Hidden Link: How AFib Directly Affects Your Memory Loss Risk

Atrial fibrillation (AF) and memory loss share a concerning relationship that extends beyond coincidental occurrence. AF is the most common clinically significant arrhythmia in the world. In the United States alone, about 2.5 million people have it. Men are 1.5 times more likely than women to experience it. Recent evidence demonstrates that AF is associated with a 39% increased risk of cognitive impairment in the general population and, more alarmingly, a 2.70-fold increased risk in post-stroke patients.
The connection between AF and dementia presents a substantial public health challenge. While more than 20% of individuals over 70 years of age have mild cognitive impairment (MCI), the presence of an irregular heartbeat appears to accelerate this decline. Furthermore, can AFib cause memory loss directly? The data suggest it can—AF is associated with a 1.38 increased risk of dementia and a 1.40 increased risk of cognitive impairment with or without stroke.
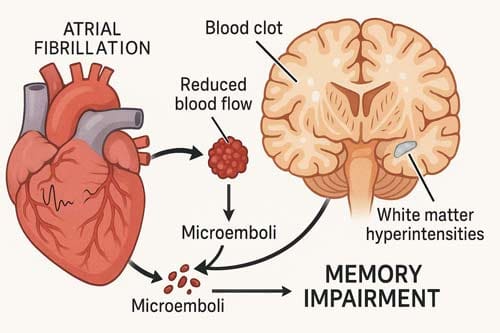
While the exact mechanisms are still being studied, researchers have found possible pathways such as microemboli, temporary cerebral hypoperfusion caused by beat-to-beat variability, and the onset of cerebral small vessel disease, which shows up as white matter hyperintensities and cerebral microbleeds. Simultaneously, short-term memory impairments may signify initial indicators of cognitive decline in patients with atrial fibrillation (AF).
Around 800,000 people in the US develop MCI, and more than 500,000 develop dementia each year. As the number of dementia cases worldwide is expected to reach 115.4 million by 2050, it is becoming more and more important to understand the link between AFib and cognition. This article analyses the evidence linking atrial fibrillation (AFib) to memory impairment, investigates the underlying pathophysiological mechanisms, delineates significant risk factors, and evaluates potential protective strategies to alleviate cognitive decline in patients afflicted with this prevalent arrhythmia.
Introduction
AFib and Memory Loss: What the Research Shows
Studies investigating the correlation between atrial fibrillation and cognitive deterioration have significantly increased in recent years. Numerous studies consistently indicate that patients with atrial fibrillation experience heightened risks of cognitive impairment across diverse demographics, even when accounting for conventional risk factors.
Cross-sectional studies on AFib and cognitive scores
Cross-sectional research offers substantial evidence for the AFib-cognition relationship. In the Rotterdam study, individuals with atrial fibrillation exhibited markedly elevated odds of cognitive impairment (OR = 1.7, 95% CI 1.2-2.5) and dementia (OR = 2.3, 95% CI 1.4-3.7) relative to those without the arrhythmia. This correlation remained significant regardless of cardiovascular risk factors and prior stroke history. Additionally, another cross-sectional study found that men with AF had lower cognitive scores than men without AF, and this relationship stayed the same even when stroke patients were taken out of the equation.
The impact varies based on AF type. Findings from the Atherosclerosis Risk in Communities (ARIC) study indicated that persistent atrial fibrillation (AF) was linked to diminished cognitive function, in contrast to paroxysmal AF, which exhibited no such correlation. It suggests that the burden of AF, beyond its mere existence, may affect cognitive outcomes.
Cross-sectional data from the Framingham Heart Study revealed specific cognitive vulnerabilities in reasoning, attention, and spatial abilities among patients with atrial fibrillation. For men in particular, AF was linked to worse scores on tests of global cognition, immediate and delayed memory, attention, and executive function.
Longitudinal data linking AFib to dementia risk
Longitudinal studies provide even more substantial evidence of the AF-dementia connection. Data from 15 population-based studies encompassing 46,637 participants showed that AF patients had 2.4 times higher dementia risk (95% CI 1.7-3.5) following stroke. Even in broader populations without stroke, AF was associated with a 1.6-fold increased dementia risk (95% CI 1.0-2.7).
