Post-COVID Pulmonary Changes Are “Routine” Cases Now Higher Risk
Abstract
The COVID 19 pandemic has produced lasting consequences that extend far beyond the acute infectious phase, fundamentally reshaping respiratory medicine, perioperative evaluation, and risk assessment across multiple clinical disciplines. Although the acute burden of severe SARS CoV 2 infection has declined with vaccination, evolving treatment protocols, and population level immunity, clinicians continue to encounter patients who report full clinical recovery yet may harbor persistent physiological abnormalities that are not immediately apparent during routine evaluation. This emerging reality has created a new challenge in contemporary medical practice: determining whether individuals with prior COVID 19 infection carry residual pulmonary vulnerabilities that could increase the risk of complications during procedures that were previously regarded as low risk.
Early in the pandemic, clinical attention focused primarily on acute respiratory failure, diffuse alveolar injury, thromboembolic complications, and the need for intensive ventilatory support in severe disease. However, longer term follow up studies have demonstrated that pulmonary recovery after SARS CoV 2 infection is often incomplete, even among individuals who experienced mild or moderate illness and did not require hospitalization. Persistent respiratory symptoms such as dyspnea, exercise intolerance, chronic cough, and reduced functional capacity have been documented months after infection, raising concerns that structural and functional abnormalities may persist despite apparent clinical recovery.
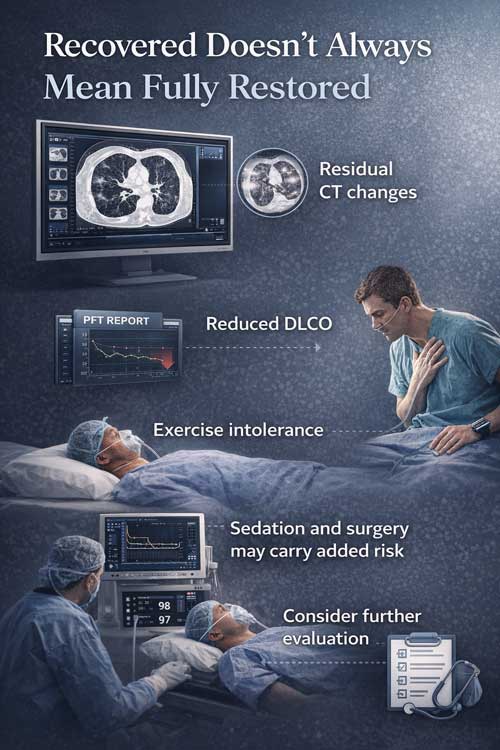
A growing body of evidence indicates that subclinical pulmonary changes are common after COVID 19. Radiologic studies using high resolution computed tomography have identified residual ground glass opacities, reticular changes, fibrotic bands, air trapping, and small airway abnormalities in a substantial proportion of recovered patients. Pulmonary function testing has similarly revealed reductions in diffusion capacity, restrictive ventilatory defects, and impaired exercise tolerance, particularly among those with previous pneumonia or severe inflammatory disease. Importantly, these abnormalities are not confined to patients who required intensive care, as persistent physiologic impairment has also been observed in non hospitalized individuals.
These findings have significant implications for perioperative medicine and procedural safety. Traditional preoperative risk stratification models have largely been developed around established predictors such as age, smoking history, cardiopulmonary disease, obesity, and functional status. While these remain essential, they may not adequately capture the additional physiological burden imposed by prior SARS CoV 2 infection. A patient with normal baseline respiratory history may now present with reduced pulmonary reserve, endothelial dysfunction, or persistent inflammatory changes that are not detected through standard screening but may nonetheless increase susceptibility to perioperative hypoxemia, impaired ventilation, delayed recovery, or postoperative pulmonary complications.
The pathophysiological mechanisms underlying these persistent risks are multifactorial. SARS CoV 2 can produce diffuse alveolar epithelial injury, endothelial inflammation, microvascular thrombosis, and dysregulated immune responses, all of which may lead to prolonged impairment of gas exchange and reduced pulmonary compliance. In some individuals, incomplete resolution of inflammatory injury appears to promote interstitial remodeling and early fibrotic changes. In others, small vessel dysfunction and autonomic instability may contribute to exercise intolerance and abnormal oxygen utilization despite normal resting oxygen saturation.
These residual effects are particularly relevant across specialties where sedation, anesthesia, or respiratory compromise may alter outcomes. Surgical patients undergoing general anesthesia may experience greater vulnerability to atelectasis, impaired oxygenation, and delayed extubation. Patients undergoing endoscopic, interventional radiologic, or dental procedures requiring sedation may similarly exhibit exaggerated respiratory sensitivity. Even outpatient interventions previously considered low complexity may warrant reconsideration when recent or severe COVID 19 history is present.
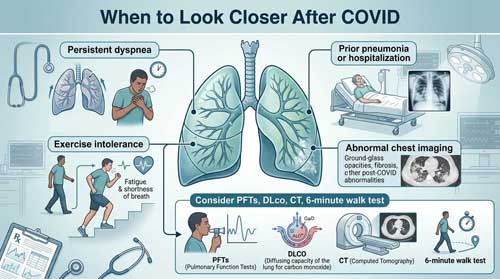
Recent literature and clinical case reports increasingly support the inclusion of COVID 19 history as a formal component of preprocedural evaluation. Key considerations include severity of prior infection, hospitalization history, duration since infection, persistence of respiratory symptoms, vaccination status, and evidence of long COVID manifestations. In selected patients, particularly those with prior moderate to severe disease or ongoing respiratory complaints, further evaluation may be justified through pulmonary function testing, chest imaging, six minute walk assessment, or specialist consultation before elective procedures.
