Predicting Rheumatoid Arthritis Treatment Success New Biomarker Breakthrough

Key Takeaways
Rheumatoid arthritis is a chronic systemic autoimmune disease affecting approximately 1 percent of the global population. Despite major advances in biologic and targeted synthetic disease modifying antirheumatic drugs, treatment success remains highly variable. Approximately 40 percent of patients fail to respond adequately to first line biologic disease modifying antirheumatic drugs, and tumor necrosis factor inhibitors do not achieve remission in up to 70 percent of treated individuals. This variability reflects the biological heterogeneity of rheumatoid arthritis and underscores a central challenge in contemporary rheumatology: the absence of robust, clinically validated biomarkers capable of reliably predicting therapeutic response.
Unlike oncology, where molecular profiling frequently guides targeted therapy selection, rheumatology continues to rely largely on empirical treatment algorithms. Current clinical guidelines provide structured sequencing strategies but offer limited direction for matching individual patients with the therapy most likely to achieve rapid remission. As a result, treatment decisions often follow a trial and error approach. This paradigm contributes to prolonged disease activity, irreversible joint damage, unnecessary exposure to ineffective medications, and escalating healthcare costs.
Recent advances in biomarker discovery and computational analytics are beginning to transform this landscape. Emerging data suggest that specific molecular and cellular signatures may predict treatment response with clinically meaningful accuracy. For example, elevated pre treatment levels of the protein MZB1 have been associated with the development of antidrug antibodies and subsequent failure of adalimumab therapy. Identification of such biomarkers prior to treatment initiation could enable clinicians to avoid ineffective therapies and select alternative agents more likely to succeed.
Multi biomarker panels have demonstrated superior predictive performance compared with traditional serologic markers such as rheumatoid factor and anti cyclic citrullinated peptide antibodies alone. Integrative approaches combining cytokine profiles, gene expression patterns, genetic polymorphisms, and clinical parameters provide a more comprehensive representation of disease biology. Tissue based signatures further refine this strategy. Synovial biopsy analyses have revealed that B cell rich inflammatory phenotypes may predict response to B cell depleting therapies such as rituximab, while macrophage dominant profiles may inform responsiveness to interleukin 6 inhibition with agents such as tocilizumab. In some studies, these tissue signatures have achieved predictive accuracies exceeding 80 percent, suggesting substantial clinical utility when feasible.
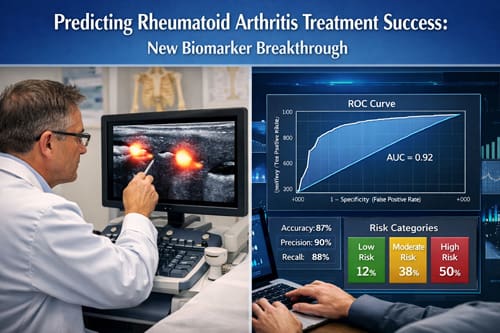
Parallel to advances in molecular profiling, machine learning methodologies are reshaping predictive modeling in rheumatoid arthritis. Computational models analyzing multidimensional patient datasets have achieved area under the curve values ranging from 0.54 to 0.92, with a mean around 0.71. Boosted tree algorithms and neural networks frequently demonstrate superior performance, while random forest classifiers have achieved AUC values as high as 0.86 in certain cohorts. By integrating laboratory markers, imaging findings, demographic variables, and longitudinal clinical data, these algorithms identify complex interaction patterns that may not be evident through conventional statistical approaches.
The potential economic implications of biomarker guided therapy are substantial. Modeling studies estimate that more precise treatment selection could reduce unnecessary medication exposure and improve treatment success rates by more than 30 percent among eligible patients. Annual healthcare savings have been projected to reach hundreds of millions of dollars, with some estimates approaching 850 million dollars in large healthcare systems. Beyond cost containment, earlier achievement of disease control may prevent structural joint damage, preserve functional status, and enhance long term quality of life.
Despite these promising developments, several challenges remain before precision medicine becomes standard practice in rheumatoid arthritis. Validation of predictive biomarkers in large, diverse populations is essential. Standardization of assay platforms, integration into clinical workflows, reimbursement considerations, and ethical management of patient level data all require careful attention. Moreover, prospective clinical trials are needed to confirm that biomarker guided therapy selection improves real world outcomes compared with conventional treatment strategies.
The future of rheumatoid arthritis management lies in transitioning from empiric sequencing toward biologically informed, individualized care. By combining molecular biomarkers, tissue characterization, and advanced computational modeling, clinicians may soon be able to match patients with optimal therapies from the outset. Such an approach has the potential to reduce disease progression, minimize therapeutic delays, lower healthcare expenditures, and ultimately redefine standards of care through precision medicine strategies.
Understanding Rheumatoid Arthritis Treatment Response Variability
Treatment response variability represents a fundamental challenge in rheumatoid arthritis management. Patient populations exhibit substantial heterogeneity in clinical trials, making it difficult to compare efficacies across different therapeutic interventions [1]. This heterogeneity extends beyond clinical trials into daily practice, where patient-specific factors markedly impact treatment outcomes.
