al., 2016).
Q: How do SGLT2 inhibitors work in non-diabetic patients?
A: SGLT2 inhibitors provide kidney protection through multiple mechanisms independent of glucose lowering. These include reducing glomerular hyperfiltration through improved tubuloglomerular feedback, decreasing inflammation, improving energy metabolism, and potentially direct anti-fibrotic effects. Benefits are observed in non-diabetic CKD patients at similar rates to those with diabetes (Vallon & Thomson, 2020).
References: 
Baigent, C., Blackwell, L., Emberson, J., Holland, L. E., Reith, C., Bhala, N., … & Collins, R. (2011). Efficacy and safety of more intensive lowering of LDL cholesterol: A meta-analysis of data from 170,000 participants in 26 randomised trials. The Lancet, 378(9793), 719-728.
Baigent, C., Keech, A., Kearney, P. M., Blackwell, L., Buck, G., Pollicino, C., … & Emberson, J. (2005). Efficacy and safety of cholesterol-lowering treatment: Prospective meta-analysis of data from 90,056 participants in 14 randomised trials of statins. The Lancet, 366(9493), 1267-1278.
Baigent, C., Landray, M. J., Reith, C., Emberson, J., Wheeler, D. C., Tomson, C., … & Sharp Investigators. (2011). The effects of lowering LDL cholesterol with simvastatin plus ezetimibe in patients with chronic kidney disease (Study of Heart and Renal Protection): A randomised placebo-controlled trial. The Lancet, 377(9784), 2181-2192.
Bakris, G. L., Weir, M. R., Shanifar, S., Zhang, Z., Douglas, J., van Dijk, D. J., & Brenner, B. M. (2003). Effects of blood pressure level on progression of diabetic nephropathy: Results from the RENAAL study. Archives of Internal Medicine, 163(13), 1555-1565.
Beam, A. L., & Kohane, I. S. (2018). Big data and machine learning in health care. JAMA, 319(13), 1317-1318.
Beck, L. H., Bonegio, R. G., Lambeau, G., Beck, D. M., Powell, D. W., Cummins, T. D., … & Salant, D. J. (2009). M-type phospholipase A2 receptor as target antigen in idiopathic membranous nephropathy. New England Journal of Medicine, 361(1), 11-21.
Bidani, A. K., & Griffin, K. A. (2004). Pathophysiology of hypertensive renal damage: Implications for therapy. Hypertension, 44(5), 595-601.
Block, G. A., Martin, K. J., De Francisco, A. L., Turner, S. A., Avram, M. M., Suranyi, M. G., … & Bone, H. G. (2004). Cinacalcet for secondary hyperparathyroidism in patients receiving hemodialysis. New England Journal of Medicine, 350(15), 1516-1525.
Bolignano, D., Lacquaniti, A., Coppolino, G., Donato, V., Campo, S., Fazio, M. R., … & Buemi, M. (2009). Neutrophil gelatinase-associated lipocalin (NGAL) and progression of chronic kidney disease. Clinical Journal of the American Society of Nephrology, 4(2), 337-344.
Bomback, A. S., Markowitz, G. S., Appel, G. B., Radhakrishnan, J., D’Agati, V. D., & Canetta, P. A. (2016). Complement-mediated glomerular diseases: A tale of 3 pathways. Kidney International Reports, 1(3), 148-155.
Bonventre, J. V. (2009). Kidney injury molecule-1 (KIM-1): A urinary biomarker and much more. Nephrology Dialysis Transplantation, 24(11), 3265-3268.
Chebib, F. T., & Torres, V. E. (2016). Autosomal dominant polycystic kidney disease: Core curriculum 2016. American Journal of Kidney Diseases, 67(5), 792-810.
Chronic Kidney Disease Prognosis Consortium. (2019). Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: A collaborative meta-analysis. The Lancet, 375(9731), 2073-2081.
Cornec-Le Gall, E., Audrézet, M. P., Chen, J. M., Hourmant, M., Morin, M. P., Perrichot, R., … & Le Meur, Y. (2013). Type of PKD1 mutation influences renal outcome in ADPKD. Journal of the American Society of Nephrology, 24(6), 1006-1013.