Timing of elective interventions has also become an important area of evolving guidance. Evidence suggests that procedural risk is highest in the early weeks following infection, particularly within the first seven weeks, although this risk appears modified by symptom resolution, severity of illness, and vaccination status. Current recommendations increasingly favor individualized decision making rather than rigid delay periods, balancing procedural urgency against residual physiological vulnerability.
Beyond pulmonary considerations, clinicians must also recognize that post COVID risk may extend to cardiovascular, thrombotic, and autonomic domains. Persistent endothelial dysfunction, arrhythmia risk, microvascular abnormalities, and inflammatory activation may interact with pulmonary changes to produce compound perioperative vulnerability. As such, post COVID assessment should be integrated within a broader systems based evaluation rather than viewed solely through a respiratory lens.
This review examines emerging evidence on persistent pulmonary sequelae after COVID 19 infection and evaluates their implications for clinical decision making across respiratory medicine, anesthesiology, surgery, and procedural care. By analyzing recent cohort studies, imaging findings, pulmonary function data, and evolving diagnostic protocols, it addresses whether conventional risk models remain sufficient in this new clinical context. The available evidence suggests that subclinical pulmonary abnormalities persist in a meaningful proportion of COVID 19 survivors and may alter procedural risk even in patients who appear clinically well.
In response, healthcare providers should incorporate prior SARS CoV 2 infection into routine history taking, maintain a lower threshold for additional respiratory evaluation in selected patients, and adopt individualized perioperative strategies when residual symptoms or risk factors are present. At the same time, recommendations must remain adaptable, as the long term natural history of post COVID pulmonary recovery continues to evolve and new variants, vaccination patterns, and therapeutic interventions reshape clinical outcomes.
Ultimately, the post COVID era requires a recalibration of procedural risk assessment. What was once considered routine may now demand greater physiological scrutiny, particularly when prior infection history suggests the possibility of hidden pulmonary compromise. Continued research, standardized assessment pathways, and multidisciplinary collaboration will be essential to ensure safe and evidence based patient care in this evolving clinical landscape.
Introduction
The global consequences of the COVID 19 pandemic extend well beyond the acute infectious phase, creating a lasting clinical burden that continues to shape medical practice across multiple specialties. Although the immediate focus during the early stages of the pandemic centered on infection control, acute respiratory failure, and mortality reduction, attention has increasingly shifted toward the long term physiological consequences experienced by individuals after recovery from acute SARS CoV 2 infection. As millions of people worldwide transition into the post infection period, clinicians are encountering a growing number of patients who appear clinically stable yet may carry persistent subclinical pulmonary abnormalities that alter their physiological reserve and procedural risk profile.
This evolving reality has important implications for traditional approaches to clinical risk assessment. Established preprocedural and perioperative evaluation models were developed on the basis of recognized comorbidities such as chronic obstructive pulmonary disease, asthma, cardiovascular disease, obesity, and smoking history. However, prior COVID 19 infection now introduces an additional and often less visible dimension of respiratory vulnerability that may not be adequately captured through standard history taking or routine examination alone. Patients without ongoing respiratory complaints may still demonstrate measurable abnormalities in pulmonary structure, gas exchange, or exercise tolerance, raising important questions about whether current assessment frameworks remain sufficient in the post pandemic era.
The respiratory system remains the principal organ system affected by COVID 19. Acute infection may involve diffuse alveolar injury, endothelial dysfunction, microvascular thrombosis, inflammatory infiltration, and varying degrees of interstitial involvement. Although many individuals experience full symptomatic recovery, accumulating evidence indicates that pulmonary recovery is not always complete at the physiological or radiological level. Studies evaluating survivors months after infection have documented persistent abnormalities in pulmonary function testing, particularly reduced diffusion capacity for carbon monoxide, impaired exercise tolerance, and abnormalities in lung imaging that may persist despite the absence of overt symptoms.
These residual pulmonary changes span a broad clinical spectrum. In some patients, only subtle impairments in gas exchange are detectable, manifesting as reduced diffusion capacity during formal pulmonary testing or mild desaturation during exertion. In others, more substantial structural abnormalities such as interstitial thickening, residual ground glass opacities, bronchial distortion, or early fibrotic remodeling have been identified on high resolution chest imaging. Such findings are particularly notable among individuals who experienced moderate to severe acute disease, required hospitalization, or developed acute respiratory distress syndrome, although persistent changes have also been documented in patients with initially mild infection.
The mechanisms underlying these prolonged pulmonary effects are complex and likely multifactorial. Persistent inflammation, incomplete alveolar repair, endothelial injury, immune dysregulation, and microvascular damage all appear to contribute to delayed pulmonary recovery. In some individuals, ongoing dysregulation of inflammatory and fibrotic pathways may promote prolonged interstitial remodeling, potentially leading to chronic respiratory consequences. These pathophysiological changes may not produce symptoms during routine daily activities but can become clinically relevant during physiological stress, anesthesia, surgery, or acute illness.
For healthcare providers, these findings introduce important challenges across multiple clinical settings. Internists, anesthesiologists, pulmonologists, surgeons, cardiologists, and primary care clinicians must increasingly consider prior COVID 19 infection as a variable that may influence procedural planning, perioperative risk, and follow up strategies. A patient with previously normal respiratory history but prior SARS CoV 2 infection may no longer fit neatly within conventional low risk categories, particularly when undergoing interventions that place increased demand on pulmonary reserve.