Why Treatment Success Rates Differ Between Patients
Clinical response to rheumatoid arthritis treatment varies markedly based on numerous patient and disease characteristics. Furthermore, approximately 50% of people with early rheumatoid arthritis do not achieve remission at 6-month follow-up [1]. Several key factors influence treatment response:
- Patient characteristics: Greater age, longer disease duration, higher baseline Sharp score, and steroid use correlate with lower response rates [1]. Additionally, RF-negative status has been associated with increased likelihood of maintaining remission or low disease activity with monotherapy [2].
- Disease parameters: Higher baseline DAS28-CRP score, methotrexate/biologic naivety, baseline ESR, and anticitrullinated peptide antibody (ACPA) seropositivity correlate with greater ACR response [1]. Moreover, patients with shorter disease duration (≤ 5 years) demonstrated significantly higher odds of maintaining remission [2].
- Medication-specific factors: Biomarker presence such as shared epitope positivity (SE+) may predict response to specific drugs like abatacept [3], indicating the potential value of personalized treatment approaches.
These predictive factors collectively explained 29%, 37%, and 53% of variance in ACR20, ACR50, and ACR70 responses, respectively, highlighting both progress and limitations in predicting outcomes [1].
The Cost of Trial-and-Error Treatment Approaches
The financial implications of ineffective treatment sequencing are substantial. Indeed, the annual direct medical costs associated with rheumatoid arthritis reach approximately USD 12,509 for all patients, escalating to more than USD 36,000 for patients using biological DMARDs [3]. This burden extends beyond financial metrics:
Between 30% and >60% of patients do not experience adequate response to biological DMARDs, necessitating treatment changes [3]. In 2017, the weighted average cost per quality-adjusted life year (QALY) for targeted rheumatoid arthritis therapy was USD 212,423 [4].
Patient out-of-pocket spending represents an ongoing concern, especially without maximum spending limits in Medicare Part D’s catastrophic phase [4]. Although patient costs decreased from USD 585.008 in 2010 to USD 480.001 in 2019, this modest reduction fails to substantially alleviate financial burden [4].
Treatment switching remains common, with one study of Medicare patients estimating approximately 10% switch rate in the first year of therapy alone [4]. Each treatment change typically increases costs, particularly when patients transition to newer, more expensive medications.
Current Gaps in Predicting Treatment Outcomes
Neither the American College of Rheumatology nor the European League Against Rheumatism guidelines provide definitive guidance for optimizing treatment selection after initial therapy failure [3]. This absence of standardized prediction tools leads to several critical gaps:
Currently, difficult-to-treat rheumatoid arthritis (D2T RA) affects approximately 5%-28% of patients and poses a significant clinical challenge [5]. The success rate after 12 months of treatment with newly started biological or targeted synthetic DMARDs in D2T RA was merely 31% compared with 63% in non-D2T RA [6].
Early identification of patients at risk for becoming difficult-to-treat represents a promising strategy for improving outcomes [5]. Nevertheless, current approaches remain insufficient for guiding clinicians in identifying which patients will benefit from which treatment options.
Standardization issues persist in biomarker testing, integration into electronic health records, and economic evaluation of personalized approaches. Therefore, predicting treatment response remains an urgent unmet need in rheumatoid arthritis management [7].

Traditional Biomarkers Used in Rheumatoid Arthritis Treatment Guidelines
Laboratory biomarkers form the cornerstone of rheumatoid arthritis diagnosis, classification, and treatment response evaluation. Current guidelines rely heavily on autoantibodies and inflammatory markers to inform therapeutic decisions, yet these traditional tests present inherent limitations that affect clinical confidence.
Rheumatoid Factor and Anti-CCP Antibodies
Serum autoantibodies remain essential components in both diagnostic and prognostic assessment of rheumatoid arthritis. Rheumatoid factor (RF), an autoantibody primarily of the IgM class that targets the Fc portion of IgG, demonstrates sensitivity between 60-80% with specificity ranging from 70-85% [8]. However, RF positivity occurs in other autoimmune conditions, chronic infections, and even healthy elderly individuals, limiting its diagnostic precision [1].
Anti-cyclic citrullinated peptide (anti-CCP) antibodies offer comparable sensitivity (60-75%) but substantially higher specificity (95-99%) [1]. These antibodies target proteins that have undergone citrullination—a post-translational modification where arginine converts to citrulline [2]. Notably, second-generation anti-CCP (CCP2) assays predominate in Europe, while third-generation (CCP3) tests are more common in the United States [2].
The combination of both biomarkers provides superior diagnostic confidence. Double-positive patients have a higher likelihood ratio for developing rheumatoid arthritis and typically experience faster progression to clinically active disease [8]. Furthermore, these autoantibodies often precede clinical symptoms by up to 18 years [2], offering a potential window for early intervention.
Inflammatory Markers: CRP and ESR Levels
Acute phase reactants provide objective measures of systemic inflammation and constitute key components in treatment guidelines:
- C-reactive protein (CRP) is produced by the liver primarily in response to IL-6 stimulation. Normal levels typically remain below 3.0 mg/L [6]. Values between 10-100 mg/L suggest moderate inflammation consistent with active rheumatoid arthritis [6].
- Erythrocyte sedimentation rate (ESR) measures the rate at which red blood cells settle in plasma. Unlike CRP, ESR values are influenced by additional factors including age, sex, immunoglobulin levels, and anemia [2]. Normal ranges vary by age and gender—generally 0-15 mm/hr for men and 0-20 mm/hr for women under age 50 [9].