The risk relationship appears especially pronounced for specific demographics:
- Younger patients: The Rotterdam study found AF associated with elevated dementia risk primarily among people under 67 years (HR = 1.81, 95% CI 1.11-2.94)
- Similarly, the US Intermountain Heart Collaborative Study showed the highest dementia risk in AF patients younger than 70 years.
- UK Biobank data demonstrated a 21% increased dementia risk in those diagnosed with AF before age 70 and a 36% higher risk of early-onset dementia.
A systematic review of 8 longitudinal studies with 77,668 patients without acute stroke found AF independently associated with a 40% increased dementia risk (HR = 1.4, 95% CI 1.2-1.7). Another meta-analysis examining the relationship between AF and cognitive decline included 21 studies with either cross-sectional or longitudinal designs, confirming that AF correlated with a twofold increased risk of dementia or cognitive impairment after stroke (HR = 2.7, 95% CI 1.8-4.0).
Notably, even without stroke history, the association persisted, with AF patients showing a 37% higher dementia risk (RR = 1.37, 95% CI 1.08-1.73).
AFib and short-term memory loss: observational trends
Observational research indicates AF’s impact on memory begins early and progresses steadily. In the Cardiovascular Health Study, cognitive function declined faster in patients who developed incident AF compared to those without AF, even in the absence of clinical stroke. Additionally, the study demonstrated that cognitive decline was more rapid after incident AF was identified than before diagnosis.
For older adults, the effect becomes increasingly pronounced with age. In models using the Modified Mini-Mental State Examination (3MSE), participants aged 75 and above experienced cognitive decline approximately 3 points faster per 5 years after developing AF. Similarly, in Digital Symbol Substitution Test (DSST) models, those 70 and older declined about 2 points faster per 5 years following AF onset.
This accelerated decline means AF patients reach cognitive impairment thresholds earlier. For instance, participants without AF reached the dementia-suggestive 3MSE score of <78 at age 87 on average, whereas those with AF reached this threshold by age 85—two years earlier.
Beyond general cognitive effects, AF appears particularly detrimental to executive function. Both prevalent and interim AF have been associated with greater decline in executive function capabilities, indicating that memory processing and organizational abilities may be especially vulnerable to AF’s effects.

How AFib Affects the Brain’s Structure and Function
Beyond the clinical manifestations of cognitive decline, atrial fibrillation causes measurable structural and functional changes in the brain. Neuroimaging studies have revealed multiple pathological alterations that help explain the mechanism behind AFib’s impact on memory and cognition.
Silent cerebral infarcts in AFib patients
Silent cerebral infarcts (SCIs) are small areas of brain damage that happen without any obvious clinical signs. In contrast to strokes that cause immediate neurological deficits, these “silent” injuries accumulate over time, ultimately impairing cognitive function. Patients with atrial fibrillation (AFib) have much higher rates of spinal cord injuries (SCIs) than patients without AFib.
Magnetic resonance imaging (MRI) studies show that people with AFib are 2.2 times more likely to get new SCIs than people with a normal sinus rhythm. Additionally, these silent infarcts transpire in patients undergoing optimal anticoagulation therapy, signifying that factors beyond thromboembolism contribute to cerebral injury. Consequently, between 15-26% of AFib patients show evidence of SCIs on neuroimaging, yet remain clinically asymptomatic.
The distribution of these infarcts often follows cortical and subcortical patterns typical of embolic events. Indeed, the cumulative effect of multiple SCIs over time creates a burden of cerebrovascular disease that eventually manifests as cognitive impairment. It helps explain why AFib patients can develop memory problems despite never experiencing a clinically apparent stroke.
White matter hyperintensities and microbleeds
White matter hyperintensities (WMHs) appear as bright areas on T2-weighted MRI scans and reflect small vessel disease affecting brain connectivity. In AFib patients, these abnormalities occur with greater frequency and progress more rapidly than in the general population.
Numerous studies demonstrate that AFib is independently correlated with elevated WMH volume, even after controlling for conventional vascular risk factors. The distribution usually impacts periventricular and deep white matter areas essential for cognitive processing. Longitudinal studies also show that people with AFib have faster WMH progression over time, with annual increases in WMH volume that are about 0.12 ml greater than those of matched controls.