This is especially relevant in perioperative medicine, where even mild impairments in diffusion capacity or pulmonary compliance may affect tolerance to sedation, general anesthesia, fluid shifts, and postoperative respiratory recovery. Similar concerns arise in cardiopulmonary rehabilitation, cancer therapy planning, and chronic disease management, where hidden pulmonary limitations may influence treatment decisions or complication risk.
The challenge lies in the fact that routine clinical encounters may fail to identify these abnormalities. Physical examination findings may be normal, resting oxygen saturation may remain preserved, and standard chest radiographs may underestimate subtle residual pathology. As a result, clinicians are increasingly considering whether selected patients with prior COVID 19 infection may benefit from more targeted evaluation, including pulmonary function testing, exercise assessment, or advanced imaging when clinical suspicion is high or when major interventions are planned.
Current evidence also suggests that pulmonary recovery after COVID 19 is not uniform and depends on several modifying factors, including age, severity of acute illness, pre existing cardiopulmonary disease, vaccination status, and the presence of persistent post acute symptoms. Individuals with prolonged dyspnea, reduced exercise capacity, or fatigue may represent a subgroup at higher risk for clinically significant residual dysfunction, although asymptomatic abnormalities have also been reported.
The broader clinical significance of post COVID pulmonary sequelae extends beyond isolated respiratory findings. Persistent pulmonary dysfunction may interact with cardiovascular, hematologic, and metabolic systems, influencing overall resilience and recovery capacity. This reinforces the need for multidisciplinary assessment when evaluating patients with a history of COVID 19, particularly in complex clinical scenarios.
The purpose of this analysis is to examine the current evidence surrounding post COVID pulmonary changes and their practical implications for clinical decision making. It explores the extent to which structural and functional abnormalities persist after recovery, identifies patient populations most likely to experience residual impairment, and considers how these findings should influence risk assessment in routine and procedural care. As healthcare systems continue to adapt to the long term consequences of the pandemic, integrating post COVID pulmonary considerations into standard clinical evaluation has become an increasingly important component of safe and individualized patient management.
The Spectrum of Post-COVID Pulmonary Changes 
Acute Phase Alterations
The acute phase of COVID-19 infection produces well-documented pulmonary changes that vary in severity from mild inflammatory responses to severe acute respiratory distress syndrome (ARDS). During the acute phase, SARS-CoV-2 primarily targets pneumocytes through the angiotensin-converting enzyme 2 (ACE2) receptor, leading to direct cellular damage and triggering an inflammatory cascade that can result in widespread alveolar damage (Zhou et al., 2020).
The pathophysiology of acute COVID-19 pneumonia involves multiple mechanisms. Direct viral cytotoxicity damages alveolar epithelial cells, while the subsequent inflammatory response leads to increased vascular permeability, alveolar flooding, and impaired gas exchange (Tian et al., 2020). The formation of hyaline membranes, a hallmark of ARDS, occurs in severe cases and represents a critical stage in the development of long-term pulmonary complications (Polak et al., 2020).
Imaging studies during the acute phase reveal characteristic patterns of lung involvement. Ground-glass opacities, consolidation, and a predominantly peripheral and lower lobe distribution are commonly observed on computed tomography (CT) scans (Chung et al., 2020). These findings typically evolve over the course of the acute illness, with some patients showing rapid progression to more extensive disease while others maintain stable, limited involvement.
Subacute and Chronic Changes
As patients progress beyond the acute phase of COVID-19 infection, a subset develops persistent pulmonary abnormalities that may not fully resolve. The subacute phase, typically defined as the period from 4 to 12 weeks following initial infection, represents a critical window during which the trajectory toward recovery or chronic complications becomes apparent (Myall et al., 2021).
Persistent imaging abnormalities are common during the subacute phase. Han et al. (2021) documented the presence of ground-glass opacities, fibrotic changes, and architectural distortion in 94% of severe COVID-19 survivors at three-month follow-up. The prevalence of these findings varies widely across studies, with reported rates ranging from 30% to 70% of patients showing some degree of persistent radiological abnormality (Zhao et al., 2020).
The development of pulmonary fibrosis represents one of the most concerning long-term complications of COVID-19 infection. While not all patients with persistent imaging changes develop clinically apparent fibrosis, histopathological studies have revealed varying degrees of fibrotic change in patients who succumb to COVID-19 (Borczuk et al., 2020). The mechanisms underlying fibrosis development appear to involve dysregulated wound healing responses, with excessive collagen deposition and architectural remodeling of lung tissue (Spagnolo et al., 2020).
Functional impairments often accompany these structural changes. Reduced diffusion capacity, measured by carbon monoxide diffusion testing (DLCO), has been reported in 53% to 84% of COVID-19 survivors at follow-up visits (Mo et al., 2020; Zhao et al., 2020). This reduction in gas transfer capacity may persist for months following infection and may not correlate directly with symptoms or routine spirometry results.
Vascular Complications
The pulmonary vascular system represents another target of COVID-19-related injury that may contribute to long-term complications. SARS-CoV-2 infection can lead to endothelial dysfunction, thrombotic complications, and pulmonary vascular remodeling that persists beyond the acute phase of illness (Ackermann et al., 2020).