Yet these inflammatory markers demonstrate important limitations. Remarkably, in a large cohort of patients with high disease activity according to Clinical Disease Activity Index, more than half maintained normal CRP levels [2]. Correspondingly, joint inflammation on histology has been observed in nearly half of patients with normal CRP values [2].
Disease Activity Scores and Their Limitations
Composite disease activity measures incorporate laboratory biomarkers with clinical parameters to standardize assessment. The Disease Activity Score 28 (DAS28) utilizes either CRP or ESR alongside joint counts and patient global assessment [1]. Similarly, the Simplified Disease Activity Index (SDAI) and Clinical Disease Activity Index (CDAI) integrate these measures to guide treatment decisions [2].
Nevertheless, these scoring systems exhibit substantial shortcomings. First, clinical-serological discordance occurs frequently—many patients with active disease present with normal acute phase reactants [1]. Conversely, elevated inflammatory markers may reflect comorbidities rather than joint inflammation [1]. Additionally, approximately 20-30% of rheumatoid arthritis patients remain seronegative for both RF and anti-CCP, potentially delaying diagnosis and appropriate treatment initiation [1].
Given these limitations, rheumatoid arthritis treatment guidelines increasingly recognize the need for supplementary biomarkers that better predict individual treatment responses and disease trajectories [2].
Emerging Biomarkers for Treatment Response Prediction 
Beyond conventional laboratory tests, several promising biomarker categories have emerged to predict treatment outcomes in rheumatoid arthritis. These novel indicators offer potential for guiding therapeutic decisions before medication initiation, thus saving valuable time in disease management.
Genetic Markers and Pharmacogenomics
Pharmacogenetic variations substantially influence drug metabolism and efficacy in rheumatoid arthritis treatment. For methotrexate (MTX), polymorphisms in folate pathway genes predict both effectiveness and adverse reactions. The methylenetetrahydrofolate reductase (MTHFR) gene exhibits two primary variants—677C>T and 1298A>C—that affect enzyme activity. Patients carrying the MTHFR 677C>T variant experience approximately 70% increased toxicity risk compared to those without this polymorphism [10]. Meanwhile, reduced folate carrier-1 (RFC-1) variants, specifically the SLC19A1 80G>A polymorphism, show improved MTX response with homozygous 80A/A patients responding three times better than wild-type 80G/G homozygous individuals [11].
For TNF inhibitors, specific genetic biomarkers consistently predict response patterns. The PDE3A-SLCO1C1 locus shows strong association with response to infliximab and etanercept, reaching genome-wide significance with odds ratios of 2.91 [12]. Likewise, the PTPRC variant predicts response to specific TNF inhibitors in seropositive patients, albeit accounting for merely 0.5% of response variance [12].
Cytokine Profiles as Predictive Indicators
Serum cytokine levels before treatment initiation can forecast therapeutic outcomes. In regards to TNF inhibitor therapy:
- TNF and IL-15 levels are elevated in responders versus non-responders (p<0.05) [13]
- MCP-1 and IL-6 show a trend toward higher baseline concentration in responders (p<0.1) [13]
- IL-6/IL-2 ratio demonstrates superior prediction capacity than either cytokine alone [4]
A predictive model incorporating IL-6, IL-2, CRP, and DAS28-ESR achieved remarkable accuracy metrics: ROC-AUC 0.80-0.89, precision 0.83-1.00, and F1 scores 0.75-0.87 [4]. Henceforth, these markers could potentially guide initial therapy selection, reducing trial-and-error approaches commonly employed in rheumatoid arthritis management.
MicroRNA Expression Patterns
MicroRNAs—small non-coding RNA molecules—show considerable promise as biomarkers for treatment response. miR-103a-3p expression is particularly noteworthy, with first-degree relatives of rheumatoid arthritis patients showing approximately 7.6-fold increase compared to healthy controls and 1.96-fold increase compared to patients with active disease [3]. Empirical analysis revealed this marker effectively distinguishes between healthy controls and at-risk individuals (AUC=0.935, 92% specificity, 83% sensitivity) [3].
Other microRNAs correlate with both disease activity and treatment response. miR-146a levels predict response to anti-TNF therapy when combined with CRP measurements [14]. Meanwhile, miR-425-5p, miR-21-5p, and miR-212-3p show significantly reduced expression in patients treated with corticosteroids [14], potentially serving as monitoring markers during therapy.
DNA Methylation Signatures
DNA methylation patterns in peripheral blood increasingly appear valuable for predicting treatment outcomes. In studies of etanercept response, four CpG sites showed differential methylation patterns meeting false discovery rate thresholds of 0.05 [15]. Notably, the most differentially methylated position mapped to the LRPAP1 gene (p=1.46×10⁻⁸), which influences TGF-β activity [15].
For adalimumab therapy, researchers identified 27 CpGs whose collective methylation pattern predicted treatment response, with 20 showing p-values below 0.05 after adjusting for confounding factors [5]. These predictive methylation sites annotated to immunologically relevant genes including ADAP1, MRPL28, and GNA12, among others [5]. Subsequently, pathway analysis revealed enrichment in macrophage networks and inflammatory response pathways, underlining their biological relevance [5].
Together, these emerging biomarkers represent promising tools for moving rheumatoid arthritis treatment toward a more personalized medicine approach, potentially transforming current standardized treatment algorithms.