Cornec-Le Gall, E., Alam, A., & Perrone, R. D. (2019). Autosomal dominant polycystic kidney disease. The Lancet, 393(10174), 919-935.
Cruz, N. M., Song, X., Czerniecki, S. M., Gulieva, R. E., Churchill, A. J., Kim, Y. K., … & Freedman, B. S. (2017). Organoid cystogenesis reveals a critical role of microenvironment in human polycystic kidney disease. Nature Materials, 16(11), 1112-1119.
Cushman, W. C., Evans, G. W., Byington, R. P., Goff Jr, D. C., Grimm Jr, R. H., Cutler, J. A., … & Ismail-Beigi, F. (2010). Effects of intensive blood-pressure control in type 2 diabetes mellitus. New England Journal of Medicine, 362(17), 1575-1585.
D’Agati, V. D., Kaskel, F. J., & Falk, R. J. (2011). Focal segmental glomerulosclerosis. New England Journal of Medicine, 365(25), 2398-2411.
D’Amico, G. (1992). Natural history of idiopathic IgA nephropathy: Role of clinical and histological prognostic factors. American Journal of Kidney Diseases, 20(3), 315-327.
de Zeeuw, D., Coll, B., Andress, D., Brennan, J. J., Tang, H., Houser, M., … & Vaidya, V. S. (2014). The endothelin antagonist atrasentan lowers residual albuminuria in patients with type 2 diabetic nephropathy. Journal of the American Society of Nephrology, 25(5), 1083-1093.
Denic, A., Glassock, R. J., & Rule, A. D. (2016). Structural and functional changes with the aging kidney. Advances in Chronic Kidney Disease, 23(1), 19-28.
Diamantidis, C. J., & Becker, S. (2014). Health information technology (IT) to improve the care of patients with chronic kidney disease (CKD). BMC Nephrology, 15(1), 7.
The EMPA-KIDNEY Collaborative Group. (2023). Empagliflozin in patients with chronic kidney disease. New England Journal of Medicine, 388(2), 117-127.
Fogazzi, G. B., Ponticelli, C., & Ritz, E. (1999). The urinary sediment: An integrated view. Nephrology Dialysis Transplantation, 14(11), 2667-2681.
Freedman, B. I., Dustin, M. L., Pilz, D. T., Borsa, N. G., Nassar, G. M., & De Luca, J. L. (2018). APOL1 genotype and kidney transplantation outcomes from deceased African American donors. Transplantation, 102(2), 194-202.
Fried, L. F., Emanuele, N., Zhang, J. H., Brophy, M., Conner, T. A., Duckworth, W., … & Peduzzi, P. (2013). Combined angiotensin inhibition for the treatment of diabetic nephropathy. New England Journal of Medicine, 369(20), 1892-1903.
Gansevoort, R. T., Correa-Rotter, R., Hemmelgarn, B. R., Jafar, T. H., Heerspink, H. J. L., Mann, J. F., … & Wen, C. P. (2013). Chronic kidney disease and cardiovascular risk: Epidemiology, mechanisms, and prevention. The Lancet, 382(9889), 339-352.
Glassock, R. J., Warnock, D. G., & Delanaye, P. (2017). The global burden of chronic kidney disease: Estimates, variability and pitfalls. Nature Reviews Nephrology, 13(2), 104-114.
Gruppo Italiano di Studi Epidemiologici in Nefrologia (GISEN). (1997). Randomised placebo-controlled trial of effect of ramipril on decline in glomerular filtration rate and risk of terminal renal failure in proteinuric, non-diabetic nephropathy. The Lancet, 349(9069), 1857-1863.
Gutierrez, O., Isakova, T., Rhee, E., Shah, A., Holmes, J., Collerone, G., … & Wolf, M. (2005). Fibroblast growth factor-23 mitigates hyperphosphatemia but accentuates calcitriol deficiency in chronic kidney disease. Journal of the American Society of Nephrology, 16(7), 2205-2215.
Harris, P. C., & Torres, V. E. (2009). Polycystic kidney disease. Annual Review of Medicine, 60, 321-337.