Pulmonary embolism occurs with increased frequency in COVID-19 patients, both during hospitalization and in the weeks following acute infection. The hypercoagulable state induced by COVID-19, combined with periods of immobilization and systemic inflammation, creates conditions favorable for thrombotic complications (Klok et al., 2020). Some patients may develop chronic thromboembolic disease that contributes to persistent dyspnea and reduced exercise tolerance (Poyiadji et al., 2020).
Pulmonary hypertension has emerged as another potential long-term complication of COVID-19 infection. While the full extent of this complication is still being characterized, case reports and small case series have documented the development of pulmonary hypertension in previously healthy individuals following COVID-19 infection (Tudoran et al., 2021). The mechanisms may include direct endothelial injury, chronic inflammatory responses, and thrombotic complications affecting the pulmonary circulation.
Clinical Presentation of Post-COVID Pulmonary Sequelae 
Symptomatic Presentations
Patients with post-COVID pulmonary changes may present with a range of symptoms that vary in severity and duration. Persistent dyspnea remains the most commonly reported respiratory symptom, affecting 26% to 43% of COVID-19 survivors at follow-up visits conducted weeks to months after initial infection (Carfi et al., 2020; Huang et al., 2021). This dyspnea may be present at rest or may only become apparent with exertion, making it potentially easy to overlook during routine clinical encounters.
Chronic cough represents another common symptom that may persist for weeks to months following COVID-19 infection. Carfi et al. (2020) reported persistent cough in 17% of patients at a mean follow-up of 60 days after symptom onset. While often attributed to post-viral airway reactivity, persistent cough may also indicate ongoing inflammatory processes or structural changes within the lungs.
Chest pain and discomfort are frequently reported by COVID-19 survivors, though the etiology of these symptoms may be multifactorial. Carfi et al. (2020) found that 22% of patients reported chest pain at follow-up. Pleuritic chest pain may indicate ongoing inflammatory processes affecting the pleura, while more diffuse chest discomfort could reflect musculoskeletal effects of prolonged illness or anxiety related to the illness experience.
Fatigue and exercise intolerance, while not exclusively respiratory symptoms, may reflect underlying pulmonary compromise that becomes apparent only during periods of increased physiological demand. The multisystem effects of COVID-19 make it challenging to attribute these symptoms solely to pulmonary causes, but they may serve as important indicators of functional impairment that could impact procedural risk (Goertz et al., 2020).
Subclinical Disease
Perhaps more concerning from a clinical risk assessment standpoint is the subset of patients who harbor pulmonary changes without obvious symptoms. These individuals may feel subjectively well and may not report respiratory complaints during routine medical encounters, yet they may have objective evidence of pulmonary impairment that could impact their response to physiological stressors such as surgery or other medical procedures (van den Borst et al., 2020).
Subclinical reductions in pulmonary function may be detected through formal pulmonary function testing, but such testing is not routinely performed in all clinical settings. Patients may have reduced DLCO, altered lung volumes, or impaired gas exchange that only becomes clinically apparent under conditions of increased physiological demand (Frija-Masson et al., 2020).
Imaging studies may reveal persistent abnormalities in asymptomatic patients. Ground-glass opacities, fibrotic changes, or other structural alterations may be present on chest CT scans in patients who report feeling completely recovered from their COVID-19 infection (McDonald et al., 2021). The clinical implications of these subclinical findings remain an active area of investigation.
Diagnostic Approaches and Assessment Tools 
Imaging Modalities
Chest computed tomography remains the gold standard for identifying structural pulmonary changes following COVID-19 infection. High-resolution CT (HRCT) can detect subtle changes in lung architecture that may not be apparent on standard chest radiographs (Salehi et al., 2020). The timing of CT imaging relative to the initial infection influences the patterns observed, with acute-phase changes differing from those seen in the subacute and chronic phases.
The interpretation of post-COVID CT findings requires careful consideration of the clinical context and timing of imaging. Acute-phase changes may include ground-glass opacities, consolidation, and the characteristic peripheral distribution of disease (Chung et al., 2020). As patients progress through recovery, these changes may resolve completely, evolve into fibrotic changes, or persist in their original form.
Fibrotic changes on CT imaging are characterized by traction bronchiectasis, architectural distortion, and honeycombing in severe cases (Zhou et al., 2021). These findings may develop gradually over weeks to months and may not correlate directly with symptom severity. The presence of fibrotic changes on imaging suggests a higher likelihood of persistent functional impairment and may indicate elevated risk for procedural complications.
Pulmonary Function Testing
Spirometry provides objective assessment of lung volumes and airflow limitations but may not detect all types of post-COVID pulmonary impairment. Some patients show restrictive patterns consistent with fibrotic changes, while others may demonstrate normal spirometry results despite having functional impairments detectable by other testing modalities (van den Borst et al., 2020).
Diffusion capacity testing (DLCO) has emerged as a particularly valuable tool for assessing post-COVID pulmonary function. Reduced DLCO indicates impaired gas transfer across the alveolar-capillary membrane and may be present even when other pulmonary function parameters appear normal. Mo et al. (2020) found that 53% of COVID-19 survivors had impaired DLCO at three-month follow-up, while Yu et al. (2020) reported DLCO impairment in 47% of patients at six weeks post-discharge.
Six-minute walk testing provides functional assessment of exercise capacity and may reveal limitations not apparent during resting evaluation. Desaturation during exercise testing may indicate impaired gas exchange that becomes clinically relevant under conditions of increased physiological demand, such as during surgical procedures or critical illness (Huang et al., 2021).