Machine Learning Models in Treatment Prediction
Machine learning techniques are ushering in a new era of personalized rheumatoid arthritis treatment through sophisticated predictive models. These computational approaches move beyond traditional statistical methods by identifying complex patterns in patient data that may otherwise remain undetected.
How ML Algorithms Analyze Patient Data
Machine learning (ML) models in rheumatoid arthritis research typically follow a structured analytical process. Initially, these systems leverage variables available in electronic health records from large patient cohorts [7]. The data preparation phase involves cleaning and organizing information, removing duplicates, and addressing anomalies. Feature selection—a crucial step—identifies notable predictors through statistical filtering, wrapper methods, or embedded techniques [16].
Common predictor variables include:
- Demographics (age, sex, race, ethnicity)
- Clinical measurements (previous CDAI scores, joint counts)
- Laboratory values (lymphocytes, neutrophils, ALT)
- Social determinants (per capita income, median home value) [7]
For missing data, various imputation methods are applied prior to model training. Subsequently, the processed dataset undergoes splitting into training and validation sets—typically in an 80:20 ratio [7]. The prediction pipeline then constructs models that output probability estimates for treatment response, which are compared against predetermined thresholds to obtain final binary outcomes [17].
Random Forest and Boosted Tree Performance
Tree-based methods dominate the machine learning landscape in rheumatoid arthritis research, employed in approximately 88% of relevant studies [2]. These algorithms create decision pathways based on patient characteristics to predict treatment outcomes.
In comparative analyzes, neural networks demonstrate superior performance with median AUC values of 0.79, followed by boosted tree algorithms at 0.73 and random forests at 0.70 [2]. XGBoost frequently emerges as a top performer—in one study examining TNF inhibitor response, CatBoost achieved an AUROC of 0.72 on the training set (ESPOIR cohort) [17]. Even more impressive, AdaBoost has reached accuracy levels of 85.71% with appropriate calibration [18].
The performance advantage of tree-based models over linear approaches suggests the presence of complex non-linear interactions between variables in rheumatoid arthritis treatment response [17]. Interestingly, results often differ based on medication class; models analyzing etanercept separately from monoclonal antibodies frequently yield improved predictions [17].
Validation Studies and AUC Results
Most machine learning studies in rheumatoid arthritis employ internal validation techniques, with only about 25% conducting external validation using independent cohorts [2]. Five-fold cross-validation repeated ten times represents a common approach for robust performance assessment [18].
Across multiple studies, AUC values range from 0.54 to 0.92, with a mean of 0.71 and median of 0.70 [2]. For difficult-to-treat rheumatoid arthritis prediction, models have demonstrated accuracy and AUC ranges of 0.693–0.730 and 0.763–0.776 respectively two years before meeting the D2T definition [8].
Model calibration remains essential for clinical utility. Techniques including Platt scaling, isotonic regression, spline calibration, and beta calibration ensure predicted probabilities accurately reflect observed outcomes [18]. Eventually, these calibrated probabilities allow for stratifying patients into low-, medium-, and high-risk groups, enabling nuanced clinical prioritization [18].
Integration of Clinical and Molecular Data
The combination of clinical and molecular data substantially enhances predictive performance. SHapley Additive exPlanations (SHAP) analysis consistently identifies key predictive variables, including disease activity measures (DAS28-ESR, CDAI), inflammatory markers (CRP), and patient-reported outcomes (HAQ) [8].
In one comprehensive analysis, DAS28-ESR emerged as the most influential feature (mean rank = 2.3), followed by CDAI, duration of biologic treatment, CRP, and HAQ [8]. For TNF inhibitor response prediction specifically, baseline disease activity, lymphocytes, ALT, neutrophils, age, weight, and smoking status proved most predictive [17].
Looking ahead, multi-omics integration—combining transcriptomic, proteomic, and metabolomic approaches with clinical data—represents the frontier of predictive modeling in rheumatoid arthritis [19]. This integrated approach may finally deliver the precision necessary to match patients with optimal treatments from the outset.

Breakthrough Discovery: MZB1 and Antidrug Antibody Formation 
Recent research has identified MZB1 (Marginal Zone B and B1 cell-specific protein) as a pivotal biomarker capable of predicting treatment outcomes in rheumatoid arthritis patients receiving biologic therapies. This breakthrough provides a molecular basis for understanding treatment failure mechanisms in certain patient populations.
The Role of MZB1 in B Cell Function
MZB1 functions as an endoplasmic reticulum (ER)-resident protein predominantly expressed in plasma cells and specialized B cell subsets, including marginal zone B cells and B1 cells [20]. As a molecular chaperone, MZB1 supports B cell differentiation by maintaining ER function and facilitating the transition from mature B cells to antibody-producing plasma cells [9].
In essence, MZB1 promotes immunoglobulin production through several mechanisms:
- Assists in proper folding and assembly of immunoglobulin heavy chains within the ER [9]
- Interacts with tail pieces of IgM and IgA heavy chains to enhance their secretion [21]
- Forms complexes with GRP94 and BiP to facilitate oxidative folding of antibodies [9]
- Regulates calcium signaling through interaction with the SERCA pump [22]
The protein’s role extends beyond antibody production to managing ER stress responses in B cells, which are tightly linked to immunoglobulin production capacity [21].