Cerebral microbleeds, which are small bleeding spots that can be seen on gradient-echo MRI sequences, happen more often in AFib. These microbleeds indicate cerebral amyloid angiopathy or hypertensive arteriopathy and signify an increased risk of future haemorrhage. Microbleeds in AFib patients are linked to worse performance on cognitive tests, especially in areas like executive function and processing speed.
Anticoagulation therapy is a double-edged sword because it can stop thromboembolic events but also raise the risk of microbleeds. This fragile balance highlights the difficulty of treating brain problems caused by AFib.
Reduced hippocampal and gray matter volume
Perhaps most directly linked to memory impairment, AFib patients demonstrate measurable reductions in hippocampal volume. The hippocampus, which is essential for forming and retrieving memories, shows faster atrophy in AFib, which may explain the early memory problems seen in patients.
Volumetric MRI analyses indicate that patients with AFib have hippocampal volumes that are approximately 3-5% smaller than those of age-matched controls. This decrease is unrelated to overt cerebrovascular disease, indicating direct impacts of AFib on brain structure that extend beyond those caused by infarction.
In addition to the hippocampus, the total volume of grey matter decreases more rapidly in AFib. This widespread thinning of the cortex affects areas that are important for executive function, language processing, and visual-spatial skills. Diffusion tensor imaging studies further demonstrate compromised white matter integrity, as indicated by decreased fractional anisotropy values in the principal white matter tracts connecting these regions.
Chronic cerebral hypoperfusion, caused by irregular cardiac output, inflammatory processes, and oxidative stress, is probably the pathophysiological mechanism that causes these changes in volume. These structural changes work together to create a neuroanatomical basis for the cognitive decline seen in people with AFib.
The evidence indicates that AFib is a condition that adversely affects brain structure via various mechanisms—silent infarcts, white matter disease, microbleeds, and volume loss—each contributing to the clinical manifestation of memory impairment and cognitive dysfunction.
Mechanisms Behind AFib-Related Cognitive Decline
The pathophysiological processes connecting atrial fibrillation to cognitive deterioration involve multiple interrelated mechanisms. Understanding these pathways helps explain why AFib patients experience memory loss even in the absence of a clinically apparent stroke.
Cerebral hypoperfusion due to an irregular heartbeat
The irregularly irregular rhythm characteristic of AFib creates unstable cerebral blood flow patterns. During a normal sinus rhythm, the ventricles have adequate time to fill with blood and perfuse end organs with stable pulsatility. However, AFib disrupts this pattern, creating varying diastolic filling times—some allowing complete ventricular filling, others too short for adequate filling. This inconsistency leads to a 17% drop in cardiac output during exercise in AFib patients compared to those in normal sinus rhythm.
The result is a series of hypo-hyperperfusion events affecting cerebral microvasculature. Over time, these fluctuations trigger vascular adaptations that eventually develop into fulminant vascular disease. Patients with ventricular rates below 50 or above 90 beats per minute during AFib face a 7-fold increased risk of dementia compared to those with moderate ventricular response rates.
Furthermore, flow-mediated dilation becomes impaired in AFib patients, strongly indicating arterial endothelial dysfunction. This impairment worsens as AFib progresses from paroxysmal to persistent forms. Consequently, despite normal left ventricular stroke volume, cerebral tissue may experience hypoperfusion due to this abnormal vascular response.
Microemboli and thromboembolism from AFib
Thromboembolism constitutes a significant mechanism underlying cognitive decline associated with atrial fibrillation. AFib raises the risk of arterial thromboembolic events, primarily by causing clots to form in the left atrium. These emboli can be big enough to cause a stroke that is visible on a CT scan or small enough to cause silent cerebral infarcts.
Transcranial Doppler monitoring reveals that positive micro-embolic signals correlate with:
- Higher CHA₂DS₂-VASc scores (particularly ≥4)
- Elevated D-dimer levels
- Increased left atrial diameter
- Higher rates of silent cerebral ischemia
Patients with AFib suffer pulmonary embolism more frequently than those without, though this difference appears explained mainly by age and comorbidity rather than AFib itself. Nevertheless, the risk of venous thromboembolism peaks in the first 3-6 months after AFib diagnosis and decreases over time, paralleling the risk pattern for thromboembolic stroke.