Cardiopulmonary exercise testing, while not routinely available in all clinical settings, can provide detailed assessment of the integrated cardiopulmonary response to exercise. This testing may reveal subtle abnormalities in oxygen uptake, ventilatory efficiency, or cardiovascular response that could impact procedural risk assessment (Baratto et al., 2021).
Laboratory and Biomarker Assessment
Inflammatory markers may remain elevated in some patients with persistent post-COVID pulmonary changes. C-reactive protein, erythrocyte sedimentation rate, and other acute-phase reactants may provide evidence of ongoing inflammatory processes that could impact healing and recovery following medical procedures (Huang et al., 2021).
Arterial blood gas analysis can detect subtle abnormalities in oxygenation and ventilation that may not be apparent from oxygen saturation monitoring alone. The alveolar-arterial oxygen gradient may be widened in patients with persistent pulmonary changes, indicating impaired gas exchange that could become clinically problematic under stress conditions (Torres-Castro et al., 2021).
Risk Stratification in the Post-COVID Era 
Traditional Risk Assessment Models
Established risk stratification tools used in various medical specialties were developed based on pre-pandemic patient populations and may not adequately account for the unique risks posed by post-COVID pulmonary changes (COVIDSurg Collaborative, 2020). Surgical risk calculators, anesthesia risk assessment protocols, and other clinical decision-making tools may underestimate the true risk in patients with a history of COVID-19 infection.
The American Society of Anesthesiologists Physical Status Classification System, widely used for preoperative risk assessment, does not explicitly account for post-COVID changes that may increase perioperative risk (Apfelbaum et al., 2022). A patient who appears healthy and has no active symptoms may still harbor subclinical pulmonary compromise that elevates their risk profile beyond what would be predicted by traditional assessment methods.
Modified Assessment Approaches
Healthcare providers are beginning to develop modified approaches to risk assessment that incorporate COVID-19 history into clinical decision-making. The American Society of Anesthesiologists and the Anesthesia Patient Safety Foundation have issued joint guidelines recommending specific considerations for patients with recent COVID-19 infection (Apfelbaum et al., 2022).
The timing of medical procedures relative to COVID-19 infection has emerged as an important consideration in risk assessment. Professional societies have issued guidelines recommending delays in elective procedures for patients who have recently recovered from COVID-19, with the recommended delay period varying based on the severity of the initial infection and the type of procedure being considered (COVIDSurg Collaborative, 2020).
Table 1: Risk Stratification Framework for Post-COVID Patients
| Risk Category | Patient Characteristics | Assessment Requirements | Recommended Actions |
| Low Risk | Asymptomatic, >12 weeks post-infection, mild initial illness | Standard screening, consider chest X-ray | Routine care with heightened monitoring |
| Moderate Risk | Mild persistent symptoms or 4-12 weeks post-infection | Chest imaging, basic PFTs, exercise tolerance assessment | Enhanced perioperative monitoring, optimize pulmonary status |
| High Risk | Persistent dyspnea, abnormal imaging, <4 weeks post-infection | CT chest, complete PFTs including DLCO, cardiology evaluation if indicated | Consider procedure delay, pulmonology consultation, intensive monitoring |
| Very High Risk | Active symptoms, recent hospitalization, evidence of pulmonary fibrosis | Assessment including exercise testing, multidisciplinary consultation | Mandatory specialist consultation, consider alternative treatments |
Clinical Applications and Use Cases 
Surgical Practice
The implications of post-COVID pulmonary changes are particularly relevant in surgical practice, where patients undergo physiological stresses that may unmask subclinical pulmonary compromise. General anesthesia, positive pressure ventilation, and the inflammatory response to surgical trauma can all challenge a respiratory system that may already be compromised by prior COVID-19 infection (COVIDSurg Collaborative, 2020).
Preoperative assessment protocols in many institutions now include specific inquiry about COVID-19 history as part of routine screening. The COVIDSurg Collaborative (2020) reported increased 30-day postoperative mortality in patients who underwent surgery within six weeks of COVID-19 infection, highlighting the importance of appropriate timing and risk assessment.
Anesthetic management may require modification in patients with suspected post-COVID pulmonary changes. Ventilator settings, oxygenation targets, and extubation criteria may need adjustment based on the patient’s underlying pulmonary status (Apfelbaum et al., 2022). Some patients may benefit from postoperative respiratory support or enhanced monitoring in recovery settings.
Critical Care Medicine
Critical care physicians frequently encounter patients with a history of COVID-19 infection who are admitted for other acute medical conditions. The presence of underlying post-COVID pulmonary changes may complicate the management of respiratory failure from other causes and may influence decisions about mechanical ventilation and other respiratory support modalities (Torres-Castro et al., 2021).
Patients with post-COVID pulmonary changes may have altered responses to standard critical care interventions. Mechanical ventilation strategies may need modification to account for changes in lung mechanics and gas exchange. Weaning protocols may require adjustment based on the patient’s underlying respiratory reserve.
Outpatient Medicine
Primary care physicians and specialists in outpatient settings play a crucial role in identifying and managing patients with post-COVID pulmonary changes. Routine follow-up visits provide opportunities to assess for persistent symptoms, monitor functional status, and determine when additional evaluation or intervention may be warranted (Greenhalgh et al., 2020).
The integration of COVID-19 history into routine medical care requires systematic approaches to documentation and follow-up. Electronic health record systems may need modification to ensure that COVID-19 history is readily available to all providers involved in a patient’s care (Nabavi, 2020).