Heerspink, H. J. L., Stefánsson, B. V., Correa-Rotter, R., Chertow, G. M., Greene, T., Hou, F. F., … & DAPA-CKD Trial Committees and Investigators. (2020). Dapagliflozin in patients with chronic kidney disease. New England Journal of Medicine, 383(15), 1436-1446.
Heerspink, H. J. L., Radhakrishnan, J., Alpers, C. E., Barratt, J., Bieler, S., Diva, U., … & DUPLEX Trial Investigators. (2023). Sparsentan in patients with IgA nephropathy: A prespecified interim analysis from a randomised, double-blind, active-controlled clinical trial. The Lancet, 401(10388), 1584-1594.
Hogan, J. J., Mocanu, M., & Berns, J. S. (2016). The native kidney biopsy: Update and evidence for best practice. Clinical Journal of the American Society of Nephrology, 11(2), 354-362.
Hou, F. F., Zhang, X., Zhang, G. H., Xie, D., Chen, P. Y., Zhang, W. R., … & Liang, M. (2006). Efficacy and safety of benazepril for advanced chronic renal insufficiency. New England Journal of Medicine, 354(2), 131-140.
Hu, S., & Liu, G. (2017). Targeted therapy for chronic kidney disease. Advances in Experimental Medicine and Biology, 1005, 135-166.
Ikizler, T. A., Burrowes, J. D., Byham-Gray, L. D., Campbell, K. L., Carrero, J. J., Chan, W., … & Cuppari, L. (2020). KDOQI clinical practice guideline for nutrition in CKD: 2020 update. American Journal of Kidney Diseases, 76(3), S1-S107.
Inker, L. A., Eneanya, N. D., Coresh, J., Tighiouart, H., Wang, D., Sang, Y., … & Levey, A. S. (2021). New creatinine-and cystatin C–based equations to estimate GFR without race. New England Journal of Medicine, 385(19), 1737-1749.
Jager, K. J., Kovesdy, C., Langham, R., Rosenberg, M., Jha, V., & Zoccali, C. (2019). A single number for advocacy and communication—worldwide more than 850 million individuals have kidney diseases. Nephrology Dialysis Transplantation, 34(11), 1803-1805.
Kalantar-Zadeh, K., Gutekunst, L., Mehrotra, R., Kovesdy, C. P., Bross, R., Shinaberger, C. S., … & Kopple, J. D. (2010). Understanding sources of dietary phosphorus in the treatment of patients with chronic kidney disease. Clinical Journal of the American Society of Nephrology, 5(3), 519-530.
Kang, H. M., Ahn, S. H., Choi, P., Ko, Y. A., Han, S. H., Chinga, F., … & Susztak, K. (2015). Defective fatty acid oxidation in renal tubular epithelial cells has a key role in kidney fibrosis development. Nature Medicine, 21(1), 37-46.
KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. (2024). Kidney International, 105(4S), S117-S314.
Ketteler, M., Block, G. A., Evenepoel, P., Fukagawa, M., Herzog, C. A., McCann, L., … & Tonelli, M. (2017). Executive summary of the 2017 KDIGO Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD) Guideline Update: What’s changed and why it matters. Kidney International, 92(1), 26-36.
Kohan, D. E., Rossi, N. F., Inscho, E. W., & Pollock, D. M. (2011). Regulation of blood pressure and salt homeostasis by endothelin. Physiological Reviews, 91(1), 1-77.
Lazarus, B., Chen, Y., Wilson, F. P., Sang, Y., Chang, A. R., Coresh, J., & Grams, M. E. (2016). Proton pump inhibitor use and the risk of chronic kidney disease. JAMA Internal Medicine, 176(2), 238-246.
Legendre, C. M., Licht, C., Muus, P., Greenbaum, L. A., Babu, S., Bedrosian, C., … & Ariceta, G. (2013). Terminal complement inhibitor eculizumab in atypical hemolytic-uremic syndrome. New England Journal of Medicine, 368(23), 2169-2181.
Levey, A. S., Stevens, L. A., Schmid, C. H., Zhang, Y. L., Castro III, A. F., Feldman, H. I., … & Coresh, J. (2009). A new equation to estimate glomerular filtration rate. Annals of Internal Medicine, 150(9), 604-612.