Inflammation and endothelial dysfunction
AFib activates inflammatory pathways that contribute directly to cognitive impairment. Elevated levels of inflammatory markers—including C-reactive protein, interleukins, and TNF-α—correlate with AFib and can increase oxidative stress and endothelial injury. This inflammatory milieu appears associated with the arrhythmia itself rather than underlying structural heart disease.
Endothelial dysfunction, measured by reduced flow-mediated dilatation, is consistently observed in AFib patients and correlates with adverse outcomes. The mechanisms responsible include:
- Loss of organized atrial contraction leading to low blood flow and decreased endothelial nitric oxide synthase expression
- Atrial inflammation increases C-reactive protein and cytokines, exerting proinflammatory effects on endothelial cells
- Activation of the renin-angiotensin-aldosterone system contributes to myocardial and vascular oxidative stress
Chronic inflammation has been clearly linked to several forms of dementia, including Alzheimer’s disease. Importantly, patients with chronic AFib demonstrate more impaired flow-mediated dilation compared to those with paroxysmal AFib (4.20±1.66% vs. 6.27±1.94%), suggesting a dose-response relationship between AFib burden and vascular dysfunction.

Risk Factors That Worsen Memory Loss in AFib
Several specific risk factors amplify the relationship between atrial fibrillation and cognitive decline. These factors not only increase the likelihood of memory loss but can also accelerate its progression in patients with this common arrhythmia.
High CHA2DS2-VASc scores and dementia correlation
Mounting evidence demonstrates a robust association between elevated CHA2DS2-VASc scores and cognitive deterioration. In an extensive study involving 6,030 patients undergoing long-term warfarin therapy, researchers discovered that the risk of dementia escalated progressively with elevated CHADS2 scores. This trend continued even after adjusting for multiple variables, with AF patients exhibiting significantly higher dementia incidence across all CHADS2 score strata compared to non-AF controls.
Later studies have confirmed this connection using the improved CHA2DS2-VASc scoring system. Higher CHA2DS2-VASc scores (≥3 vs ≤1) were significantly associated with elevated dementia hazard ratios in both women (HR 7.77, 95% CI 5.94-10.17) and men (HR 4.75, 95% CI 4.15-5.44). The association persisted robustly even after adjusting for potential confounding variables.
Even in AF patients with very low embolic risk (CHA2DS2-VASc 0 or 1), those receiving oral anticoagulants exhibited lower dementia risk than untreated patients. This finding suggests that proper anticoagulation management may partially mitigate cognitive risks, even in low-risk individuals.
Persistent vs paroxysmal AFib and cognitive outcomes
The temporal pattern of AF directly impacts cognitive function. Persistent AF consistently demonstrates worse cognitive outcomes than paroxysmal AF. In fact, patients with persistent AF show lower global cognitive abilities and poorer performance on visuospatial-executive testing compared to those with paroxysmal AF.
An analysis of community-dwelling elderly individuals revealed that those with 100% AF burden (persistent AF) had lower executive and verbal cognitive test scores than those without AF. Conversely, individuals with only 1-6% AF burden (paroxysmal AF) showed no such deficits. These associations remained intact even after adjusting for both clinical stroke and silent cerebral infarcts, suggesting additional mechanisms beyond cerebrovascular events.
The longitudinal Cardiovascular Health Study further supports this relationship, noting that cognitive test scores declined by 10.3 points over five years (ages 80-85) in individuals who developed AF, versus only 6.4 points in matched controls without AF.
Comorbidities: diabetes, hypertension, and heart failure
Specific comorbidities substantially magnify memory impairment in AF patients. Heart failure, present in 35% of persistent AF patients versus only 5% of those in sinus rhythm, creates a hazardous combination. The coexistence of AF and heart failure leads to reduced cerebral perfusion, potentially creating a cycle of worsening cognitive function.
Hitherto overlooked comorbidities that warrant attention include:
- Diabetes mellitus: Associated with slightly increased dementia risk in AF patients, though this relationship appears less robust in multivariate analyses
- Hypertension: Creates a U-shaped relationship with dementia risk in AF patients, where both hypo- and hypertension raise dementia likelihood
- Chronic kidney disease: Present in 20-25% of AF patients versus 9% in those with sinus rhythm.
In essence, Cardiovascular risk factors fundamentally exert a multiplicative influence on cognitive outcomes. Having high blood pressure, diabetes, being overweight, having high cholesterol, and smoking at the same time as AF greatly increases the risk of cognitive impairment. This connection helps us understand why cognitive impairment is often linked to frailty or pre-frailty in people with AF.