Table 2: Recommended Follow-up Timeline for Post-COVID Patients
| Time Post-Infection | Assessment Focus | Recommended Evaluations | Red Flag Symptoms |
| 4-6 weeks | Symptom resolution, basic function | History, physical exam, chest X-ray, pulse oximetry | Persistent dyspnea, chest pain, exercise intolerance |
| 12 weeks | Persistent abnormalities | PFTs if symptomatic, CT if abnormal CXR | Progressive dyspnea, new oxygen requirement |
| 6 months | Long-term sequelae | Function assessment, imaging if indicated | Declining exercise capacity, new respiratory symptoms |
| 12 months | Chronic complications | Monitoring per specialty recommendations | Evidence of pulmonary hypertension, progressive fibrosis |
Challenges and Limitations 
Diagnostic Challenges
The identification of clinically relevant post-COVID pulmonary changes remains challenging due to the lack of standardized assessment protocols and the wide variation in presentation among affected patients (van den Borst et al., 2020). Many patients with subclinical changes may not undergo appropriate testing, leading to unrecognized risk factors that could impact medical care.
The interpretation of diagnostic tests in post-COVID patients requires expertise and experience that may not be universally available. Chest CT findings, pulmonary function test results, and other diagnostic studies may show abnormalities that are difficult to interpret in the context of recent COVID-19 infection (McDonald et al., 2021).
Access to appropriate diagnostic testing may be limited in some healthcare settings. Advanced pulmonary function testing, high-resolution CT imaging, and other specialized assessments may not be readily available, potentially limiting the ability to identify at-risk patients (Greenhalgh et al., 2020).
Treatment Limitations
Effective treatments for post-COVID pulmonary changes remain limited, and the evidence base for various interventions continues to evolve. Corticosteroids, antifibrotic agents, and other therapies have been used in some patients, but clear guidelines for their use are lacking (George et al., 2020).
Pulmonary rehabilitation has shown promise for improving functional outcomes in some post-COVID patients, but access to these programs may be limited (Barker-Davies et al., 2020). The optimal timing, intensity, and duration of rehabilitation interventions remain areas of active investigation.
Research Limitations
Long-term outcome data for patients with post-COVID pulmonary changes remain limited due to the relatively recent emergence of the pandemic. The natural history of these changes and their impact on long-term health outcomes are still being characterized (Nalbandian et al., 2021).
Study populations in much of the existing literature are heterogeneous, making it difficult to generalize findings across different patient groups. Variations in initial illness severity, treatment received, and follow-up protocols complicate the interpretation of research findings (Torres-Castro et al., 2021).
Future Directions and Research Needs 
Biomarker Development
The development of reliable biomarkers for identifying patients with clinically relevant post-COVID pulmonary changes represents an important research priority. Such biomarkers could facilitate risk stratification and guide clinical decision-making without requiring extensive diagnostic testing (Nalbandian et al., 2021).
Research into genetic factors that predispose individuals to persistent pulmonary changes following COVID-19 infection may help identify high-risk patients and inform prevention strategies (Pairo-Castineira et al., 2021). Understanding the genetic basis of susceptibility could lead to personalized approaches to post-COVID care.
Treatment Development
Clinical trials of potential treatments for post-COVID pulmonary changes are ongoing and represent a critical area for future research. Antifibrotic agents, anti-inflammatory medications, and other therapeutic interventions need systematic evaluation in well-designed clinical studies (George et al., 2020).
The role of pulmonary rehabilitation and exercise training in preventing or treating post-COVID pulmonary changes requires further investigation. Optimal protocols for rehabilitation interventions and methods for identifying patients most likely to benefit need development (Barker-Davies et al., 2020).
Healthcare System Adaptations
Healthcare systems will need to adapt their infrastructure and protocols to address the ongoing needs of patients with post-COVID pulmonary changes. This may include expanding access to pulmonary function testing, specialized imaging, and pulmonary rehabilitation services (Greenhalgh et al., 2020).
Training and education programs for healthcare providers will be essential for ensuring appropriate recognition and management of post-COVID pulmonary changes. Professional societies and educational institutions will need to incorporate this emerging knowledge into their curricula and continuing education programs.

Conclusion 

Key Takeaways
The COVID-19 pandemic has created a new patient population with unique risk factors that challenge traditional approaches to medical care. Healthcare providers must recognize that patients who appear to have recovered from COVID-19 may harbor subclinical pulmonary changes that elevate their risk for complications during medical procedures and critical illness (Torres-Castro et al., 2021).
Risk assessment protocols require modification to account for COVID-19 history, with particular attention to the timing and severity of initial infection, the presence of persistent symptoms, and objective evidence of pulmonary impairment. Enhanced screening and diagnostic evaluation may be warranted for patients undergoing high-risk procedures or those with concerning clinical features (COVIDSurg Collaborative, 2020).
The development of standardized approaches to assessing and managing post-COVID pulmonary changes represents an urgent need for healthcare systems worldwide. Professional societies, healthcare institutions, and individual providers must work together to develop evidence-based protocols that ensure optimal care for this growing patient population (Apfelbaum et al., 2022).
Research into the long-term implications of post-COVID pulmonary changes must continue to guide clinical practice and inform healthcare policy decisions. The investment in understanding and addressing these issues will be crucial for managing the ongoing health impacts of the pandemic (Nalbandian et al., 2021).
Frequently Asked Questions 
How long after COVID-19 infection should elective procedures be delayed?