Predicting Adalimumab Treatment Response
Elevated pre-treatment whole-blood MZB1 transcript levels consistently predict non-response to adalimumab in rheumatoid arthritis patients [23]. This connection was discovered through machine learning approaches, as random forest classifiers achieved predictive accuracy with area under the receiver operating characteristic curves up to 0.86 [6].
Interestingly, network analysis identified MZB1 as a novel biomarker not captured by machine learning alone, highlighting the value of complementary analytical approaches [6]. Through stability selection gradient boosting, researchers identified additional methylation-based biomarkers with AUC values of 0.76 for predicting adalimumab response [5].
Association with Immunogenicity Development
The mechanistic link between MZB1 expression and treatment failure involves antidrug antibody (ADA) formation. At the 12-month mark, approximately 38.2% of patients treated with anti-TNF monoclonal antibodies tested positive for antidrug antibodies [24]. For adalimumab specifically, anti-drug antibodies developed in 28% of patients, with the majority (67%) forming during the first 28 weeks of treatment [25].
Patients with anti-adalimumab antibodies showed multiple concerning outcomes:
- Higher discontinuation rates (HR 3.0; 95% CI 1.6-5.5) [25]
- Consistently higher DAS28 scores after adjustment for confounders (0.4; 95% CI 0.2-0.6) [25]
- Reduced likelihood of achieving sustained minimal disease activity (HR 3.6, 95% CI 1.8-7.2) [25]
Clinical Validation and Performance Metrics
In clinical validation studies, MZB1 expression demonstrates a robust association with immunogenicity development risk [23]. As a result, MZB1 offers a potential pre-treatment screening tool to identify patients likely to develop treatment-limiting antibodies.
Through univariate analysis, researchers found an inverse relationship between antidrug antibody positivity and EULAR response at the 12-month mark for all bDMARDs (odds ratio 0.19; 95% CI, 0.09-0.38; p<0.001) [24]. This relationship persisted in generalized estimating equation models (OR 0.35; 95% CI, 0.18-0.65; p<0.001) for all visits from month 6 onward [24].
Along with antidrug antibody positivity (OR 0.41; 95% CI, 0.20-0.83), BMI ≥25 kg/m² (OR 0.35; 95% CI, 0.18-0.66) and rheumatoid factor positivity (OR 0.42; 95% CI, 0.20-0.87) were independently associated with poor treatment response [24].
Synovial Tissue Biomarkers and Cell Signatures
Examining the synovial tissue offers unique insights into rheumatoid arthritis pathophysiology and treatment outcomes. Unlike peripheral blood biomarkers, tissue-level analysis directly assesses cellular activity at the primary disease site.
Fibroblast Subset Identification
Recent analyzes have identified distinct synovial fibroblast subpopulations with specialized roles in joint pathology. Four primary subtypes exist with unique marker profiles: PRG4+ fibroblasts (expressing CD55, MMP3, PRG4, THY1neg) typically reside in the lining layer; CXCL12+ fibroblasts (expressing CXCL12, CCL2, ADAMTS1, THY1low) show enrichment for inflammatory pathways; POSTN+ fibroblasts (expressing POSTN and collagen genes) demonstrate intermediate THY1 expression; and CXCL14+ fibroblasts (expressing CXCL14, C3, ASPN, CD34, THY1) [26].
The proportions of these subsets differ between disease states and correlate with clinical parameters. For instance, POSTN+ fibroblasts correlate with higher joint counts and CRP levels in lymphoid pathotype synovium [27]. In contrast, CD34+, CD90+, cadherin11+ fibroblasts produce IL-6 and chemokines, yet CD34-, CD90-, cadherin11+ fibroblasts promote osteoclastogenesis [28].
Macrophage Populations in Treatment Response
Macrophages represent crucial cellular components in rheumatoid arthritis pathogenesis. The number of synovial sublining macrophages consistently correlates with:
- Disease activity indicators including CRP, ESR, and joint swelling count [29]
- Radiological severity and erosive burden [30]
- Treatment response across multiple therapy types [31]
Throughout disease progression, the ratio of pro-inflammatory M1 versus anti-inflammatory M2 macrophages increases [29]. Importantly, M1 macrophages exhibit enhanced anti-apoptotic capabilities compared to M2 counterparts, contributing to their persistence in inflamed joints [29].
B-Cell Poor vs B-Cell Rich Pathotypes
Synovial B-cell content predicts response to specific therapies. In patients receiving rituximab, 83% of those with B-cell poor synovium failed to achieve clinical response [1]. Yet, even within B-cell rich patients, the presence of germinal centers (GC) appears critical – 83% of GC-positive patients showed resistance to rituximab therapy [1].
In relation to pathotypes, patients with diffuse-myeloid pathotype (myeloid predominant but B-cell poor) demonstrated substantially higher response to tocilizumab (81%) versus rituximab (35%) [32]. Furthermore, patients poor in B-cells but rich in macrophages/mDCs showed 77% response to tocilizumab compared to merely 14% for rituximab [32].
Limitations of Tissue-Based Biomarker Testing
Regarding practical implementation, tissue-based biomarkers face several hurdles. Obtaining synovial biopsies remains invasive and technically challenging compared to blood-based testing [33]. Additionally, approximately 10% of synovial biopsies yield insufficient material for comprehensive analysis [34].