Lewis, E. J., Hunsicker, L. G., Clarke, W. R., Berl, T., Pohl, M. A., Lewis, J. B., … & Collaborative Study Group. (2001). Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. New England Journal of Medicine, 345(12), 851-860.
Liu, Y. (2006). Renal fibrosis: New insights into the pathogenesis and therapeutics. Kidney International, 69(2), 213-217.
Matsushita, K., van der Velde, M., Astor, B. C., Woodward, M., Levey, A. S., de Jong, P. E., … & Chronic Kidney Disease Prognosis Consortium. (2010). Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: A collaborative meta-analysis. The Lancet, 375(9731), 2073-2081.
Naesens, M., Kuypers, D. R., & Sarwal, M. (2009). Calcineurin inhibitor nephrotoxicity. Clinical Journal of the American Society of Nephrology, 4(2), 481-508.
Nderitu, P., Doos, L., Jones, P. W., Davies, S. J., & Kadam, U. T. (2013). Non-steroidal anti-inflammatory drugs and chronic kidney disease progression: A systematic review. Family Practice, 30(3), 247-255.
Norton, J. M., Moxey-Mims, M. M., Eggers, P. W., Narva, A. S., Star, R. A., Kimmel, P. L., & Rodgers, G. P. (2016). Social determinants of racial disparities in CKD. Journal of the American Society of Nephrology, 27(9), 2576-2595.
O’Hare, A. M., Glidden, D. V., Fox, C. S., & Hsu, C. Y. (2004). High prevalence of peripheral arterial disease in persons with renal insufficiency: Results from the National Health and Nutrition Examination Survey 1999-2000. Circulation, 109(3), 320-323.
O’Neill, W. C., Robbin, M. L., Bae, K. T., Grantham, J. J., Chapman, A. B., Guay-Woodford, L. M., … & Bennett, W. M. (2005). Sonographic assessment of the severity and progression of autosomal dominant polycystic kidney disease: The Consortium of Renal Imaging Studies in Polycystic Kidney Disease (CRISP). American Journal of Kidney Diseases, 46(6), 1058-1064.
Orth, S. R., & Hallan, S. I. (2008). Smoking: A risk factor for progression of chronic kidney disease and for cardiovascular morbidity and mortality in renal patients—absence of evidence or evidence of absence? Clinical Journal of the American Society of Nephrology, 3(1), 226-236.
Perkovic, V., Jardine, M. J., Neal, B., Bompoint, S., Heerspink, H. J. L., Charytan, D. M., … & CREDENCE Trial Investigators. (2019). Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. New England Journal of Medicine, 380(24), 2295-2306.
Pugh, D., Gallacher, P. J., & Dhaun, N. (2019). Management of hypertension in chronic kidney disease. Drugs, 79(4), 365-379.
Reinders, M. E., de Fijter, J. W., Roelofs, H., Bajema, I. M., de Vries, D. K., Schaapherder, A. F., … & Rabelink, T. J. (2013). Autologous bone marrow-derived mesenchymal stromal cells for the treatment of allograft rejection after renal transplantation: Results of a phase I study. Stem Cells Translational Medicine, 2(2), 107-111.
Remuzzi, G., Benigni, A., & Remuzzi, A. (2006). Mechanisms of progression and regression of renal lesions of chronic nephropathies and diabetes. Journal of Clinical Investigation, 116(2), 288-296.
Ritz, E., Rychlík, I., Locatelli, F., & Halimi, S. (1999). End-stage renal failure in type 2 diabetes: A medical catastrophe of worldwide dimensions. American Journal of Kidney Diseases, 34(5), 795-808.
Rodrigues, J. C., Haas, M., & Reich, H. N. (2017). IgA nephropathy. Clinical Journal of the American Society of Nephrology, 12(4), 677-686.
Ruggenenti, P., Cravedi, P., & Remuzzi, G. (2010). The RAAS in the pathogenesis and treatment of diabetic nephropathy. Nature Reviews Nephrology, 6(6), 319-330.