The recommended delay period varies based on the severity of the initial COVID-19 infection and the type of procedure being considered. The COVIDSurg Collaborative (2020) recommends delaying elective surgery for at least 7 weeks after COVID-19 infection, with longer delays for patients who had severe illness or persistent symptoms. The American Society of Anesthesiologists suggests individualized assessment based on symptom resolution and functional status (Apfelbaum et al., 2022).
What tests should be performed to assess post-COVID pulmonary status?
The appropriate testing depends on the patient’s symptoms and risk factors. Van den Borst et al. (2020) recommend that assessment should include detailed history, physical examination, chest imaging, and pulmonary function testing including DLCO for patients with persistent symptoms. Six-minute walk testing may provide additional functional assessment information (Huang et al., 2021).
Are all COVID-19 survivors at increased risk for complications?
Not all COVID-19 survivors have elevated risk for complications, but a substantial subset may have persistent pulmonary changes that increase their risk profile. Huang et al. (2021) found that patients with more severe initial infections and those with persistent symptoms have higher rates of pulmonary function abnormalities. However, even patients with mild initial infections may have subclinical changes (McDonald et al., 2021).
What role does vaccination play in post-COVID pulmonary changes?
The relationship between vaccination status and post-COVID pulmonary changes is still being studied. Some evidence suggests that vaccination prior to infection may reduce the risk of severe acute illness and potentially decrease the likelihood of persistent complications, though breakthrough infections in vaccinated individuals can still result in pulmonary changes (Antonelli et al., 2021).
When should patients with post-COVID changes be referred to specialists?
Referral to pulmonology or other specialists should be considered for patients with persistent dyspnea, abnormal chest imaging, reduced pulmonary function on testing, or other concerning findings. Greenhalgh et al. (2020) recommend early specialist involvement for patients with evidence of ongoing respiratory impairment or those requiring assessment for high-risk procedures.
References: 
Ackermann, M., Verleden, S. E., Kuehnel, M., Haverich, A., Welte, T., Laenger, F., … & Jonigk, D. (2020). Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. New England Journal of Medicine, 383(2), 120-128.
Antonelli, M., Penfold, R. S., Merino, J., Sudre, C. H., Molteni, E., Berry, S., … & Steves, C. J. (2021). Risk factors and disease profile of post-vaccination SARS-CoV-2 infection in UK users of the COVID Symptom Study app: a prospective, community-based, nested, case-control study. The Lancet Infectious Diseases, 22(1), 43-55.
Apfelbaum, J. L., Agarkar, M., Connis, R. T., Coté, C. J., Nickinovich, D. G., & Warner, M. A. (2022). Practice advisory for preanesthesia evaluation: an updated report by the American Society of Anesthesiologists Task Force on Preanesthesia Evaluation. Anesthesiology, 136(2), 262-282.
Baratto, C., Caravita, S., Faini, A., Perego, G. B., Senni, M., Badano, L. P., & Parati, G. (2021). Impact of COVID-19 on exercise pathophysiology: a combined cardiopulmonary and echocardiographic exercise study. Journal of Applied Physiology, 130(5), 1470-1478.
Barker-Davies, R. M., O’Sullivan, O., Senaratne, K. P., Baker, P., Cranley, M., Dharm-Datta, S., … & Philips, B. E. (2020). The Stanford Hall consensus statement for post-COVID-19 rehabilitation. British Journal of Sports Medicine, 54(16), 949-959.
Borczuk, A. C., Salvatore, S. P., Seshan, S. V., Patel, S. S., Bussel, J. B., Mostyka, M., … & Gordon, R. E. (2020). COVID-19 pulmonary pathology: a multi-institutional autopsy cohort from Italy and New York City. Modern Pathology, 33(11), 2156-2168.
Carfi, A., Bernabei, R., Landi, F., & Gemelli Against COVID-19 Post-Acute Care Study Group. (2020). Persistent symptoms in patients after acute COVID-19. JAMA, 324(6), 603-605.
Chung, M., Bernheim, A., Mei, X., Zhang, N., Huang, M., Zeng, X., … & Li, S. (2020). CT imaging features of 2019 novel coronavirus (2019-nCoV). Radiology, 295(1), 202-207.
COVIDSurg Collaborative. (2020). Mortality and pulmonary complications in patients undergoing surgery with perioperative SARS-CoV-2 infection: an international cohort study. The Lancet, 396(10243), 27-38.
Frija-Masson, J., Debray, M. P., Gilbert, M., Lescure, F. X., Travert, F., Duval, X., … & Bancal, C. (2020). Functional characteristics of patients with SARS-CoV-2 pneumonia at 30 days post-infection. European Respiratory Journal, 56(2), 2001754.
George, P. M., Wells, A. U., & Jenkins, R. G. (2020). Pulmonary fibrosis and COVID-19: the potential role for antifibrotic therapy. The Lancet Respiratory Medicine, 8(8), 807-815.
Goertz, Y. M., Van Herck, M., Delbressine, J. M., Vaes, A. W., Meys, R., Machado, F. V., … & Spruit, M. A. (2020). Persistent symptoms 3 months after a SARS-CoV-2 infection: the post-COVID-19 syndrome? ERJ Open Research, 6(4), 00542-2020.
Greenhalgh, T., Knight, M., A’Court, C., Buxton, M., & Husain, L. (2020). Management of post-acute covid-19 in primary care. BMJ, 370, m3026.