Standardization issues persist across testing methodologies and interpretation guidelines. The R4RA and STRAP trials, despite using standardized protocols, produced somewhat conflicting results regarding B-cell signatures [34]. Essentially, the optimal method for stratifying patients based on tissue characteristics remains unresolved, limiting widespread clinical adoption [34].
Biomarker-Guided Selection of Rheumatoid Arthritis Treatment Drugs
Selective biomarker profiles enable precision in rheumatoid arthritis drug selection, moving treatment approaches beyond trial-and-error strategies. Recent advances have identified specific indicators that predict response to major therapeutic classes, allowing for more targeted intervention strategies.
Matching Patients to TNF Inhibitors
Tumor necrosis factor inhibitor (TNFi) therapies show excellent results in first-line treatment, yet only about one-third of patients achieve strong response (ACR70) [35]. Several biomarkers consistently predict TNFi responsiveness:
- MRP8/14 (calprotectin) serves as a powerful predictor across multiple TNFi drugs including adalimumab and infliximab [35]
- PTPRC gene variants represent the most reproducible genetic biomarker for TNFi response, with the RS10919563 variant linked to remission improvement [35]
- miRNA-5196 outperforms CRP in predicting clinical response to anti-TNF therapy, particularly during early disease stages [35]
Forthwith, the molecular signature response classifier (MSRC) test evaluates 23 biomarkers to identify patients unlikely to respond to TNFi therapies, potentially saving over $850 million annually while improving ACR50 response rates by up to 31.3% [36].
IL-6 Pathway Inhibitors and Predictive Markers
For tocilizumab (TCZ), an IL-6 receptor antagonist, baseline CRP values correlate with improved patient VAS and CDAI scores (rr = 0.40, p = 0.017; rr = 0.44, p = 0.016) [37]. In fact, yearly progression of erosion Sharp score strongly associates with CDAI improvement (OR, 1.5; 95% CI, 1.03–2.07) [37].
Pre-treatment IL-6 measurement aids in estimating residual disease activity after TCZ treatment [35]. Notwithstanding these benefits, multi-biomarker disease activity scores during TCZ treatment should be interpreted cautiously since IL-6 directly influences these measurements [35].
JAK Inhibitor Response Indicators
Janus kinase inhibitors (JAK-I) demonstrate varied response patterns based on specific biomarkers. Elevated pre-treatment plasma levels of IL-12B, IL-17A, and IFN-γ associate with poor JAKi responses [38]. Specifically, high IFN-γ and IL-12B levels predict poor baricitinib response, till high IL-17A levels correlate with poor tofacitinib outcomes [38].
Soluble urokinase Plasminogen Activator Receptor (suPAR) and circulating calprotectin (cCLP) levels guide therapeutic choices between upadacitinib and filgotinib, with these biomarkers suggesting two distinct RA pathophenotypes—directing filgotinib toward lymphocyte-poor forms and upadacitinib toward myeloid forms [10].
B-Cell Depletion Therapy Selection
Rituximab, a chimeric anti-CD20 monoclonal antibody, demonstrates superior efficacy in seropositive patients [11]. Since not all patients respond, newer B-cell depletion approaches offer more extensive depletion beyond conventional mechanisms [39].
Circulating mir-125b levels correlate with excellent response to rituximab treatment, with high levels at disease onset linked to good response three months later [35].
Comparison Table: Biomarkers by Drug Class
| Drug Class | Key Predictive Biomarkers | Response Association |
| TNF Inhibitors | MRP8/14, PTPRC gene, miR-5196 | Higher levels = Better response |
| IL-6 Inhibitors | Baseline CRP, erosion Sharp score | Higher levels = Better response |
| JAK Inhibitors | IL-12B, IL-17A, IFN-γ | Lower levels = Better response |
| B-Cell Therapies | RF/ACPA status, miR-125b | Seropositivity, higher miR-125b = Better response |
Clinical Implementation Challenges and Future Directions 
Despite advances in biomarker discovery, successful clinical implementation faces multiple hurdles. The transition from research to routine practice requires overcoming several key challenges to realize the potential benefits of personalized rheumatoid arthritis treatment.
Standardization of Biomarker Testing
Laboratory methodologies for biomarker assessment often lack uniform protocols across clinical settings. Current testing approaches vary considerably between centers, limiting result comparability and clinical confidence. Furthermore, the quality of biomarker samples and measurement techniques requires rigorous standardization to ensure reliable outcomes across diverse healthcare environments.
External Validation Requirements
Many predictive models demonstrate adequate performance in development cohorts yet underperform in external populations. Models for difficult-to-treat rheumatoid arthritis show moderate discrimination and calibration, highlighting the need for further refinement [12]. Most importantly, while internal validation remains common, only about 25% of studies conduct proper external validation using independent cohorts.
Integration into Electronic Health Records
EHRs present opportunities to streamline biomarker implementation through:
- Disease activity calculators that help clinicians track patient status [40]
- Visualization tools that improve medication adherence [40]
- Natural language processing to extract valuable clinical information [41]
Nevertheless, patients report inconsistent data collection at clinic visits, hampering their engagement with outcome measures [42].
Cost-Effectiveness Considerations
The MSRC test (PrismRA) costs approximately $117,103 per patient response [43], yet could potentially reduce the cost per quality-adjusted life year by 33.9% to $140,502 [36]. Biomarker-guided therapy selection might yield noteworthy cost savings through improved treatment targeting and reduced medication wastage [4].