In the end, this proof shows that AF-related cognitive decline is caused by more than one thing and that managing all of these risk factors is essential for keeping cognitive function.
Can AFib Treatments Help Protect Your Memory?
Emerging research points to several treatment strategies that may protect cognitive function in patients with atrial fibrillation. Evidence suggests that proper management of AFib might help mitigate associated memory loss risks through multiple mechanisms.
Oral anticoagulants and reduced dementia risk
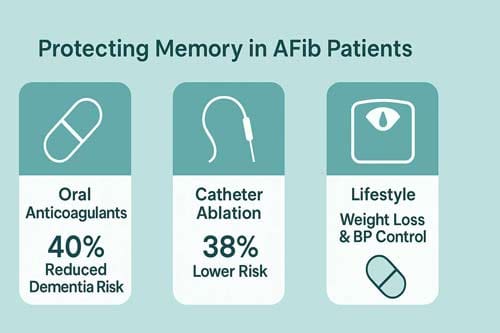
Adequate anticoagulation therapy offers significant cognitive protection for patients with AFib. Individuals on oral anticoagulants (OACs) exhibit a roughly 40% reduced risk of developing dementia in comparison to those not undergoing anticoagulation therapy. This protective effect is different for each type of medicine. Compared to vitamin K antagonists like warfarin, direct-acting oral anticoagulants (DOACs) have been shown to lower the risk of all-cause dementia by 16%. Apixaban and edoxaban seem to be the most helpful DOACs because they reduce the risk of dementia significantly compared to warfarin.
The level of protection rises with the right choice of patients. One study found that people who took DOACs had about 50% fewer cases of dementia than people who didn’t take OACs. Similarly, individuals utilising DOACs exhibited a 54% reduction in dementia risk compared to those on warfarin. Anticoagulation provides cognitive advantages even in patients without a previous history of stroke, potentially averting up to 54% of dementia cases in those with atrial fibrillation.
AFib ablation and rhythm control strategies
Rhythm control approaches show particular promise for cognitive protection. Catheter ablation, which aims to restore normal heart rhythm, correlates with improved cognitive outcomes compared to medical therapy alone. In a randomized trial, 14% of ablation patients demonstrated improved cognitive scores at 12 months versus none in the medical therapy group.
A meta-analysis involving 193,830 AFib patients found rhythm control associated with 26% lower dementia risk compared to rate control strategies. AFib ablation stands out as an efficient approach, linked to 38% reduced overall dementia risk. The cognitive benefits extend across dementia subtypes – ablation correlates with 22% lower Alzheimer’s disease risk and 42% decreased vascular dementia risk.
Lifestyle interventions: weight loss and blood pressure control
Beyond medications and procedures, lifestyle modifications offer cognitive protection. Weight management proves particularly valuable, as sustained weight loss correlates with AFib burden reduction. AFib patients who lose more than 10% of body weight become six times more likely to maintain a normal heart rhythm.
Intensive blood pressure control likewise yields cognitive benefits for AFib patients. Research demonstrates that targeted systolic blood pressure management below 120 mmHg reduces mild cognitive impairment risk regardless of diastolic pressure levels. Moreover, a clustering of healthy behaviors—including physical activity, alcohol abstinence, and smoking cessation—correlates with progressively decreasing dementia risk in AFib patients. Those following all three healthy behaviors demonstrated a 37.8% lower dementia risk compared to those following none.
These findings suggest that a comprehensive approach incorporating anticoagulation, rhythm control, and lifestyle modifications offers the best strategy for protecting memory in AFib patients.
When to Screen for Cognitive Impairment in AFib
Detecting cognitive decline early in atrial fibrillation patients requires systematic screening approaches. Recent data suggest heightened vigilance as AFib diagnosis correlates with a 45% increased risk of mild cognitive impairment (MCI).
Montreal Cognitive Assessment (MoCA) in AFib patients
The MoCA has emerged as the preferred tool for cognitive screening in AFib populations. Studies demonstrate that MoCA detects cognitive decline more effectively than the Mini-Mental State Examination (MMSE) in these patients. This superior sensitivity stems from MoCA’s comprehensive assessment of executive functions, which are particularly vulnerable to vascular cerebral damage. In patients with persistent AFib, cognitive decline can be identified with MoCA independently of the presence of heart failure or chronic kidney disease. For perspective, cognitive decline was detected via MoCA in 80% of patients with persistent AFib versus 60% with paroxysmal AFib.