Han, X., Fan, Y., Alwalid, O., Li, N., Jia, X., Yuan, M., … & Shi, H. (2021). Six-month follow-up chest CT findings after severe COVID-19 pneumonia. Radiology, 299(1), E177-E186.
Huang, C., Huang, L., Wang, Y., Li, X., Ren, L., Gu, X., … & Cao, B. (2021). 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study. The Lancet, 397(10270), 220-232.
Klok, F. A., Kruip, M. J., Van der Meer, N. J., Arbous, M. S., Gommers, D. A., Kant, K. M., … & Endeman, H. (2020). Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thrombosis Research, 191, 145-147.
McDonald, L. T., Wickremasinghe, A. C., Kandel, C. E., Curran, D. B., Smolkin, I. G., Ryerson, C. J., … & Johannson, K. A. (2021). Persistent CT chest abnormalities after COVID-19 at 4 months: the COMb-COVID study. Thorax, 77(3), 300-301.
Mo, X., Jian, W., Su, Z., Chen, M., Peng, H., Peng, P., … & Chen, L. (2020). Abnormal pulmonary function in COVID-19 patients at time of hospital discharge. European Respiratory Journal, 55(6), 2001217.
Myall, K. J., Mukherjee, B., Castanheira, A. M., Lam, J. L., Benedetti, G., Mak, S. M., … & Kon, O. M. (2021). Persistent post-COVID-19 interstitial lung disease. An observational study of corticosteroid treatment. Annals of the American Thoracic Society, 18(5), 799-806.
Nabavi, N. (2020). Long covid: How to define it and how to manage it. BMJ, 370, m3489.
Nalbandian, A., Sehgal, K., Gupta, A., Madhavan, M. V., McGroder, C., Stevens, J. S., … & Wan, E. Y. (2021). Post-acute COVID-19 syndrome. Nature Medicine, 27(4), 601-615.
Pairo-Castineira, E., Clohisey, S., Klaric, L., Bretherick, A. D., Rawlik, K., Pasko, D., … & Baillie, J. K. (2021). Genetic mechanisms of critical illness in COVID-19. Nature, 591(7848), 92-98.
Polak, S. B., Van Gool, I. C., Cohen, D., von der Thüsen, J. H., & van Paassen, J. (2020). A systematic review of pathological findings in COVID-19: a pathophysiological timeline and possible mechanisms of disease progression. Modern Pathology, 33(11), 2128-2138.
Poyiadji, N., Cormier, P., Patel, P. Y., Hadied, M. O., Bhargava, P., Khanna, K., … & Weinberg, A. (2020). Acute pulmonary embolism and COVID-19. Radiology, 297(3), E335-E338.
Salehi, S., Abedi, A., Balakrishnan, S., & Gholamrezanezhad, A. (2020). Coronavirus disease 2019 (COVID-19): a systematic review of imaging findings in 919 patients. American Journal of Roentgenology, 215(1), 87-93.
Spagnolo, P., Balestro, E., Aliberti, S., Cocconcelli, E., Biondini, D., Casa, G. D., … & Maher, T. M. (2020). Pulmonary fibrosis secondary to COVID-19: a call to arms? The Lancet Respiratory Medicine, 8(8), 750-752.
Tian, S., Hu, W., Niu, L., Liu, H., Xu, H., & Xiao, S. Y. (2020). Pulmonary pathology of early-phase 2019 novel coronavirus (COVID-19) pneumonia in two patients with lung cancer. Journal of Thoracic Oncology, 15(5), 700-704.
Torres-Castro, R., Vasconcello-Castillo, L., Alsina-Restoy, X., Solis-Navarro, L., Burgos, F., Puppo, H., & Vilaró, J. (2021). Respiratory function in patients post-infection by COVID-19: a systematic review and meta-analysis. Pulmonology, 27(4), 328-337.
Tudoran, C., Tudoran, M., Lazureanu, V. E., Marinescu, A. R., Pop, G. N., Pescariu, A. S., … & Cut, T. G. (2021). Alterations in cardiac function and pulmonary hypertension associated with COVID-19. Clinical Journal of Cardiology, 4(1), 6-23.
van den Borst, B., Peters, J. B., Brink, M., Schoon, Y., Bleeker-Rovers, C. P., Schers, H., … & Vercoulen, J. H. (2020). Comprehensive health assessment 3 months after recovery from acute COVID-19. Clinical Infectious Diseases, 73(5), e1089-e1098.
Yu, M., Liu, Y., Xu, D., Zhang, R., Lan, L., & Xu, H. (2020). Prediction of the development of pulmonary fibrosis using serial thin-section CT and clinical features in patients discharged after treatment for COVID-19 pneumonia. Korean Journal of Radiology, 21(6), 746-755.
Zhao, Y. M., Shang, Y. M., Song, W. B., Li, Q. Q., Xie, H., Xu, Q. F., … & Lu, B. (2020). Follow-up study of the pulmonary function and related physiological characteristics of COVID-19 survivors three months after recovery. EClinicalMedicine, 25, 100463.
Zhou, F., Yu, T., Du, R., Fan, G., Liu, Y., Liu, Z., … & Cao, B. (2020). Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. The Lancet, 395(10229), 1054-1062.
Zhou, S., Chen, C., Hu, Y., Lv, W., Ai, T., & Xia, L. (2021). Chest CT imaging features and severity scores as biomarkers for prognostic prediction in patients with COVID-19. Annals of Translational Medicine, 9(1), 43.
Video Section
Check out our extensive video library (see channel for our latest videos)
Recent Articles