Practical Steps for Clinicians Using New Biomarkers
Effective utilization of biomarkers in clinical practice requires methodical approaches to patient selection, result interpretation, and treatment modification. Physicians must carefully navigate these emerging tools to optimize rheumatoid arthritis treatment outcomes.
Patient Selection Criteria for Biomarker Testing
Biomarker testing should target specific patient populations to maximize clinical utility. Testing patients with nonspecific arthralgia, myalgia, or osteoarthritis is explicitly discouraged [13]. First, consider biomarker evaluation for:
- Patients with clear signs and symptoms of inflammatory polyarthritis [44]
- Individuals with high pre-test probability of disease [13]
- Patients requiring disease progression monitoring during active treatment [44]
- Cases where treatment response assessment is needed for therapy modification
Interpreting Biomarker Results
Result interpretation demands contextual consideration beyond simple numeric values. Importantly, RF titer fluctuations do not reliably correlate with disease activity [13]. In many instances, ESR and CRP values demonstrate discordance in approximately 26% of patients despite active disease [13]. Presently, when these inflammatory markers show discordant results, their predictive value for radiographic progression decreases substantially [13].
Adjusting Treatment Plans Based on Predictions
Treatment modifications should follow specific biomarker patterns. High-titer anti-CCP2 antibodies correlate with improved clinical response to rituximab and abatacept, potentially guiding biologic selection [13]. Secondarily, RF positivity may enhance response to B-cell depleting therapies [13]. Hereafter, clinicians should recognize that Multi-Biomarker Disease Activity (MBDA) scores during tocilizumab treatment require cautious interpretation due to IL-6 concentration increases that may counteract decreases in other inflammatory markers [3].
Monitoring Response with Biomarkers
Continuous biomarker monitoring provides objective assessment advantages over symptom evaluation alone. Clinical symptoms frequently lag behind biological changes, whereas biomarkers offer real-time feedback on disease activity [14]. Hence, strategic monitoring enables clinicians to identify silent inflammation, predict relapse risk, and detect emerging drug resistance [14]. In parallel, biomarkers help determine optimal timing for treatment escalation, tapering, or changing therapeutic approach based on molecular evidence rather than subjective reporting [14].
Conclusion 

Predicting treatment response in rheumatoid arthritis represents a pivotal frontier in rheumatology that continues to evolve through biomarker innovation. The transition from traditional trial-and-error approaches toward precision medicine offers substantial benefits for patient outcomes and healthcare economics. Emerging biomarker technologies now allow clinicians to match patients with optimal therapeutic options before treatment initiation, potentially saving valuable time in disease management while reducing unnecessary medication exposure.
Several key developments highlight the transformative potential of biomarker-guided therapy:
- MZB1 expression serves as a powerful predictor of antidrug antibody formation and subsequent treatment failure with biological DMARDs • Machine learning algorithms analyze complex patient data with remarkable accuracy, achieving AUC values up to 0.92 through integration of clinical and molecular parameters • Synovial tissue analysis reveals distinct cellular signatures that correspond with differential responses to specific drug classes • Multi-biomarker panels outperform individual markers, reflecting the heterogeneous nature of rheumatoid arthritis pathophysiology
These advances address a fundamental challenge in rheumatology practice. Unlike oncology, where molecular pathology routinely guides targeted therapy selection, rheumatoid arthritis management has lacked reliable prediction tools until recently. Thus, patients often endure prolonged disease activity while clinicians work through sequential treatment options.
Practical implementation of biomarker testing requires careful consideration of patient selection, timing, and result interpretation. Patients with clear inflammatory polyarthritis and those failing first-line therapy represent ideal candidates for advanced biomarker assessment. Though promising, current approaches face standardization hurdles across laboratory methodologies, integration into electronic health records, and economic evaluation of personalized strategies.
Clinical application of biomarker-guided therapy demands both technical knowledge and thoughtful consideration of patient-specific factors. The ideal approach combines objective molecular data with clinical expertise, recognizing that biomarkers should inform rather than dictate treatment decisions. Clinicians must balance emerging biomarker evidence against patient preferences, comorbidities, and practical considerations such as cost and accessibility.
The field of rheumatoid arthritis biomarkers will undoubtedly expand as technologies advance and larger validation studies emerge. Additional research focusing on external validation, standardization, and cost-effectiveness analysis remains essential before widespread clinical adoption becomes feasible. Ultimately, biomarker-guided treatment selection promises to transform rheumatoid arthritis management from empiric approaches toward truly personalized medicine, offering patients the right treatment at the right time while optimizing healthcare resources.
Frequently Asked Questions: 
FAQs
Q1. What are some promising biomarkers for predicting rheumatoid arthritis treatment response? Several emerging biomarkers show potential for predicting treatment outcomes, including genetic markers like MTHFR and RFC-1 variants, cytokine profiles such as TNF and IL-15 levels, microRNA expression patterns like miR-146a, and DNA methylation signatures. Additionally, the MZB1 protein has been identified as a key predictor of antidrug antibody formation.
Q2. How can machine learning improve rheumatoid arthritis treatment prediction? Machine learning models can analyze complex patient data to identify patterns that may not be apparent through traditional methods. These algorithms integrate clinical, laboratory, and molecular data to predict treatment outcomes with high accuracy. Some models have achieved area under the curve (AUC) values up to 0.92, remarkably outperforming conventional approaches.