Role of MRI in detecting silent infarcts
MRI remains the gold standard for identifying cerebrovascular abnormalities in AFib patients. Approximately 28% of AFib patients exhibit silent infarcts on MRI, yet only 39% of these individuals receive anticoagulation despite having CHA₂DS₂-VASc scores indicating treatment. Furthermore, 85% of new brain infarcts in anticoagulated AFib patients occur without clinical symptoms. MRI can identify cardinal features of cerebral small vessel disease that strongly correlate with current and future cognitive performance.
Who should be screened and when
According to consensus recommendations, cognitive assessment should initially target AFib patients expressing memory concerns. Subsequently, clinicians should implement a staged approach—beginning with simple screening questions about memory difficulties, followed by formal cognitive testing with MoCA when problems arise. Hence, screening for unknown AFib should be considered in patients with cognitive impairment. Generally, cognitive screening becomes particularly important for those over 74 years old, as AFib and MCI frequently coincide with multiple comorbidities at this age.
Conclusion 
The complex interplay between atrial fibrillation and cognitive decline constitutes a significant healthcare challenge necessitating increased clinical focus. Recent evidence unequivocally identifies atrial fibrillation (AFib) as an independent risk factor for memory loss and dementia via various pathophysiological mechanisms. Cerebral hypoperfusion, microemboli formation, and systemic inflammation collectively lead to progressive cognitive decline, even in individuals without clinically evident stroke.
Factors that amplify cognitive risk deserve particular consideration during patient assessment. Persistent AFib confers greater cognitive hazards than paroxysmal forms, while elevated CHA₂DS₂-VASc scores correlate strongly with accelerated cognitive decline. Furthermore, comorbidities such as heart failure, diabetes, and hypertension create multiplicative effects on cognitive outcomes, necessitating comprehensive management approaches.
Early intervention seems necessary for cognitive preservation. Anticoagulation therapy, particularly with direct-acting oral anticoagulants, exhibits significant protective effects against the onset of dementia. Similarly, rhythm control strategies, especially catheter ablation, exhibit encouraging outcomes for the preservation of cognitive function. These findings highlight the necessity of evaluating cognitive outcomes when determining treatment strategies for AFib patients.
Routine cognitive screening ought to be established as a standard procedure for the management of AFib, particularly in elderly individuals and those indicating memory-related issues. The Montreal Cognitive Assessment has a better ability to identify problems than other screening tools. However, an MRI is still necessary to find silent brain injuries that often occur before cognitive issues become apparent in a person. Early intervention appears crucial for cognitive preservation. Anticoagulation therapy, especially with direct-acting oral anticoagulants, demonstrates substantial protective effects against dementia development. Likewise, rhythm control strategies—particularly catheter ablation—show promising results for cognitive function maintenance. These findings underscore the need to consider cognitive outcomes when selecting treatment strategies for AFib patients.
Routine cognitive screening should become standard practice for AFib management, especially among elderly populations and those expressing memory concerns. The Montreal Cognitive Assessment offers superior detection sensitivity compared to traditional screening tools, while MRI remains essential for identifying silent cerebral injuries that often precede clinical cognitive manifestations.
Future research must focus on optimizing therapeutic strategies specifically targeting cognitive preservation in AFib patients. Additionally, longitudinal studies examining the impact of early rhythm control and aggressive risk factor modification on long-term cognitive trajectories will prove invaluable. Therefore, clinicians should recognize AFib not merely as a cardiac arrhythmia but as a condition with profound neurological implications requiring vigilant monitoring and proactive management to protect cognitive health.
Key Takeaways
Understanding the connection between atrial fibrillation and memory loss can help patients and healthcare providers take proactive steps to protect cognitive health.
- AFib significantly increases dementia risk – Patients with atrial fibrillation face a 39% higher risk of cognitive impairment and a 2.7-fold increased dementia risk after stroke.
- Brain damage occurs silently – AFib causes measurable brain changes, including silent infarcts, white matter damage, and reduced hippocampal volume, even without obvious stroke symptoms.