Shlipak, M. G., Matsushita, K., Ärnlöv, J., Inker, L. A., Katz, R., Polkinghorne, K. R., … & CKD Prognosis Consortium. (2013). Cystatin C versus creatinine in determining risk based on kidney function. New England Journal of Medicine, 369(10), 932-943.
Smart, N. A., & Titus, T. T. (2011). Outcomes of early versus late nephrology referral in chronic kidney disease: A systematic review. American Journal of Medicine, 124(11), 1073-1080.
Swen, J. J., Nijenhuis, M., de Boer, A., Grandia, L., Maitland-van der Zee, A. H., Mulder, H., … & van der Weide, J. (2011). Pharmacogenetics: From bench to byte—an update of guidelines. Clinical Pharmacology & Therapeutics, 89(5), 662-673.
Takasato, M., Er, P. X., Chiu, H. S., Maier, B., Baillie, G. J., Ferguson, C., … & Little, M. H. (2015). Kidney organoids from human iPS cells contain multiple lineages and model human nephrogenesis. Nature, 526(7574), 564-568.
Tesar, V., Troyanov, S., Bellur, S., Verhave, J. C., Cook, H. T., Feehally, J., … & VALIGA study of the ERA-EDTA Immunonephrology Working Group. (2015). Corticosteroids in IgA nephropathy: A retrospective analysis from the VALIGA study. Journal of the American Society of Nephrology, 26(9), 2248-2258.
Thadhani, R., Appelbaum, E., Pritchett, Y., Chang, Y., Wenger, J., Tamez, H., … & Zoccali, C. (2012). Vitamin D therapy and cardiac structure and function in patients with chronic kidney disease: The PRIMO randomized controlled trial. JAMA, 307(7), 674-684.
Torres, V. E., Chapman, A. B., Devuyst, O., Gansevoort, R. T., Grantham, J. J., Higashihara, E., … & TEMPO 3:4 Trial Investigators. (2012). Tolvaptan in patients with autosomal dominant polycystic kidney disease. New England Journal of Medicine, 367(25), 2407-2418.
United States Renal Data System. (2023). USRDS annual data report: Epidemiology of kidney disease in the United States. National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases.
Vallon, V., & Thomson, S. C. (2020). The tubular hypothesis of nephron filtration and diabetic kidney disease. Nature Reviews Nephrology, 16(6), 317-336.
Webster, A. C., Nagler, E. V., Morton, R. L., & Masson, P. (2017). Chronic kidney disease. The Lancet, 389(10075), 1238-1252.
Wesseling, C., Aragón, A., González, M., Weiss, I., Glaser, J., Bobadilla, J., … & Roncal-Jiménez, C. (2016). Heat stress, hydration and uric acid: A cross-sectional study in workers of three occupations in a hotspot of Mesoamerican nephropathy in Nicaragua. BMJ Open, 6(12), e011034.
Wright Jr, J. T., Williamson, J. D., Whelton, P. K., Snyder, J. K., Sink, K. M., Rocco, M. V., … & SPRINT Research Group. (2015). A randomized trial of intensive versus standard blood-pressure control. New England Journal of Medicine, 373(22), 2103-2116.
Wright, J. T., Bakris, G., Greene, T., Agodoa, L. Y., Appel, L. J., Charleston, J., … & African American Study of Kidney Disease and Hypertension Study Group. (2002). Effect of blood pressure lowering and antihypertensive drug class on progression of hypertensive kidney disease: Results from the AASK trial. JAMA, 288(19), 2421-2431.
Yang, T., Richards, E. M., Pepine, C. J., & Raizada, M. K. (2018). The gut microbiota and the brain–gut–kidney axis in hypertension and chronic kidney disease. Nature Reviews Nephrology, 14(7), 442-456.
Zhang, J. L., Rusinek, H., Chandarana, H., & Lee, V. S. (2013). Functional MRI of the kidneys. Journal of Magnetic Resonance Imaging, 37(2), 282-293.
Zhao, Y. Y. (2013). Metabolomics in chronic kidney disease. Clinica Chimica Acta, 422, 59-69.
Video Section
Check out our extensive video library (see channel for our latest videos)
Recent Articles