Q3. What role do synovial tissue biomarkers play in guiding treatment decisions? Synovial tissue analysis provides direct insights into disease activity at the joint level. Different fibroblast and macrophage populations, as well as B-cell content, can predict responses to specific therapies. For example, patients with B-cell poor synovium tend to respond poorly to rituximab, while those with high macrophage content may benefit more from tocilizumab.
Q4. How can clinicians practically implement biomarker testing in their practice? Clinicians should consider biomarker testing for patients with clear signs of inflammatory polyarthritis, those failing first-line therapy, or when assessing treatment response. Interpretation of results should account for potential discordances between markers and clinical symptoms. Treatment plans can be adjusted based on specific biomarker patterns, such as using high-titer anti-CCP2 antibodies to guide biologic selection.
Q5. What challenges exist in implementing biomarker-guided therapy for rheumatoid arthritis? Key challenges include standardizing biomarker testing across different laboratories, integrating results into electronic health records, and conducting external validation studies to confirm predictive accuracy. Additionally, cost-effectiveness considerations and the need for clinician education on interpreting and applying biomarker data in practice remain important hurdles to widespread adoption.
References: 
[2] – https://pmc.ncbi.nlm.nih.gov/articles/PMC12607112/
[3] – https://pmc.ncbi.nlm.nih.gov/articles/PMC4723630/
[4] – https://www.ajmc.com/view/precision-medicine-could-yield-significant-cost-savings-in-ra-treatment
[5] – https://www.frontiersin.org/journals/immunology/articles/10.3389/
fimmu.2023.1303231/full
[6] – https://pubmed.ncbi.nlm.nih.gov/40415615/
[7] – https://acrabstracts.org/abstract/predicting-disease-activity-in-rheumatoid-arthritis-patients-using-machine-learning-data-from-the-acrs-rise-registry/
[8] – https://www.nature.com/articles/s41598-025-18298-y
[9] – https://www.jcancer.org/v17p0542.htm
[10] – https://pubmed.ncbi.nlm.nih.gov/37664943/
[11] – https://pmc.ncbi.nlm.nih.gov/articles/PMC9524768/
[12] – https://pubmed.ncbi.nlm.nih.gov/40977501/
[13] – https://pmc.ncbi.nlm.nih.gov/articles/PMC8205440/
[14] – https://rheumatologyassociateshouston.com/personalized-medicine-in-rheumatology-tailoring-treatment-to-your-genes-and-biomarkers/
[15] – https://www.sciencedirect.com/science/article/abs/pii/S0003496724604607
[16] – https://pmc.ncbi.nlm.nih.gov/articles/PMC11194317/
[17] – https://rmdopen.bmj.com/content/8/2/e002442
[18] – https://www.nature.com/articles/s41598-025-09975-z
[19] – https://www.sciencedirect.com/science/article/pii/S2001037023002805
[20] – https://www.frontiersin.org/journals/immunology/articles/10.3389/
fimmu.2026.1741773/full
[21] – https://pmc.ncbi.nlm.nih.gov/articles/PMC12861871/
[22] – https://www.sciencedirect.com/science/article/abs/pii/S0141813025069569
[23] – https://pmc.ncbi.nlm.nih.gov/articles/PMC12769510/
[24] – https://www.rheumatologyadvisor.com/news/monitoring-of-antidrug-antibodies-suggested-for-patients-with-ra-nonresponsive-to-bdmards/
[25] – https://www.hopkinsarthritis.org/arthritis-news/antidrug-antibodies/
[26] – https://www.nature.com/articles/s41467-018-02892-y
[27] – https://rmdopen.bmj.com/content/8/1/e001949
[28] – https://acrabstracts.org/abstract/identification-of-synovial-fibroblast-subsets-that-define-pathology-in-rheumatoid-arthritis/
[29] – https://pmc.ncbi.nlm.nih.gov/articles/PMC7377929/
[30] – https://pmc.ncbi.nlm.nih.gov/articles/PMC130001/
[31] – https://pmc.ncbi.nlm.nih.gov/articles/PMC3263447/
[32] – https://www.nature.com/articles/s41591-022-01789-0
[33] – https://www.medrxiv.org/content/10.1101/2021.03.19.21253995v1.full-text
[34] – https://www.thelancet.com/journals/lanrhe/article/PIIS2665-9913(23)00269-2/fulltext
[35] – https://pmc.ncbi.nlm.nih.gov/articles/PMC8995470/
[36] – https://pmc.ncbi.nlm.nih.gov/articles/PMC10394192/
[37] – https://www.nature.com/articles/s41598-020-73968-3
[38] – https://acrabstracts.org/abstract/proteomic-insights-into-jak-inhibitor-therapeutic-response-in-rheumatoid-arthritis/
[39] – https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2024.1454747/full
[40] – https://pmc.ncbi.nlm.nih.gov/articles/PMC4518025/
[41] – https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0069932
[42] – https://link.springer.com/article/10.1186/s12911-024-02696-9
[43] – https://www.jmcp.org/doi/full/10.18553/jmcp.2021.21120
[44] – https://arupconsult.com/content/rheumatoid-arthritis
Video Section
Check out our extensive video library (see channel for our latest videos)
Recent Articles