- Proper anticoagulation protects memory – Oral anticoagulants reduce dementia risk by 40%, with newer DOACs showing superior cognitive protection compared to warfarin.
- Rhythm control offers cognitive benefits – AFib ablation procedures correlate with 38% lower dementia risk and improved cognitive scores compared to rate control alone.
- Early screening is essential – The Montreal Cognitive Assessment (MoCA) effectively detects cognitive decline in AFib patients, especially those over 74 years old.
The evidence strongly supports treating AFib not just as a heart rhythm disorder, but as a condition with profound neurological implications requiring comprehensive management to preserve cognitive function throughout life.
Frequently Asked Questions:
FAQs
Q1. How does atrial fibrillation impact cognitive function? Atrial fibrillation can significantly affect cognitive function, particularly in areas of learning, memory, and executive function. Studies have shown that individuals with AFib perform worse on cognitive assessments compared to those without the condition, even in the absence of stroke.
Q2. Can treating atrial fibrillation help protect against memory loss? Yes, proper treatment of atrial fibrillation can help protect against memory loss. Oral anticoagulants, especially newer direct-acting oral anticoagulants (DOACs), have been shown to reduce dementia risk by up to 40%. Additionally, rhythm control strategies like catheter ablation are associated with improved cognitive outcomes.
Q3. What are the mechanisms by which AFib affects the brain? AFib affects the brain through multiple mechanisms, including cerebral hypoperfusion due to irregular heartbeat, microemboli formation, and systemic inflammation. These processes can lead to silent cerebral infarcts, white matter damage, and reduced brain volume, all of which contribute to cognitive decline.
Q4. Are there specific risk factors that worsen memory loss in AFib patients? Yes, several factors can exacerbate memory loss in AFib patients. These include persistent AFib (as opposed to paroxysmal), high CHA2DS2-VASc scores, and comorbidities such as diabetes, hypertension, and heart failure. The presence of these factors can significantly increase the risk of cognitive decline.
Q5. When should AFib patients be screened for cognitive impairment? Cognitive screening is recommended for AFib patients who express memory concerns, particularly those over 74 years old. The Montreal Cognitive Assessment (MoCA) is the preferred screening tool due to its superior sensitivity in detecting cognitive decline in AFib populations. Regular screening can help identify cognitive issues early, allowing for timely intervention.
References:
[1] – https://pmc.ncbi.nlm.nih.gov/articles/PMC9945461/
[2] – https://www.neurology.org/doi/10.1212/WNL.0000000000006456
[4] – https://www.cardiosmart.org/news/2015/3/weight-loss-helps-control-atrial-fibrillation
[5] – https://pmc.ncbi.nlm.nih.gov/articles/PMC7877921/
[6] – https://www.ahajournals.org/doi/10.1161/CIRCULATIONAHA.121.055018
[7] – https://www.jacc.org/doi/10.1016/j.jacadv.2023.100555
[9] – https://www.sciencedirect.com/science/article/pii/S0049384820304576
[10] – https://www.aging-us.com/article/103149/text
[11] – https://www.ahajournals.org/doi/10.1161/circ.138.suppl_1.11548
[12] – https://pubmed.ncbi.nlm.nih.gov/28577686/
[13] – https://pubmed.ncbi.nlm.nih.gov/30611392/
[14] – https://www.ahajournals.org/doi/10.1161/CIRCEP.121.010462
[15] – https://www.jacc.org/doi/10.1016/j.jacc.2015.11.064
[17] – https://pmc.ncbi.nlm.nih.gov/articles/PMC7533123/
[18] – https://pmc.ncbi.nlm.nih.gov/articles/PMC11839768/
[19] – https://www.frontiersin.org/journals/cardiovascular-medicine/articles/10.3389/fcvm.2025.1527802/full
[20] – https://www.ahajournals.org/doi/10.1161/JAHA.121.023098
[21] – https://pubmed.ncbi.nlm.nih.gov/37227345/
[22] – https://pubmed.ncbi.nlm.nih.gov/38369630/
[24] – https://pmc.ncbi.nlm.nih.gov/articles/PMC9075438/
[26] – https://pmc.ncbi.nlm.nih.gov/articles/PMC9550332/
[27] – https://www.hrsonline.org/news/hrs2020science-lbct-silent-brain-lesions/

